SAVE 40% ON SINGLE IMAGE PURCHASES - OFFER ENDS SOON, USE CODE: IMAGESAVE40%
Page 1 of 1
Manganese dioxide Stock Photos and Images
 Lessons in practical electricity; principles, experiments, and arithmetical problems, an elementary text-book . Fig. 89.—Elements of Carbon Cylinder Cell with Depolarizer.Zinc, manganese dioxide in a porous cup and sal-ammoniac solution. manganese and then sealed in. The zinc rod is prevented fromtouching the carbon by being first inserted through a porce-lain insulator. About 4 to 6 ounces of sal-ammoniac are gen-erally used for cells of ordinary size. The salt is placed inthe jar, water poured in until it is abouttwo-thirds full, and then stirred till all thesalt is dissolved. When the carbo Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/lessons-in-practical-electricity-principles-experiments-and-arithmetical-problems-an-elementary-text-book-fig-89elements-of-carbon-cylinder-cell-with-depolarizerzinc-manganese-dioxide-in-a-porous-cup-and-sal-ammoniac-solution-manganese-and-then-sealed-in-the-zinc-rod-is-prevented-fromtouching-the-carbon-by-being-first-inserted-through-a-porce-lain-insulator-about-4-to-6-ounces-of-sal-ammoniac-are-gen-erally-used-for-cells-of-ordinary-size-the-salt-is-placed-inthe-jar-water-poured-in-until-it-is-abouttwo-thirds-full-and-then-stirred-till-all-thesalt-is-dissolved-when-the-carbo-image340071277.html
Lessons in practical electricity; principles, experiments, and arithmetical problems, an elementary text-book . Fig. 89.—Elements of Carbon Cylinder Cell with Depolarizer.Zinc, manganese dioxide in a porous cup and sal-ammoniac solution. manganese and then sealed in. The zinc rod is prevented fromtouching the carbon by being first inserted through a porce-lain insulator. About 4 to 6 ounces of sal-ammoniac are gen-erally used for cells of ordinary size. The salt is placed inthe jar, water poured in until it is abouttwo-thirds full, and then stirred till all thesalt is dissolved. When the carbo Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/lessons-in-practical-electricity-principles-experiments-and-arithmetical-problems-an-elementary-text-book-fig-89elements-of-carbon-cylinder-cell-with-depolarizerzinc-manganese-dioxide-in-a-porous-cup-and-sal-ammoniac-solution-manganese-and-then-sealed-in-the-zinc-rod-is-prevented-fromtouching-the-carbon-by-being-first-inserted-through-a-porce-lain-insulator-about-4-to-6-ounces-of-sal-ammoniac-are-gen-erally-used-for-cells-of-ordinary-size-the-salt-is-placed-inthe-jar-water-poured-in-until-it-is-abouttwo-thirds-full-and-then-stirred-till-all-thesalt-is-dissolved-when-the-carbo-image340071277.htmlRM2AN7GAN–Lessons in practical electricity; principles, experiments, and arithmetical problems, an elementary text-book . Fig. 89.—Elements of Carbon Cylinder Cell with Depolarizer.Zinc, manganese dioxide in a porous cup and sal-ammoniac solution. manganese and then sealed in. The zinc rod is prevented fromtouching the carbon by being first inserted through a porce-lain insulator. About 4 to 6 ounces of sal-ammoniac are gen-erally used for cells of ordinary size. The salt is placed inthe jar, water poured in until it is abouttwo-thirds full, and then stirred till all thesalt is dissolved. When the carbo
 Lessons in practical electricity; principles, experiments, and arithmetical problems, an elementary text-book . Fig. 89.—Elements of Carbon Cylinder Cell with Depolarizer.Zinc, manganese dioxide in a porous cup and sal-ammoniac solution. manganese and then sealed in. The zinc rod is prevented fromtouching the carbon by being first inserted through a porce-lain insulator. About 4 to 6 ounces of sal-ammoniac are gen-erally used for cells of ordinary size. The salt is placed inthe jar, water poured in until it is abouttwo-thirds full, and then stirred till all thesalt is dissolved. When the carbo Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/lessons-in-practical-electricity-principles-experiments-and-arithmetical-problems-an-elementary-text-book-fig-89elements-of-carbon-cylinder-cell-with-depolarizerzinc-manganese-dioxide-in-a-porous-cup-and-sal-ammoniac-solution-manganese-and-then-sealed-in-the-zinc-rod-is-prevented-fromtouching-the-carbon-by-being-first-inserted-through-a-porce-lain-insulator-about-4-to-6-ounces-of-sal-ammoniac-are-gen-erally-used-for-cells-of-ordinary-size-the-salt-is-placed-inthe-jar-water-poured-in-until-it-is-abouttwo-thirds-full-and-then-stirred-till-all-thesalt-is-dissolved-when-the-carbo-image340071951.html
Lessons in practical electricity; principles, experiments, and arithmetical problems, an elementary text-book . Fig. 89.—Elements of Carbon Cylinder Cell with Depolarizer.Zinc, manganese dioxide in a porous cup and sal-ammoniac solution. manganese and then sealed in. The zinc rod is prevented fromtouching the carbon by being first inserted through a porce-lain insulator. About 4 to 6 ounces of sal-ammoniac are gen-erally used for cells of ordinary size. The salt is placed inthe jar, water poured in until it is abouttwo-thirds full, and then stirred till all thesalt is dissolved. When the carbo Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/lessons-in-practical-electricity-principles-experiments-and-arithmetical-problems-an-elementary-text-book-fig-89elements-of-carbon-cylinder-cell-with-depolarizerzinc-manganese-dioxide-in-a-porous-cup-and-sal-ammoniac-solution-manganese-and-then-sealed-in-the-zinc-rod-is-prevented-fromtouching-the-carbon-by-being-first-inserted-through-a-porce-lain-insulator-about-4-to-6-ounces-of-sal-ammoniac-are-gen-erally-used-for-cells-of-ordinary-size-the-salt-is-placed-inthe-jar-water-poured-in-until-it-is-abouttwo-thirds-full-and-then-stirred-till-all-thesalt-is-dissolved-when-the-carbo-image340071951.htmlRM2AN7H6R–Lessons in practical electricity; principles, experiments, and arithmetical problems, an elementary text-book . Fig. 89.—Elements of Carbon Cylinder Cell with Depolarizer.Zinc, manganese dioxide in a porous cup and sal-ammoniac solution. manganese and then sealed in. The zinc rod is prevented fromtouching the carbon by being first inserted through a porce-lain insulator. About 4 to 6 ounces of sal-ammoniac are gen-erally used for cells of ordinary size. The salt is placed inthe jar, water poured in until it is abouttwo-thirds full, and then stirred till all thesalt is dissolved. When the carbo
 The earth and its inhabitants .. . s, and which will one day afford direct access to the Rio Brancoand the Amazons ; after the heavy rains the sources of an affluent of the Caronicommunicate across the low water-parting with the Cuyuni, which belongs to theEssequibo basin. Some granite blocks, blackened by manganese dioxide, are THE ORINOCO. 95 strewn along the banks of the river as far as the augostiira, or narrows,which formerly gave their name to the capital of the lower Orinoco, 260 milesfrom the Atlantic. The tides ascend to this place, but are not strong enough tostem the current, which Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-earth-and-its-inhabitants-s-and-which-will-one-day-afford-direct-access-to-the-rio-brancoand-the-amazons-after-the-heavy-rains-the-sources-of-an-affluent-of-the-caronicommunicate-across-the-low-water-parting-with-the-cuyuni-which-belongs-to-theessequibo-basin-some-granite-blocks-blackened-by-manganese-dioxide-are-the-orinoco-95-strewn-along-the-banks-of-the-river-as-far-as-the-augostiira-or-narrowswhich-formerly-gave-their-name-to-the-capital-of-the-lower-orinoco-260-milesfrom-the-atlantic-the-tides-ascend-to-this-place-but-are-not-strong-enough-tostem-the-current-which-image343175823.html
The earth and its inhabitants .. . s, and which will one day afford direct access to the Rio Brancoand the Amazons ; after the heavy rains the sources of an affluent of the Caronicommunicate across the low water-parting with the Cuyuni, which belongs to theEssequibo basin. Some granite blocks, blackened by manganese dioxide, are THE ORINOCO. 95 strewn along the banks of the river as far as the augostiira, or narrows,which formerly gave their name to the capital of the lower Orinoco, 260 milesfrom the Atlantic. The tides ascend to this place, but are not strong enough tostem the current, which Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-earth-and-its-inhabitants-s-and-which-will-one-day-afford-direct-access-to-the-rio-brancoand-the-amazons-after-the-heavy-rains-the-sources-of-an-affluent-of-the-caronicommunicate-across-the-low-water-parting-with-the-cuyuni-which-belongs-to-theessequibo-basin-some-granite-blocks-blackened-by-manganese-dioxide-are-the-orinoco-95-strewn-along-the-banks-of-the-river-as-far-as-the-augostiira-or-narrowswhich-formerly-gave-their-name-to-the-capital-of-the-lower-orinoco-260-milesfrom-the-atlantic-the-tides-ascend-to-this-place-but-are-not-strong-enough-tostem-the-current-which-image343175823.htmlRM2AX907B–The earth and its inhabitants .. . s, and which will one day afford direct access to the Rio Brancoand the Amazons ; after the heavy rains the sources of an affluent of the Caronicommunicate across the low water-parting with the Cuyuni, which belongs to theEssequibo basin. Some granite blocks, blackened by manganese dioxide, are THE ORINOCO. 95 strewn along the banks of the river as far as the augostiira, or narrows,which formerly gave their name to the capital of the lower Orinoco, 260 milesfrom the Atlantic. The tides ascend to this place, but are not strong enough tostem the current, which
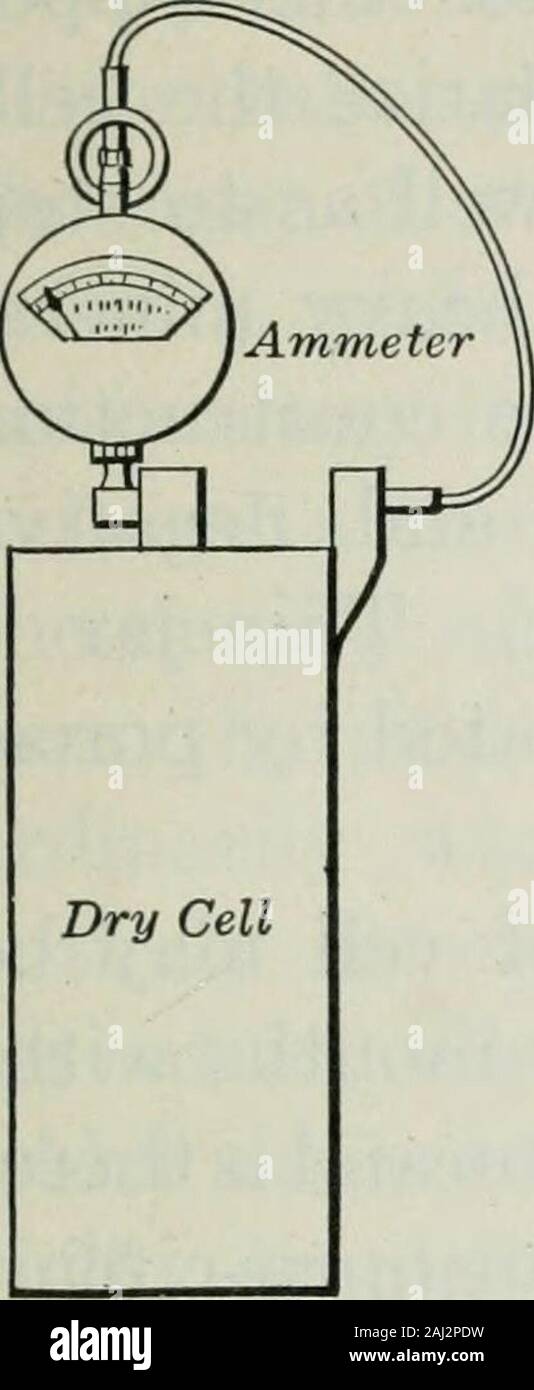 An elementary book on electricity and magnetism and their applications . of pulp board or blottingpaper which is saturated with asolution of sal ammoniac and zincchloride. The zinc chloride isnecessary to reduce the rapid de-terioration which would otherwisetake place on open circuit. Thespace between the lining and the carbon electrode is filled witha mixture of granulated carbon and manganese dioxide. Thislatter is used as the depolarizer. The top of the cell is gener-ally sealed up with a pitch composition. This cell has a volt-age of about 1.5 volts and will polarize rapidly if kept long o Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/an-elementary-book-on-electricity-and-magnetism-and-their-applications-of-pulp-board-or-blottingpaper-which-is-saturated-with-asolution-of-sal-ammoniac-and-zincchloride-the-zinc-chloride-isnecessary-to-reduce-the-rapid-de-terioration-which-would-otherwisetake-place-on-open-circuit-thespace-between-the-lining-and-the-carbon-electrode-is-filled-witha-mixture-of-granulated-carbon-and-manganese-dioxide-thislatter-is-used-as-the-depolarizer-the-top-of-the-cell-is-gener-ally-sealed-up-with-a-pitch-composition-this-cell-has-a-volt-age-of-about-15-volts-and-will-polarize-rapidly-if-kept-long-o-image338122341.html
An elementary book on electricity and magnetism and their applications . of pulp board or blottingpaper which is saturated with asolution of sal ammoniac and zincchloride. The zinc chloride isnecessary to reduce the rapid de-terioration which would otherwisetake place on open circuit. Thespace between the lining and the carbon electrode is filled witha mixture of granulated carbon and manganese dioxide. Thislatter is used as the depolarizer. The top of the cell is gener-ally sealed up with a pitch composition. This cell has a volt-age of about 1.5 volts and will polarize rapidly if kept long o Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/an-elementary-book-on-electricity-and-magnetism-and-their-applications-of-pulp-board-or-blottingpaper-which-is-saturated-with-asolution-of-sal-ammoniac-and-zincchloride-the-zinc-chloride-isnecessary-to-reduce-the-rapid-de-terioration-which-would-otherwisetake-place-on-open-circuit-thespace-between-the-lining-and-the-carbon-electrode-is-filled-witha-mixture-of-granulated-carbon-and-manganese-dioxide-thislatter-is-used-as-the-depolarizer-the-top-of-the-cell-is-gener-ally-sealed-up-with-a-pitch-composition-this-cell-has-a-volt-age-of-about-15-volts-and-will-polarize-rapidly-if-kept-long-o-image338122341.htmlRM2AJ2PDW–An elementary book on electricity and magnetism and their applications . of pulp board or blottingpaper which is saturated with asolution of sal ammoniac and zincchloride. The zinc chloride isnecessary to reduce the rapid de-terioration which would otherwisetake place on open circuit. Thespace between the lining and the carbon electrode is filled witha mixture of granulated carbon and manganese dioxide. Thislatter is used as the depolarizer. The top of the cell is gener-ally sealed up with a pitch composition. This cell has a volt-age of about 1.5 volts and will polarize rapidly if kept long o
 An essay on the history of electrotherapy and diagnosis; . Mayer and Meltzers portable battery and coil.. Althaus portable battery. manganese dioxide, two-thirds of the tube are filled with asaturated solution of ammonium chloride, and the upper partof the inside of the tube is coated with tallow to prevent the 72 AN ESSAY ON THE Contained liquid creeping up and crystallising. The upperpart is closed with a vulcanite cover, through which passes azinc rod, while a platinum wire passing to the bottom of thetube forms the carbon pole. It was early noticed that plates of ordinary zinc whenimmersed Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/an-essay-on-the-history-of-electrotherapy-and-diagnosis-mayer-and-meltzers-portable-battery-and-coil-althaus-portable-battery-manganese-dioxide-two-thirds-of-the-tube-are-filled-with-asaturated-solution-of-ammonium-chloride-and-the-upper-partof-the-inside-of-the-tube-is-coated-with-tallow-to-prevent-the-72-an-essay-on-the-contained-liquid-creeping-up-and-crystallising-the-upperpart-is-closed-with-a-vulcanite-cover-through-which-passes-azinc-rod-while-a-platinum-wire-passing-to-the-bottom-of-thetube-forms-the-carbon-pole-it-was-early-noticed-that-plates-of-ordinary-zinc-whenimmersed-image340185970.html
An essay on the history of electrotherapy and diagnosis; . Mayer and Meltzers portable battery and coil.. Althaus portable battery. manganese dioxide, two-thirds of the tube are filled with asaturated solution of ammonium chloride, and the upper partof the inside of the tube is coated with tallow to prevent the 72 AN ESSAY ON THE Contained liquid creeping up and crystallising. The upperpart is closed with a vulcanite cover, through which passes azinc rod, while a platinum wire passing to the bottom of thetube forms the carbon pole. It was early noticed that plates of ordinary zinc whenimmersed Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/an-essay-on-the-history-of-electrotherapy-and-diagnosis-mayer-and-meltzers-portable-battery-and-coil-althaus-portable-battery-manganese-dioxide-two-thirds-of-the-tube-are-filled-with-asaturated-solution-of-ammonium-chloride-and-the-upper-partof-the-inside-of-the-tube-is-coated-with-tallow-to-prevent-the-72-an-essay-on-the-contained-liquid-creeping-up-and-crystallising-the-upperpart-is-closed-with-a-vulcanite-cover-through-which-passes-azinc-rod-while-a-platinum-wire-passing-to-the-bottom-of-thetube-forms-the-carbon-pole-it-was-early-noticed-that-plates-of-ordinary-zinc-whenimmersed-image340185970.htmlRM2ANCPJX–An essay on the history of electrotherapy and diagnosis; . Mayer and Meltzers portable battery and coil.. Althaus portable battery. manganese dioxide, two-thirds of the tube are filled with asaturated solution of ammonium chloride, and the upper partof the inside of the tube is coated with tallow to prevent the 72 AN ESSAY ON THE Contained liquid creeping up and crystallising. The upperpart is closed with a vulcanite cover, through which passes azinc rod, while a platinum wire passing to the bottom of thetube forms the carbon pole. It was early noticed that plates of ordinary zinc whenimmersed
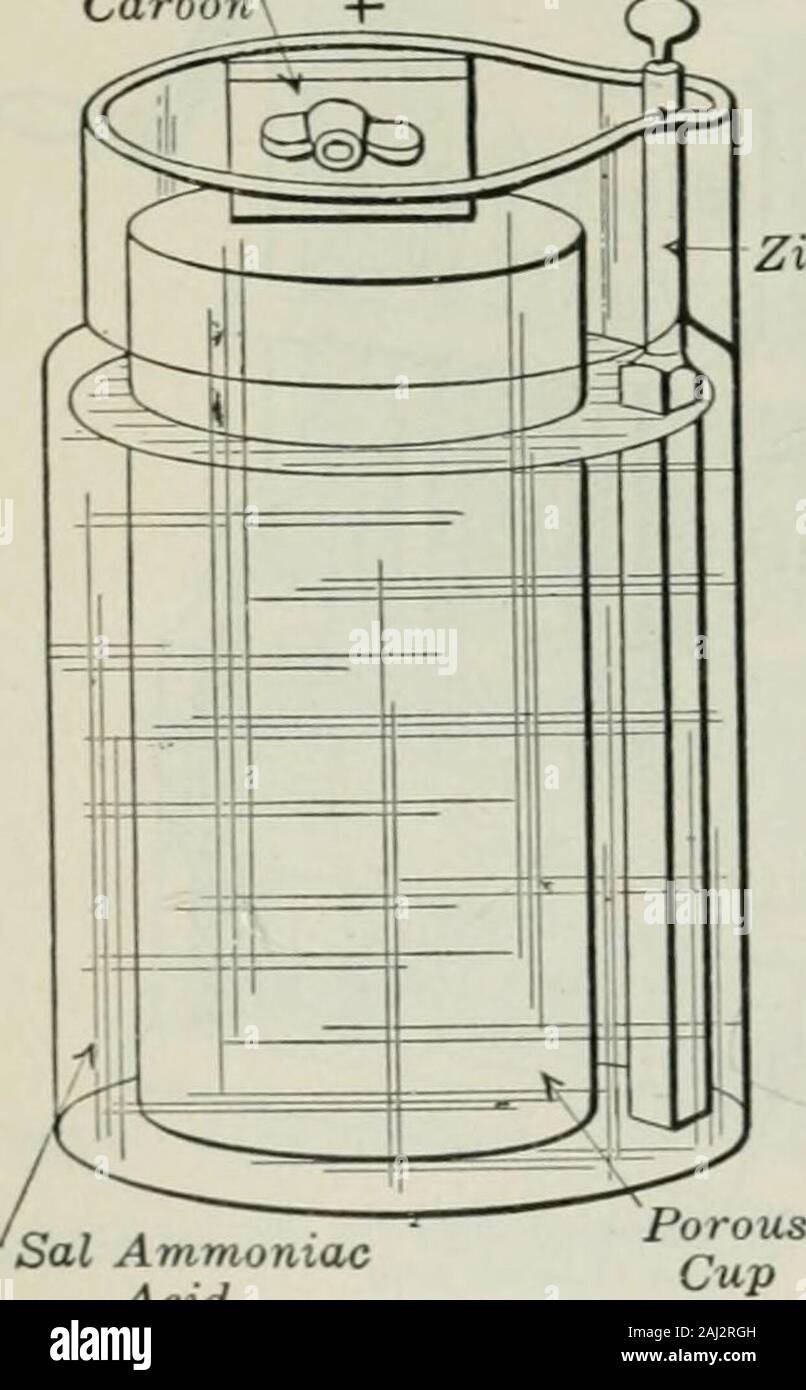 An elementary book on electricity and magnetism and their applications . j the solution ; never add the solution to theacid, as serious burns are apt to result. zinc When manganese dioxide is used asa depolarizer, it is generally brokenup into small lumps and put into aporous cup surrounding a positiveplate of carbon. When sal ammoniacdissolved in water is used as the liquidin this form of cell, it makes the famil-iar Leclanche cell (Fig. 8), which isfrequently used for ringing door bellsand doing similar service. Some-times the manganese dioxide is pul-verized and placed inside of the carbonp Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/an-elementary-book-on-electricity-and-magnetism-and-their-applications-j-the-solution-never-add-the-solution-to-theacid-as-serious-burns-are-apt-to-result-zinc-when-manganese-dioxide-is-used-asa-depolarizer-it-is-generally-brokenup-into-small-lumps-and-put-into-aporous-cup-surrounding-a-positiveplate-of-carbon-when-sal-ammoniacdissolved-in-water-is-used-as-the-liquidin-this-form-of-cell-it-makes-the-famil-iar-leclanche-cell-fig-8-which-isfrequently-used-for-ringing-door-bellsand-doing-similar-service-some-times-the-manganese-dioxide-is-pul-verized-and-placed-inside-of-the-carbonp-image338123201.html
An elementary book on electricity and magnetism and their applications . j the solution ; never add the solution to theacid, as serious burns are apt to result. zinc When manganese dioxide is used asa depolarizer, it is generally brokenup into small lumps and put into aporous cup surrounding a positiveplate of carbon. When sal ammoniacdissolved in water is used as the liquidin this form of cell, it makes the famil-iar Leclanche cell (Fig. 8), which isfrequently used for ringing door bellsand doing similar service. Some-times the manganese dioxide is pul-verized and placed inside of the carbonp Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/an-elementary-book-on-electricity-and-magnetism-and-their-applications-j-the-solution-never-add-the-solution-to-theacid-as-serious-burns-are-apt-to-result-zinc-when-manganese-dioxide-is-used-asa-depolarizer-it-is-generally-brokenup-into-small-lumps-and-put-into-aporous-cup-surrounding-a-positiveplate-of-carbon-when-sal-ammoniacdissolved-in-water-is-used-as-the-liquidin-this-form-of-cell-it-makes-the-famil-iar-leclanche-cell-fig-8-which-isfrequently-used-for-ringing-door-bellsand-doing-similar-service-some-times-the-manganese-dioxide-is-pul-verized-and-placed-inside-of-the-carbonp-image338123201.htmlRM2AJ2RGH–An elementary book on electricity and magnetism and their applications . j the solution ; never add the solution to theacid, as serious burns are apt to result. zinc When manganese dioxide is used asa depolarizer, it is generally brokenup into small lumps and put into aporous cup surrounding a positiveplate of carbon. When sal ammoniacdissolved in water is used as the liquidin this form of cell, it makes the famil-iar Leclanche cell (Fig. 8), which isfrequently used for ringing door bellsand doing similar service. Some-times the manganese dioxide is pul-verized and placed inside of the carbonp
 An essay on the history of electrotherapy and diagnosis; . Althaus portable battery. manganese dioxide, two-thirds of the tube are filled with asaturated solution of ammonium chloride, and the upper partof the inside of the tube is coated with tallow to prevent the 72 AN ESSAY ON THE Contained liquid creeping up and crystallising. The upperpart is closed with a vulcanite cover, through which passes azinc rod, while a platinum wire passing to the bottom of thetube forms the carbon pole. It was early noticed that plates of ordinary zinc whenimmersed in battery liquids became corroded, even thoug Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/an-essay-on-the-history-of-electrotherapy-and-diagnosis-althaus-portable-battery-manganese-dioxide-two-thirds-of-the-tube-are-filled-with-asaturated-solution-of-ammonium-chloride-and-the-upper-partof-the-inside-of-the-tube-is-coated-with-tallow-to-prevent-the-72-an-essay-on-the-contained-liquid-creeping-up-and-crystallising-the-upperpart-is-closed-with-a-vulcanite-cover-through-which-passes-azinc-rod-while-a-platinum-wire-passing-to-the-bottom-of-thetube-forms-the-carbon-pole-it-was-early-noticed-that-plates-of-ordinary-zinc-whenimmersed-in-battery-liquids-became-corroded-even-thoug-image340185673.html
An essay on the history of electrotherapy and diagnosis; . Althaus portable battery. manganese dioxide, two-thirds of the tube are filled with asaturated solution of ammonium chloride, and the upper partof the inside of the tube is coated with tallow to prevent the 72 AN ESSAY ON THE Contained liquid creeping up and crystallising. The upperpart is closed with a vulcanite cover, through which passes azinc rod, while a platinum wire passing to the bottom of thetube forms the carbon pole. It was early noticed that plates of ordinary zinc whenimmersed in battery liquids became corroded, even thoug Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/an-essay-on-the-history-of-electrotherapy-and-diagnosis-althaus-portable-battery-manganese-dioxide-two-thirds-of-the-tube-are-filled-with-asaturated-solution-of-ammonium-chloride-and-the-upper-partof-the-inside-of-the-tube-is-coated-with-tallow-to-prevent-the-72-an-essay-on-the-contained-liquid-creeping-up-and-crystallising-the-upperpart-is-closed-with-a-vulcanite-cover-through-which-passes-azinc-rod-while-a-platinum-wire-passing-to-the-bottom-of-thetube-forms-the-carbon-pole-it-was-early-noticed-that-plates-of-ordinary-zinc-whenimmersed-in-battery-liquids-became-corroded-even-thoug-image340185673.htmlRM2ANCP89–An essay on the history of electrotherapy and diagnosis; . Althaus portable battery. manganese dioxide, two-thirds of the tube are filled with asaturated solution of ammonium chloride, and the upper partof the inside of the tube is coated with tallow to prevent the 72 AN ESSAY ON THE Contained liquid creeping up and crystallising. The upperpart is closed with a vulcanite cover, through which passes azinc rod, while a platinum wire passing to the bottom of thetube forms the carbon pole. It was early noticed that plates of ordinary zinc whenimmersed in battery liquids became corroded, even thoug
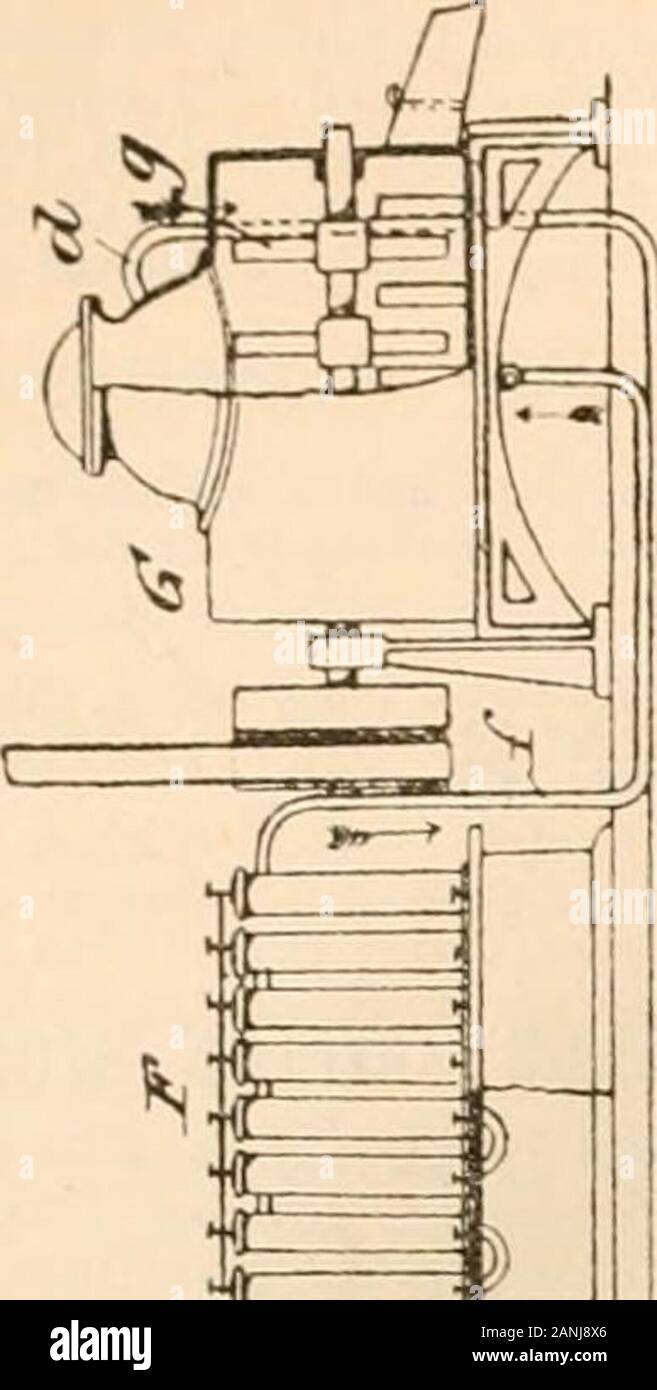 Journal . einventors chlor-ozone, obtained by electrifying a mixtureof chlorine and oxygen. The oxygen is contained in thereceiver -. The chlorine is generated in the retort B bythe action of hydrochloric acid on manganese dioxide. Themixed gases pass by the pipe n^ into the wash-bottle 6, andthence by the pipe 6- into the gas-holder C. They are thenmade to pass by the pipe c^ into the ozone generator F,where they are converted into chlor-ozone. E- representsthe battery and E the induction coil for producing thecurrent. The electrified gases then pass into the vessel G,in which the bleaching Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/journal-einventors-chlor-ozone-obtained-by-electrifying-a-mixtureof-chlorine-and-oxygen-the-oxygen-is-contained-in-thereceiver-the-chlorine-is-generated-in-the-retort-b-bythe-action-of-hydrochloric-acid-on-manganese-dioxide-themixed-gases-pass-by-the-pipe-n-into-the-wash-bottle-6-andthence-by-the-pipe-6-into-the-gas-holder-c-they-are-thenmade-to-pass-by-the-pipe-c-into-the-ozone-generator-fwhere-they-are-converted-into-chlor-ozone-e-representsthe-battery-and-e-the-induction-coil-for-producing-thecurrent-the-electrified-gases-then-pass-into-the-vessel-gin-which-the-bleaching-image340306910.html
Journal . einventors chlor-ozone, obtained by electrifying a mixtureof chlorine and oxygen. The oxygen is contained in thereceiver -. The chlorine is generated in the retort B bythe action of hydrochloric acid on manganese dioxide. Themixed gases pass by the pipe n^ into the wash-bottle 6, andthence by the pipe 6- into the gas-holder C. They are thenmade to pass by the pipe c^ into the ozone generator F,where they are converted into chlor-ozone. E- representsthe battery and E the induction coil for producing thecurrent. The electrified gases then pass into the vessel G,in which the bleaching Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/journal-einventors-chlor-ozone-obtained-by-electrifying-a-mixtureof-chlorine-and-oxygen-the-oxygen-is-contained-in-thereceiver-the-chlorine-is-generated-in-the-retort-b-bythe-action-of-hydrochloric-acid-on-manganese-dioxide-themixed-gases-pass-by-the-pipe-n-into-the-wash-bottle-6-andthence-by-the-pipe-6-into-the-gas-holder-c-they-are-thenmade-to-pass-by-the-pipe-c-into-the-ozone-generator-fwhere-they-are-converted-into-chlor-ozone-e-representsthe-battery-and-e-the-induction-coil-for-producing-thecurrent-the-electrified-gases-then-pass-into-the-vessel-gin-which-the-bleaching-image340306910.htmlRM2ANJ8X6–Journal . einventors chlor-ozone, obtained by electrifying a mixtureof chlorine and oxygen. The oxygen is contained in thereceiver -. The chlorine is generated in the retort B bythe action of hydrochloric acid on manganese dioxide. Themixed gases pass by the pipe n^ into the wash-bottle 6, andthence by the pipe 6- into the gas-holder C. They are thenmade to pass by the pipe c^ into the ozone generator F,where they are converted into chlor-ozone. E- representsthe battery and E the induction coil for producing thecurrent. The electrified gases then pass into the vessel G,in which the bleaching
 ..Debris . cular records I. SIMOX HOMER WE.WEK, CovinktTau Beta Pi; Eleclrical En- n. -:i. -Thesis; Power RequirLiI ! Because ol his unassunnn.the junior partner o( a t-r. i i ,.rr:iwriter for the leading mag.w ihlv ol lUi.do not hesitate to predict unlimited M)ci OWEN CLIVE HAWOKTH. ?sis ; Action ol Manganese Dioxide on Oxidizing Agents. • of the veteran school teachers of the class. He is older than he looks, and his ling manner and frequent bursts of wit make it all the more clear that he Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/debris-cular-records-i-simox-homer-wewek-covinkttau-beta-pi-eleclrical-en-n-i-thesis-power-requirlii-!-because-ol-his-unassunnnthe-junior-partner-o-a-t-r-i-i-rriwriter-for-the-leading-magw-ihlv-ol-luido-not-hesitate-to-predict-unlimited-mci-owen-clive-hawokth-sis-action-ol-manganese-dioxide-on-oxidizing-agents-of-the-veteran-school-teachers-of-the-class-he-is-older-than-he-looks-and-his-ling-manner-and-frequent-bursts-of-wit-make-it-all-the-more-clear-that-he-image342752333.html
..Debris . cular records I. SIMOX HOMER WE.WEK, CovinktTau Beta Pi; Eleclrical En- n. -:i. -Thesis; Power RequirLiI ! Because ol his unassunnn.the junior partner o( a t-r. i i ,.rr:iwriter for the leading mag.w ihlv ol lUi.do not hesitate to predict unlimited M)ci OWEN CLIVE HAWOKTH. ?sis ; Action ol Manganese Dioxide on Oxidizing Agents. • of the veteran school teachers of the class. He is older than he looks, and his ling manner and frequent bursts of wit make it all the more clear that he Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/debris-cular-records-i-simox-homer-wewek-covinkttau-beta-pi-eleclrical-en-n-i-thesis-power-requirlii-!-because-ol-his-unassunnnthe-junior-partner-o-a-t-r-i-i-rriwriter-for-the-leading-magw-ihlv-ol-luido-not-hesitate-to-predict-unlimited-mci-owen-clive-hawokth-sis-action-ol-manganese-dioxide-on-oxidizing-agents-of-the-veteran-school-teachers-of-the-class-he-is-older-than-he-looks-and-his-ling-manner-and-frequent-bursts-of-wit-make-it-all-the-more-clear-that-he-image342752333.htmlRM2AWHM2N–..Debris . cular records I. SIMOX HOMER WE.WEK, CovinktTau Beta Pi; Eleclrical En- n. -:i. -Thesis; Power RequirLiI ! Because ol his unassunnn.the junior partner o( a t-r. i i ,.rr:iwriter for the leading mag.w ihlv ol lUi.do not hesitate to predict unlimited M)ci OWEN CLIVE HAWOKTH. ?sis ; Action ol Manganese Dioxide on Oxidizing Agents. • of the veteran school teachers of the class. He is older than he looks, and his ling manner and frequent bursts of wit make it all the more clear that he
 An international system of electro-therapeutics : for students, general practitioners, and specialists . Fig. 72. remaining space is filled with manganese ore (manganese dioxide) andpieces of carbon. A platinum wire, P, connects the carbon with the zinc A-280 BLEYER. piece, S, which represents one of the poles of the element. A solution ofsal ammoniac is used. The manganese ore and carbon pieces are sepa-rated from each other by a layer of asphalt. A number of elementsjoined up to form a battery are shown in Fig. 72. The J. Kidder Manufacturing Company makes also a very suitableportable galvan Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/an-international-system-of-electro-therapeutics-for-students-general-practitioners-and-specialists-fig-72-remaining-space-is-filled-with-manganese-ore-manganese-dioxide-andpieces-of-carbon-a-platinum-wire-p-connects-the-carbon-with-the-zinc-a-280-bleyer-piece-s-which-represents-one-of-the-poles-of-the-element-a-solution-ofsal-ammoniac-is-used-the-manganese-ore-and-carbon-pieces-are-sepa-rated-from-each-other-by-a-layer-of-asphalt-a-number-of-elementsjoined-up-to-form-a-battery-are-shown-in-fig-72-the-j-kidder-manufacturing-company-makes-also-a-very-suitableportable-galvan-image339013629.html
An international system of electro-therapeutics : for students, general practitioners, and specialists . Fig. 72. remaining space is filled with manganese ore (manganese dioxide) andpieces of carbon. A platinum wire, P, connects the carbon with the zinc A-280 BLEYER. piece, S, which represents one of the poles of the element. A solution ofsal ammoniac is used. The manganese ore and carbon pieces are sepa-rated from each other by a layer of asphalt. A number of elementsjoined up to form a battery are shown in Fig. 72. The J. Kidder Manufacturing Company makes also a very suitableportable galvan Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/an-international-system-of-electro-therapeutics-for-students-general-practitioners-and-specialists-fig-72-remaining-space-is-filled-with-manganese-ore-manganese-dioxide-andpieces-of-carbon-a-platinum-wire-p-connects-the-carbon-with-the-zinc-a-280-bleyer-piece-s-which-represents-one-of-the-poles-of-the-element-a-solution-ofsal-ammoniac-is-used-the-manganese-ore-and-carbon-pieces-are-sepa-rated-from-each-other-by-a-layer-of-asphalt-a-number-of-elementsjoined-up-to-form-a-battery-are-shown-in-fig-72-the-j-kidder-manufacturing-company-makes-also-a-very-suitableportable-galvan-image339013629.htmlRM2AKFB9H–An international system of electro-therapeutics : for students, general practitioners, and specialists . Fig. 72. remaining space is filled with manganese ore (manganese dioxide) andpieces of carbon. A platinum wire, P, connects the carbon with the zinc A-280 BLEYER. piece, S, which represents one of the poles of the element. A solution ofsal ammoniac is used. The manganese ore and carbon pieces are sepa-rated from each other by a layer of asphalt. A number of elementsjoined up to form a battery are shown in Fig. 72. The J. Kidder Manufacturing Company makes also a very suitableportable galvan
 . American journal of pharmacy. of chlorine: i Cc. N/ioNaoSoOg = 0.003546 Gm. CI. When a liquid is to be an-alyzed, the manganese dioxideis simply added to the samplein the flask. If the chlorine in fluoridesis to be determined the pro-cedure is carried out in thesame way as for the other samples. Some HF will be evolved, but it has no effect upon thefinal results and its action upon the glass is negligible. If the sulphuric acid is free from nitric and hydrochloric acids,chlorides, nitrates and nitrous fumes, and the manganese dioxide con-tains no impurities capable of decomposing KI on volat Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/american-journal-of-pharmacy-of-chlorine-i-cc-nionaosoog-=-0003546-gm-ci-when-a-liquid-is-to-be-an-alyzed-the-manganese-dioxideis-simply-added-to-the-samplein-the-flask-if-the-chlorine-in-fluoridesis-to-be-determined-the-pro-cedure-is-carried-out-in-thesame-way-as-for-the-other-samples-some-hf-will-be-evolved-but-it-has-no-effect-upon-thefinal-results-and-its-action-upon-the-glass-is-negligible-if-the-sulphuric-acid-is-free-from-nitric-and-hydrochloric-acidschlorides-nitrates-and-nitrous-fumes-and-the-manganese-dioxide-con-tains-no-impurities-capable-of-decomposing-ki-on-volat-image336998529.html
. American journal of pharmacy. of chlorine: i Cc. N/ioNaoSoOg = 0.003546 Gm. CI. When a liquid is to be an-alyzed, the manganese dioxideis simply added to the samplein the flask. If the chlorine in fluoridesis to be determined the pro-cedure is carried out in thesame way as for the other samples. Some HF will be evolved, but it has no effect upon thefinal results and its action upon the glass is negligible. If the sulphuric acid is free from nitric and hydrochloric acids,chlorides, nitrates and nitrous fumes, and the manganese dioxide con-tains no impurities capable of decomposing KI on volat Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/american-journal-of-pharmacy-of-chlorine-i-cc-nionaosoog-=-0003546-gm-ci-when-a-liquid-is-to-be-an-alyzed-the-manganese-dioxideis-simply-added-to-the-samplein-the-flask-if-the-chlorine-in-fluoridesis-to-be-determined-the-pro-cedure-is-carried-out-in-thesame-way-as-for-the-other-samples-some-hf-will-be-evolved-but-it-has-no-effect-upon-thefinal-results-and-its-action-upon-the-glass-is-negligible-if-the-sulphuric-acid-is-free-from-nitric-and-hydrochloric-acidschlorides-nitrates-and-nitrous-fumes-and-the-manganese-dioxide-con-tains-no-impurities-capable-of-decomposing-ki-on-volat-image336998529.htmlRM2AG7H1N–. American journal of pharmacy. of chlorine: i Cc. N/ioNaoSoOg = 0.003546 Gm. CI. When a liquid is to be an-alyzed, the manganese dioxideis simply added to the samplein the flask. If the chlorine in fluoridesis to be determined the pro-cedure is carried out in thesame way as for the other samples. Some HF will be evolved, but it has no effect upon thefinal results and its action upon the glass is negligible. If the sulphuric acid is free from nitric and hydrochloric acids,chlorides, nitrates and nitrous fumes, and the manganese dioxide con-tains no impurities capable of decomposing KI on volat
 An international system of electro-therapeutics : for students, general practitioners, and specialists . Fig. 71.—Complete Celi,. The cell, B B,s made of gutta-percha, and contains a gutta-perchacylinder, G Z^ which holds the zinc rod. K is the carbon block; the. Fig. 72. remaining space is filled with manganese ore (manganese dioxide) andpieces of carbon. A platinum wire, P, connects the carbon with the zinc A-280 BLEYER. piece, S, which represents one of the poles of the element. A solution ofsal ammoniac is used. The manganese ore and carbon pieces are sepa-rated from each other by a layer Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/an-international-system-of-electro-therapeutics-for-students-general-practitioners-and-specialists-fig-71complete-celi-the-cell-b-bs-made-of-gutta-percha-and-contains-a-gutta-perchacylinder-g-z-which-holds-the-zinc-rod-k-is-the-carbon-block-the-fig-72-remaining-space-is-filled-with-manganese-ore-manganese-dioxide-andpieces-of-carbon-a-platinum-wire-p-connects-the-carbon-with-the-zinc-a-280-bleyer-piece-s-which-represents-one-of-the-poles-of-the-element-a-solution-ofsal-ammoniac-is-used-the-manganese-ore-and-carbon-pieces-are-sepa-rated-from-each-other-by-a-layer-image339013962.html
An international system of electro-therapeutics : for students, general practitioners, and specialists . Fig. 71.—Complete Celi,. The cell, B B,s made of gutta-percha, and contains a gutta-perchacylinder, G Z^ which holds the zinc rod. K is the carbon block; the. Fig. 72. remaining space is filled with manganese ore (manganese dioxide) andpieces of carbon. A platinum wire, P, connects the carbon with the zinc A-280 BLEYER. piece, S, which represents one of the poles of the element. A solution ofsal ammoniac is used. The manganese ore and carbon pieces are sepa-rated from each other by a layer Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/an-international-system-of-electro-therapeutics-for-students-general-practitioners-and-specialists-fig-71complete-celi-the-cell-b-bs-made-of-gutta-percha-and-contains-a-gutta-perchacylinder-g-z-which-holds-the-zinc-rod-k-is-the-carbon-block-the-fig-72-remaining-space-is-filled-with-manganese-ore-manganese-dioxide-andpieces-of-carbon-a-platinum-wire-p-connects-the-carbon-with-the-zinc-a-280-bleyer-piece-s-which-represents-one-of-the-poles-of-the-element-a-solution-ofsal-ammoniac-is-used-the-manganese-ore-and-carbon-pieces-are-sepa-rated-from-each-other-by-a-layer-image339013962.htmlRM2AKFBNE–An international system of electro-therapeutics : for students, general practitioners, and specialists . Fig. 71.—Complete Celi,. The cell, B B,s made of gutta-percha, and contains a gutta-perchacylinder, G Z^ which holds the zinc rod. K is the carbon block; the. Fig. 72. remaining space is filled with manganese ore (manganese dioxide) andpieces of carbon. A platinum wire, P, connects the carbon with the zinc A-280 BLEYER. piece, S, which represents one of the poles of the element. A solution ofsal ammoniac is used. The manganese ore and carbon pieces are sepa-rated from each other by a layer
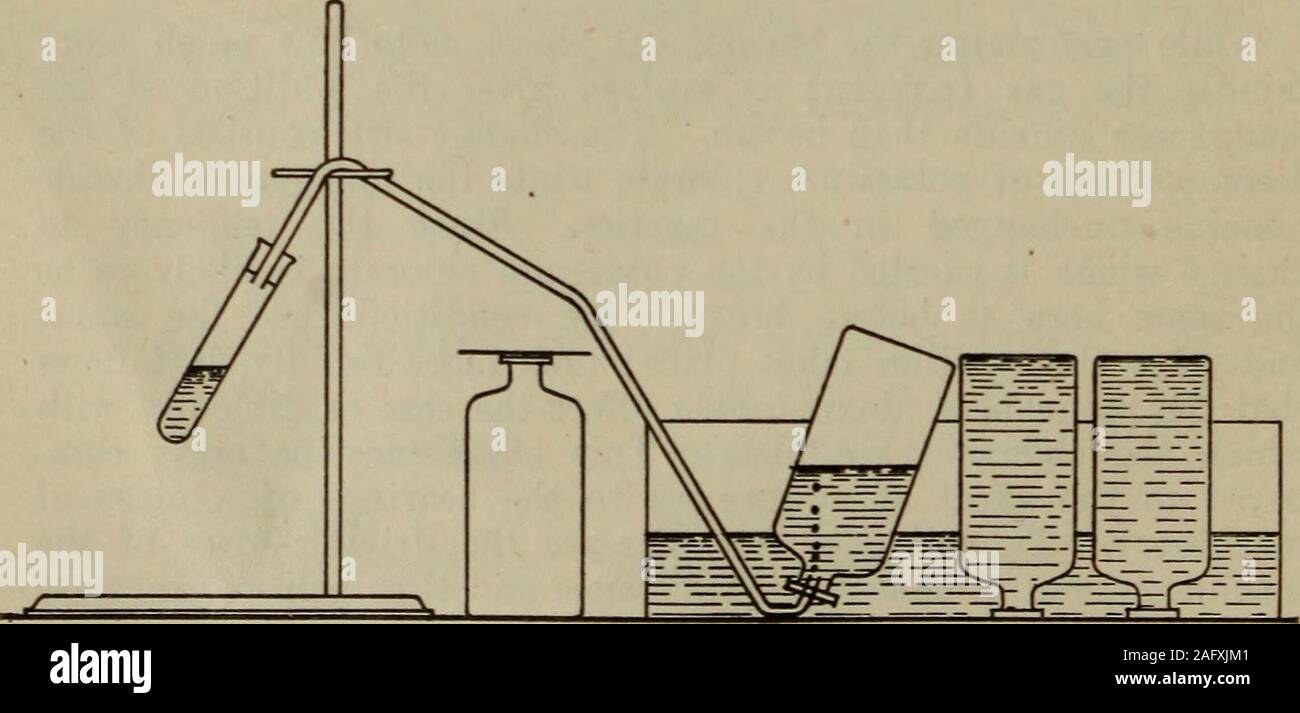 . An introductory course of lessons and exercises in chemistry (rewritten 1919). tory Chemistry mixture composed of approximately two parts of potassium chlorate andone part manganese dioxide (powdered). Be careful to keep paper,pieces of cork, and other combustibles out of this mixture. In the next operation, two students should work together. They shouldsecure a water basin, four wide-mouth bottles, and four pieces of glasslarge enough to cover the mouths of tbe bottles. Assemble the whole as ^ C G A burner for BendingGlass Tubing A Correctly Bent. B St C Incorrectly Bent- shown in the accom Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/an-introductory-course-of-lessons-and-exercises-in-chemistry-rewritten-1919-tory-chemistry-mixture-composed-of-approximately-two-parts-of-potassium-chlorate-andone-part-manganese-dioxide-powdered-be-careful-to-keep-paperpieces-of-cork-and-other-combustibles-out-of-this-mixture-in-the-next-operation-two-students-should-work-together-they-shouldsecure-a-water-basin-four-wide-mouth-bottles-and-four-pieces-of-glasslarge-enough-to-cover-the-mouths-of-tbe-bottles-assemble-the-whole-as-c-g-a-burner-for-bendingglass-tubing-a-correctly-bent-b-st-c-incorrectly-bent-shown-in-the-accom-image336802257.html
. An introductory course of lessons and exercises in chemistry (rewritten 1919). tory Chemistry mixture composed of approximately two parts of potassium chlorate andone part manganese dioxide (powdered). Be careful to keep paper,pieces of cork, and other combustibles out of this mixture. In the next operation, two students should work together. They shouldsecure a water basin, four wide-mouth bottles, and four pieces of glasslarge enough to cover the mouths of tbe bottles. Assemble the whole as ^ C G A burner for BendingGlass Tubing A Correctly Bent. B St C Incorrectly Bent- shown in the accom Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/an-introductory-course-of-lessons-and-exercises-in-chemistry-rewritten-1919-tory-chemistry-mixture-composed-of-approximately-two-parts-of-potassium-chlorate-andone-part-manganese-dioxide-powdered-be-careful-to-keep-paperpieces-of-cork-and-other-combustibles-out-of-this-mixture-in-the-next-operation-two-students-should-work-together-they-shouldsecure-a-water-basin-four-wide-mouth-bottles-and-four-pieces-of-glasslarge-enough-to-cover-the-mouths-of-tbe-bottles-assemble-the-whole-as-c-g-a-burner-for-bendingglass-tubing-a-correctly-bent-b-st-c-incorrectly-bent-shown-in-the-accom-image336802257.htmlRM2AFXJM1–. An introductory course of lessons and exercises in chemistry (rewritten 1919). tory Chemistry mixture composed of approximately two parts of potassium chlorate andone part manganese dioxide (powdered). Be careful to keep paper,pieces of cork, and other combustibles out of this mixture. In the next operation, two students should work together. They shouldsecure a water basin, four wide-mouth bottles, and four pieces of glasslarge enough to cover the mouths of tbe bottles. Assemble the whole as ^ C G A burner for BendingGlass Tubing A Correctly Bent. B St C Incorrectly Bent- shown in the accom
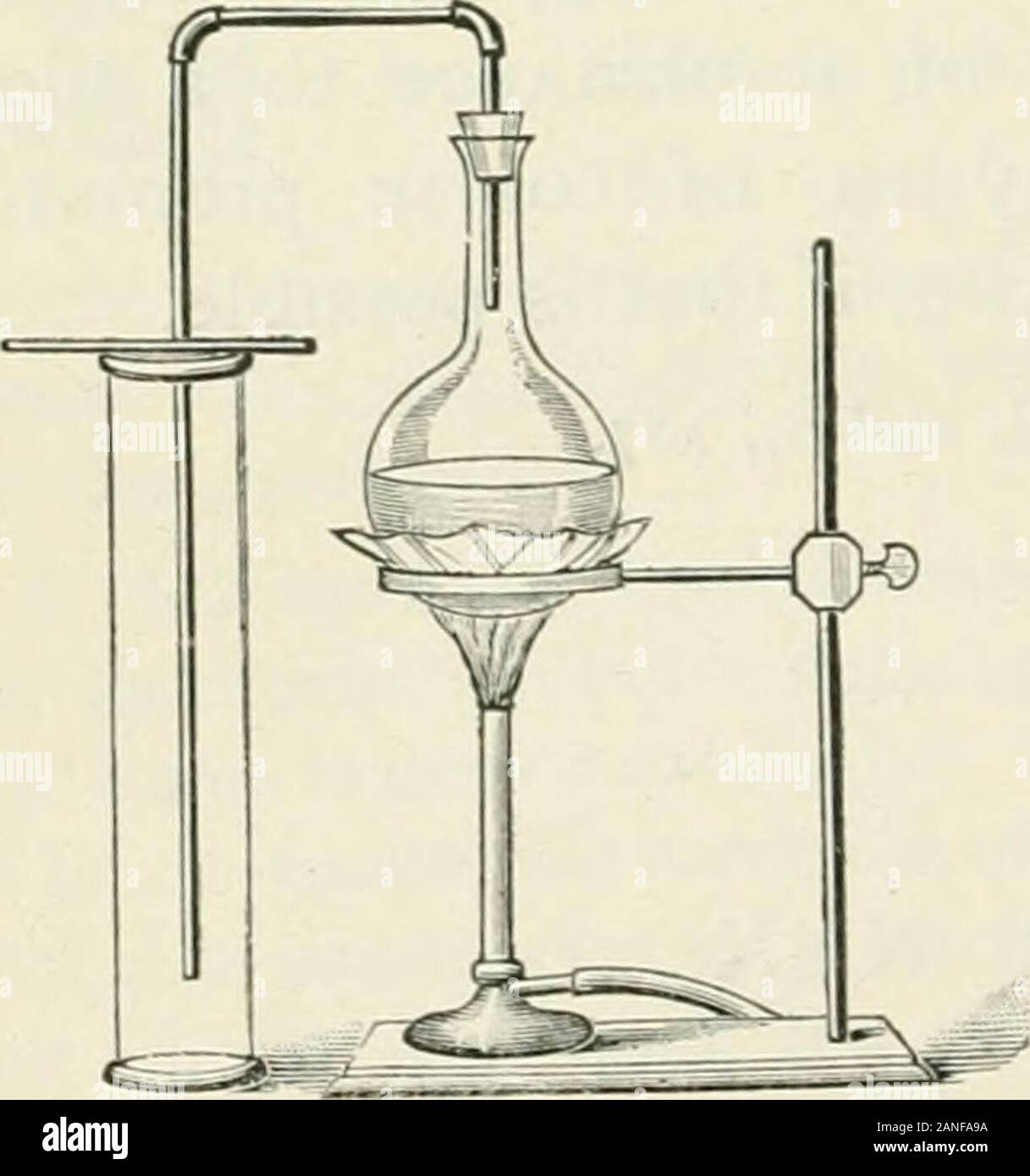 High School Chemistry . ese are sometimescalled haloid acids, and the salts which they form, haloidsalts; they are thus distinguished from salts and acidswhich contain oxygen. 2.—Experiments with Chlorine. 1. Into a test-tube put one part of manganese dioxide,two parts of salt, and three of sulphuric acid. Fit thetest-tube with a cork and delivery tube. Heat gently.Cautiously smell the gas that comes off. Note its colour. 2. To prepare the gas on a larger scale, take a 4 oz. Florence flask and place init about 20 grams of man-ganese dioxide and 100 c.c.of strong hydrochloric acid.Use fittings Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/high-school-chemistry-ese-are-sometimescalled-haloid-acids-and-the-salts-which-they-form-haloidsalts-they-are-thus-distinguished-from-salts-and-acidswhich-contain-oxygen-2experiments-with-chlorine-1-into-a-test-tube-put-one-part-of-manganese-dioxidetwo-parts-of-salt-and-three-of-sulphuric-acid-fit-thetest-tube-with-a-cork-and-delivery-tube-heat-gentlycautiously-smell-the-gas-that-comes-off-note-its-colour-2-to-prepare-the-gas-on-a-larger-scale-take-a-4-oz-florence-flask-and-place-init-about-20-grams-of-man-ganese-dioxide-and-100-ccof-strong-hydrochloric-aciduse-fittings-image340242150.html
High School Chemistry . ese are sometimescalled haloid acids, and the salts which they form, haloidsalts; they are thus distinguished from salts and acidswhich contain oxygen. 2.—Experiments with Chlorine. 1. Into a test-tube put one part of manganese dioxide,two parts of salt, and three of sulphuric acid. Fit thetest-tube with a cork and delivery tube. Heat gently.Cautiously smell the gas that comes off. Note its colour. 2. To prepare the gas on a larger scale, take a 4 oz. Florence flask and place init about 20 grams of man-ganese dioxide and 100 c.c.of strong hydrochloric acid.Use fittings Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/high-school-chemistry-ese-are-sometimescalled-haloid-acids-and-the-salts-which-they-form-haloidsalts-they-are-thus-distinguished-from-salts-and-acidswhich-contain-oxygen-2experiments-with-chlorine-1-into-a-test-tube-put-one-part-of-manganese-dioxidetwo-parts-of-salt-and-three-of-sulphuric-acid-fit-thetest-tube-with-a-cork-and-delivery-tube-heat-gentlycautiously-smell-the-gas-that-comes-off-note-its-colour-2-to-prepare-the-gas-on-a-larger-scale-take-a-4-oz-florence-flask-and-place-init-about-20-grams-of-man-ganese-dioxide-and-100-ccof-strong-hydrochloric-aciduse-fittings-image340242150.htmlRM2ANFA9A–High School Chemistry . ese are sometimescalled haloid acids, and the salts which they form, haloidsalts; they are thus distinguished from salts and acidswhich contain oxygen. 2.—Experiments with Chlorine. 1. Into a test-tube put one part of manganese dioxide,two parts of salt, and three of sulphuric acid. Fit thetest-tube with a cork and delivery tube. Heat gently.Cautiously smell the gas that comes off. Note its colour. 2. To prepare the gas on a larger scale, take a 4 oz. Florence flask and place init about 20 grams of man-ganese dioxide and 100 c.c.of strong hydrochloric acid.Use fittings
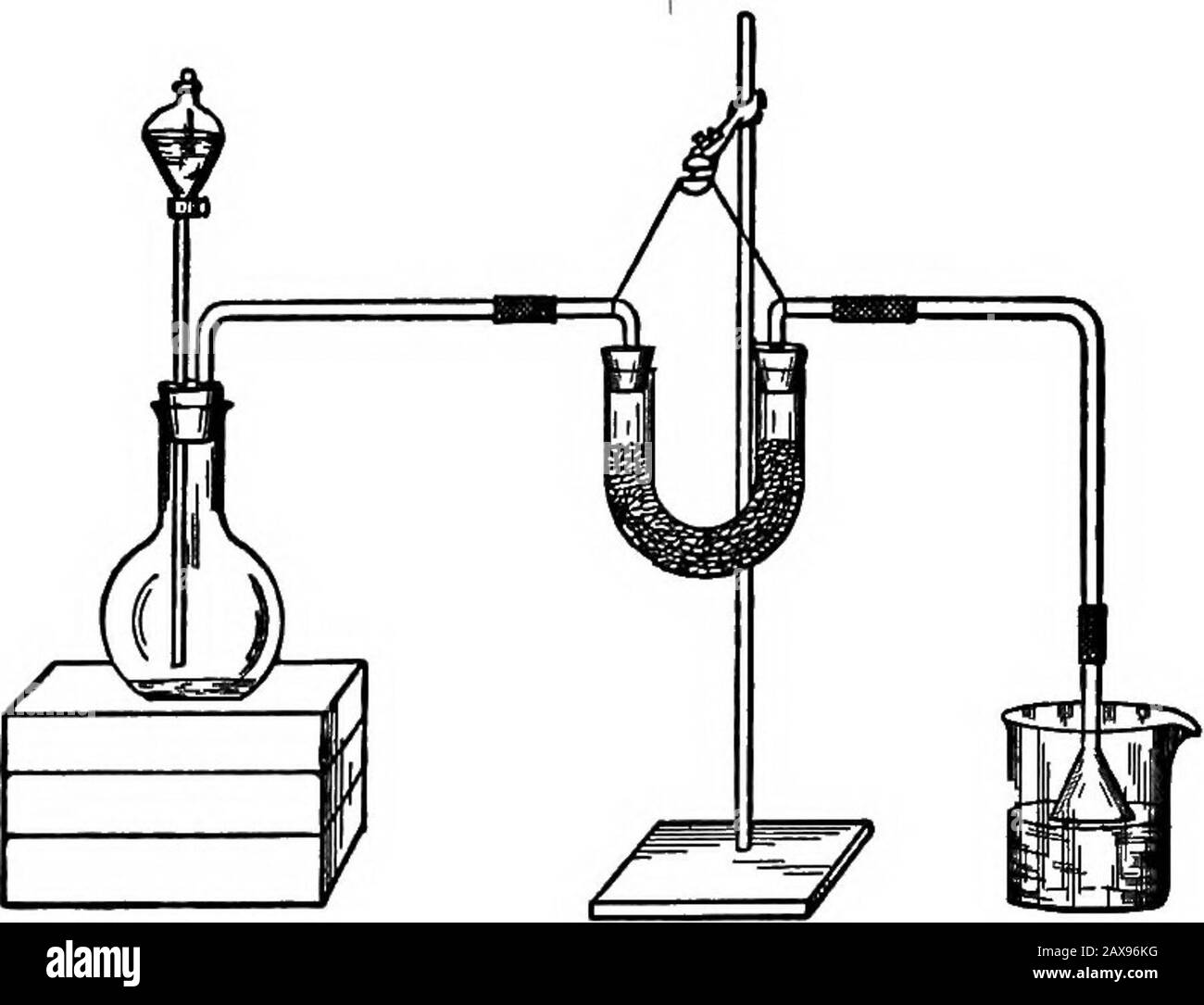 A college text-book of chemistry . ams manganese dioxide. Put the mixture into a 500 cc.. flask ; connect with a condenser (see Fig. 28). Mix 15 cc. con-centrated sulphuric acid and 90 cc. water. After cooling pourthe liquid on the mixture in the flask. Gently heat, when brominewill be given off in the form of vapor. A part of this will con-dense and collect in the receiver. Perform this experiment undera hood with a good draught. Htdrobromic Acid. ^ Experiment 82.—In a small porcelain evaporating-dish put afew crystals of potassium bromide. Pour on them a few drops ofconcentrated sulphuric ac Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/a-college-text-book-of-chemistry-ams-manganese-dioxide-put-the-mixture-into-a-500-cc-flask-connect-with-a-condenser-see-fig-28-mix-15-cc-con-centrated-sulphuric-acid-and-90-cc-water-after-cooling-pourthe-liquid-on-the-mixture-in-the-flask-gently-heat-when-brominewill-be-given-off-in-the-form-of-vapor-a-part-of-this-will-con-dense-and-collect-in-the-receiver-perform-this-experiment-undera-hood-with-a-good-draught-htdrobromic-acid-experiment-82in-a-small-porcelain-evaporating-dish-put-afew-crystals-of-potassium-bromide-pour-on-them-a-few-drops-ofconcentrated-sulphuric-ac-image343180868.html
A college text-book of chemistry . ams manganese dioxide. Put the mixture into a 500 cc.. flask ; connect with a condenser (see Fig. 28). Mix 15 cc. con-centrated sulphuric acid and 90 cc. water. After cooling pourthe liquid on the mixture in the flask. Gently heat, when brominewill be given off in the form of vapor. A part of this will con-dense and collect in the receiver. Perform this experiment undera hood with a good draught. Htdrobromic Acid. ^ Experiment 82.—In a small porcelain evaporating-dish put afew crystals of potassium bromide. Pour on them a few drops ofconcentrated sulphuric ac Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/a-college-text-book-of-chemistry-ams-manganese-dioxide-put-the-mixture-into-a-500-cc-flask-connect-with-a-condenser-see-fig-28-mix-15-cc-con-centrated-sulphuric-acid-and-90-cc-water-after-cooling-pourthe-liquid-on-the-mixture-in-the-flask-gently-heat-when-brominewill-be-given-off-in-the-form-of-vapor-a-part-of-this-will-con-dense-and-collect-in-the-receiver-perform-this-experiment-undera-hood-with-a-good-draught-htdrobromic-acid-experiment-82in-a-small-porcelain-evaporating-dish-put-afew-crystals-of-potassium-bromide-pour-on-them-a-few-drops-ofconcentrated-sulphuric-ac-image343180868.htmlRM2AX96KG–A college text-book of chemistry . ams manganese dioxide. Put the mixture into a 500 cc.. flask ; connect with a condenser (see Fig. 28). Mix 15 cc. con-centrated sulphuric acid and 90 cc. water. After cooling pourthe liquid on the mixture in the flask. Gently heat, when brominewill be given off in the form of vapor. A part of this will con-dense and collect in the receiver. Perform this experiment undera hood with a good draught. Htdrobromic Acid. ^ Experiment 82.—In a small porcelain evaporating-dish put afew crystals of potassium bromide. Pour on them a few drops ofconcentrated sulphuric ac
 Elements of inorganic chemistry : descriptive and qualitative . and oresof iron; chrome alum or other chromium salts; metallic aluminum andas many commonly occurring aluminum compounds as possible; metallicnickel and ores of nickel; cobalt ores; manganese ores; commercial formsof metallic zinc. 99. The Fourth Group Metals. Barium dioxide, hydroxide, and asmany barium-bearing minerals as possible; strontium nitrate ; many cal-cium bearing minerals; metallic magnesium ribbon, and many magnesium-bearing minerals. The Fifth Group 3Ietals are already provided for. 3G2 APPENDIX. GENERAL APPARATUS.Un Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/elements-of-inorganic-chemistry-descriptive-and-qualitative-and-oresof-iron-chrome-alum-or-other-chromium-salts-metallic-aluminum-andas-many-commonly-occurring-aluminum-compounds-as-possible-metallicnickel-and-ores-of-nickel-cobalt-ores-manganese-ores-commercial-formsof-metallic-zinc-99-the-fourth-group-metals-barium-dioxide-hydroxide-and-asmany-barium-bearing-minerals-as-possible-strontium-nitrate-many-cal-cium-bearing-minerals-metallic-magnesium-ribbon-and-many-magnesium-bearing-minerals-the-fifth-group-3ietals-are-already-provided-for-3g2-appendix-general-apparatusun-image339435694.html
Elements of inorganic chemistry : descriptive and qualitative . and oresof iron; chrome alum or other chromium salts; metallic aluminum andas many commonly occurring aluminum compounds as possible; metallicnickel and ores of nickel; cobalt ores; manganese ores; commercial formsof metallic zinc. 99. The Fourth Group Metals. Barium dioxide, hydroxide, and asmany barium-bearing minerals as possible; strontium nitrate ; many cal-cium bearing minerals; metallic magnesium ribbon, and many magnesium-bearing minerals. The Fifth Group 3Ietals are already provided for. 3G2 APPENDIX. GENERAL APPARATUS.Un Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/elements-of-inorganic-chemistry-descriptive-and-qualitative-and-oresof-iron-chrome-alum-or-other-chromium-salts-metallic-aluminum-andas-many-commonly-occurring-aluminum-compounds-as-possible-metallicnickel-and-ores-of-nickel-cobalt-ores-manganese-ores-commercial-formsof-metallic-zinc-99-the-fourth-group-metals-barium-dioxide-hydroxide-and-asmany-barium-bearing-minerals-as-possible-strontium-nitrate-many-cal-cium-bearing-minerals-metallic-magnesium-ribbon-and-many-magnesium-bearing-minerals-the-fifth-group-3ietals-are-already-provided-for-3g2-appendix-general-apparatusun-image339435694.htmlRM2AM6HKA–Elements of inorganic chemistry : descriptive and qualitative . and oresof iron; chrome alum or other chromium salts; metallic aluminum andas many commonly occurring aluminum compounds as possible; metallicnickel and ores of nickel; cobalt ores; manganese ores; commercial formsof metallic zinc. 99. The Fourth Group Metals. Barium dioxide, hydroxide, and asmany barium-bearing minerals as possible; strontium nitrate ; many cal-cium bearing minerals; metallic magnesium ribbon, and many magnesium-bearing minerals. The Fifth Group 3Ietals are already provided for. 3G2 APPENDIX. GENERAL APPARATUS.Un
Search Results for Manganese dioxide Stock Photos and Images (16)
Page 1 of 1