Quick filters:
Page 1 of 1
Alum crystals Stock Photos and Images

RMD96J3N–General view of an Alum works in the Whitby area. From George Walker 'The Costume of Yorkshire' Leeds 1814, Aquatint. Steeping calcined (burnt) shale in water in pits produce raw alum liquor which would be gravity fed to processing plant where it would be refined and boiled until alum crystals produced. These were important as a mordant (fixer) in textile dyeing industry.
RM2T8503E–Romanus Schmehlik Alum crystals, scale 25: 1. Silbergelatinepapier / Stereoformat 1912 , 1912

RF2JJTYFP–White transparent crystalline alum or potassium alum isolated on white background with hard shadows. Eco item for body care or water purification. Spa
![Alchemy Alphabet: ALUM (Alumen), also: Vitriol of Argile. Potash Alum, Potassium-Aluminium sulfate: Ch.formula=[KAl(SO₄)₂•12H₂O] or [Al₂(SO₄)₃]. Stock Vector Alchemy Alphabet: ALUM (Alumen), also: Vitriol of Argile. Potash Alum, Potassium-Aluminium sulfate: Ch.formula=[KAl(SO₄)₂•12H₂O] or [Al₂(SO₄)₃]. Stock Vector](https://c8.alamy.com/comp/2FKA8BX/alchemy-alphabet-alum-alumen-also-vitriol-of-argile-potash-alum-potassium-aluminium-sulfate-chformula=-kalso12ho-or-also-2FKA8BX.jpg)
RF2FKA8BX–Alchemy Alphabet: ALUM (Alumen), also: Vitriol of Argile. Potash Alum, Potassium-Aluminium sulfate: Ch.formula=[KAl(SO₄)₂•12H₂O] or [Al₂(SO₄)₃].
![Alchemy Alphabet: ALUM (Alumen). Potassium alum, Potassium-Aluminium sulfate, Aluminum salt: Chemical formula=[KAl(SO₄)₂•12H₂O] or [Al₂(SO₄)₃]. Stock Vector Alchemy Alphabet: ALUM (Alumen). Potassium alum, Potassium-Aluminium sulfate, Aluminum salt: Chemical formula=[KAl(SO₄)₂•12H₂O] or [Al₂(SO₄)₃]. Stock Vector](https://c8.alamy.com/comp/2F7G83T/alchemy-alphabet-alum-alumen-potassium-alum-potassium-aluminium-sulfate-aluminum-salt-chemical-formula=-kalso12ho-or-also-2F7G83T.jpg)
RF2F7G83T–Alchemy Alphabet: ALUM (Alumen). Potassium alum, Potassium-Aluminium sulfate, Aluminum salt: Chemical formula=[KAl(SO₄)₂•12H₂O] or [Al₂(SO₄)₃].

RM2WN9JY1–Harry Potter fan James Robbins, eleven, a pupil at Craigentinny Primary School, Edinburgh, gets hold of a giant blood red crystal, like the Philosophers Stone in the Harry Potter movie which is released today, at The University of Edinburgh's Chemistry Department today, Friday 16/11/01. As part of Chemistry Week which starts today James was competing with pupils from throughout Edinburgh and The Lothians in a competition to grow crystals and leapt at the chance to get hold of the giant chrome alum crystal which the University normally keeps locked up in it's museum.
![Alchemy Alphabet: FEATER ALUM, Halotrichite (Halotrichum), 'Salt hair'. Hydrated sulfate of Aluminium and Iron: Chemical formula=[FeAl₂(SO₄)₄•22H₂O]. Stock Vector Alchemy Alphabet: FEATER ALUM, Halotrichite (Halotrichum), 'Salt hair'. Hydrated sulfate of Aluminium and Iron: Chemical formula=[FeAl₂(SO₄)₄•22H₂O]. Stock Vector](https://c8.alamy.com/comp/2FKDC4R/alchemy-alphabet-feater-alum-halotrichite-halotrichum-salt-hair-hydrated-sulfate-of-aluminium-and-iron-chemical-formula=-fealso22ho-2FKDC4R.jpg)
RF2FKDC4R–Alchemy Alphabet: FEATER ALUM, Halotrichite (Halotrichum), 'Salt hair'. Hydrated sulfate of Aluminium and Iron: Chemical formula=[FeAl₂(SO₄)₄•22H₂O].

RF2HXE0D0–Potassium alum stones, or potash alum, called ame-stone, is the double sulfate of aluminum and potassium, widely used to reduce sweating

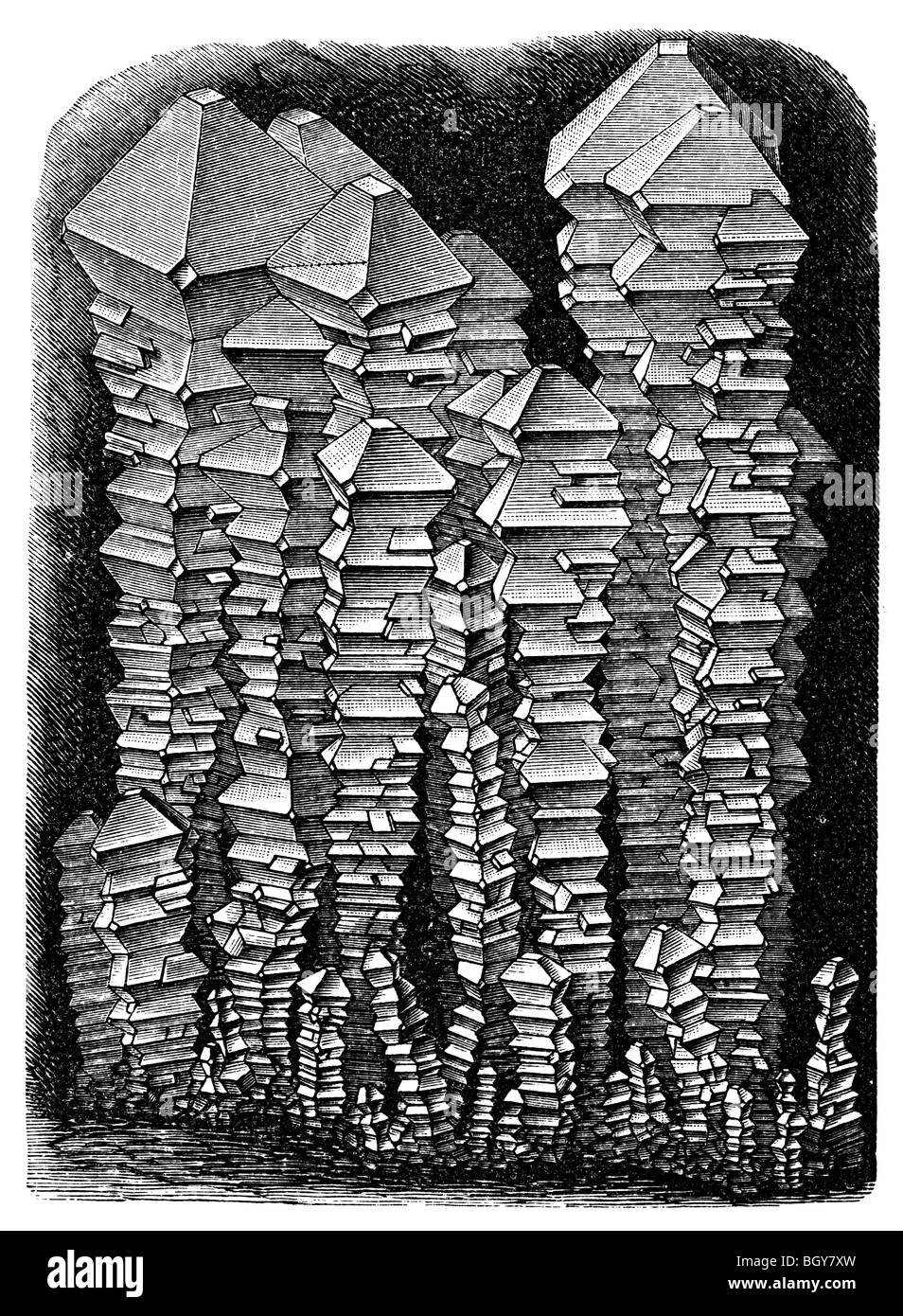
RM2AXFN9H–Scientific amusements . When clay is submitted to the influenceof sulphuric acid and chloride of pota-sium, we obtain alum, which is a sul-phate of alumina and potash. Alumis a colourless salt, which crystallizeson the surface of water in beautifuloctahedrons of striking regularity. TheCalcined alum. |^g ^^ page 21 represents a group ofalum crystals. This salt is much used in the colouring offabrics ; it is also used for the sizing of papers, and theclarification of tallow. Doctors also use it as an astrin-gent and caustic substance. When alum is submitted tothe action of heat in an earthenwa

RMPG4175–. Elements of applied microscopy. A text-book for beginners. Microscopy. FiG. 54.—Crystals of C-esidm Alum. (After Lehmann.) phate. From dilute acid solutions slender monoclinic prisms of the compound (CaS04+ 2H2O) crystallize out. Fig. 55.—Crystals of Calcium Sulphate. (After Lehmann.) (Fig. 55), showing numerous double twins, as at C, and in presence of strong acids, masses of minute needle- like crystals, as at .4.. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations may

RMG0KR9B–Brick wall & ceiling of 'Stufe' grotto entrance at Solfatara volcano. Pozzuoli near Naples Italy; Campi Flegrei volcanic area.

RMMA6YM2–. Elementary physics and chemistry: first stage . CRYSTALS AND CRYSTALLISATION—Continued. PRACTICAL WORK. Things required.—Crystals of washing-soda, sugar candy, borax, rock-crystal, blue vitriol, rock-salt, and alum. Flasks. Sand-bath. Laboratory burner. Tripod stand. Blotting paper. Magnify- ing glass. Evaporating basin. Sulphur. Iron spoon. Test-tubes. What to do. Evaporate a solu- tion of common salt by gently heating it, and, when the basin is dry, examine a little of the residue. Care- ful inspection will discover small cubes, the shape of some of which can be recognised by the unaided e

RF2JJK5KE–White transparent crystalline alum or potassium alum isolated on white background with tropical leaves and hard shadows. Eco item for body care or wat

RF2HXE0CN–Potassium alum stones, or potash alum, called ame-stone, is the double sulfate of aluminum and potassium, widely used to reduce sweating

RM2AWMEAJ–Portrait . when added inlarge quantities. One drop of a saturated solution to each ounce ofdeveloper is usually sufficient to prevent abrasionmarks, and one additional drop of bromide of potassiumto each ounce should also be added, as it tends to pre-serve the color and vigor of the print. When iodide of potassium is used in the hypo-alum toning bath it should never be added except insolution. A hypo-alum bath, to which crystals of iodide havebeen added, may cause yellow stains on the prints. 11 *P o r t r a it A Useful Electric Heater With the almost universal use of the hot hypo-alum method
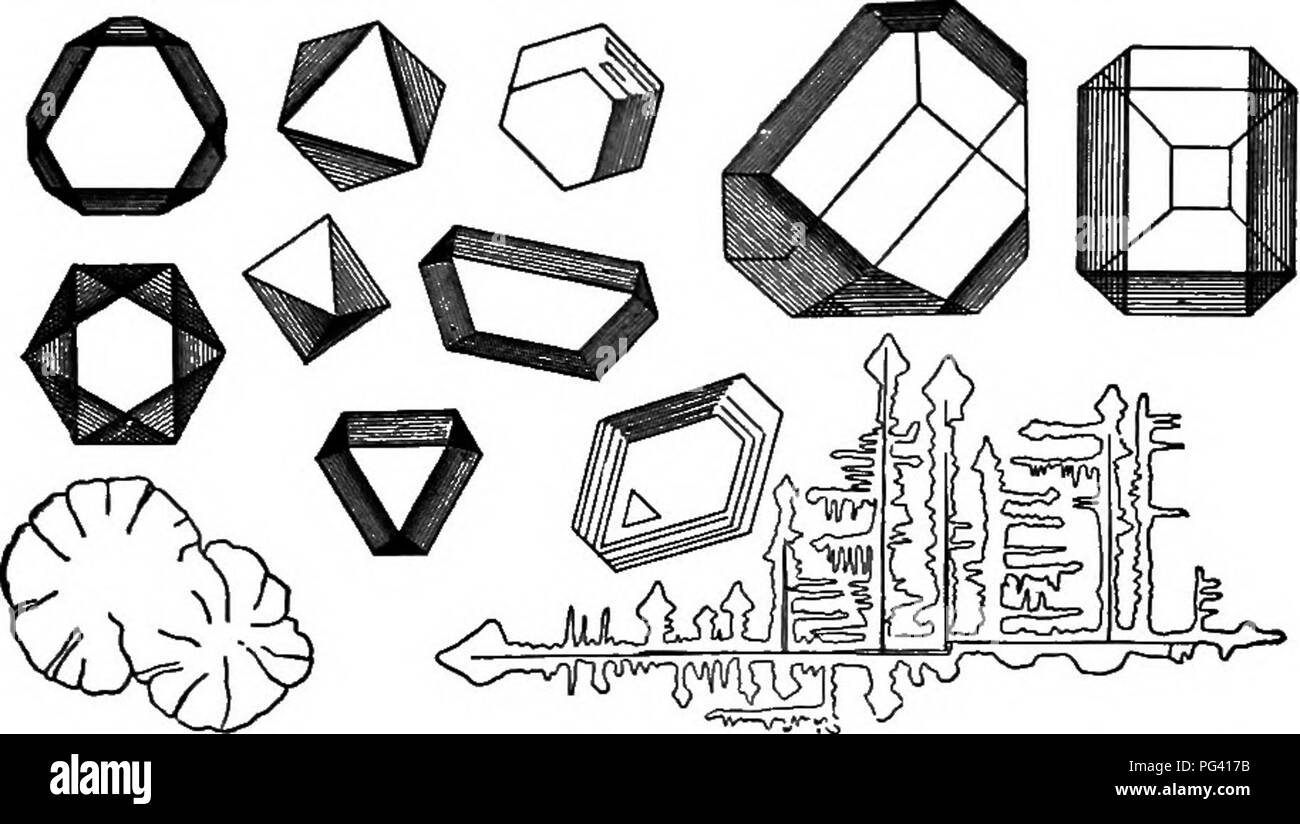
RMPG417B–. Elements of applied microscopy. A text-book for beginners. Microscopy. MICROCHEMISTR Y. 145 phuric acid, which in concentrated solution throws out short orthorhombic crystals of anhydrous calcium sul-. FiG. 54.—Crystals of C-esidm Alum. (After Lehmann.) phate. From dilute acid solutions slender monoclinic prisms of the compound (CaS04+ 2H2O) crystallize out. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations may not perfectly resemble the original work.. Winslow, C. -E.
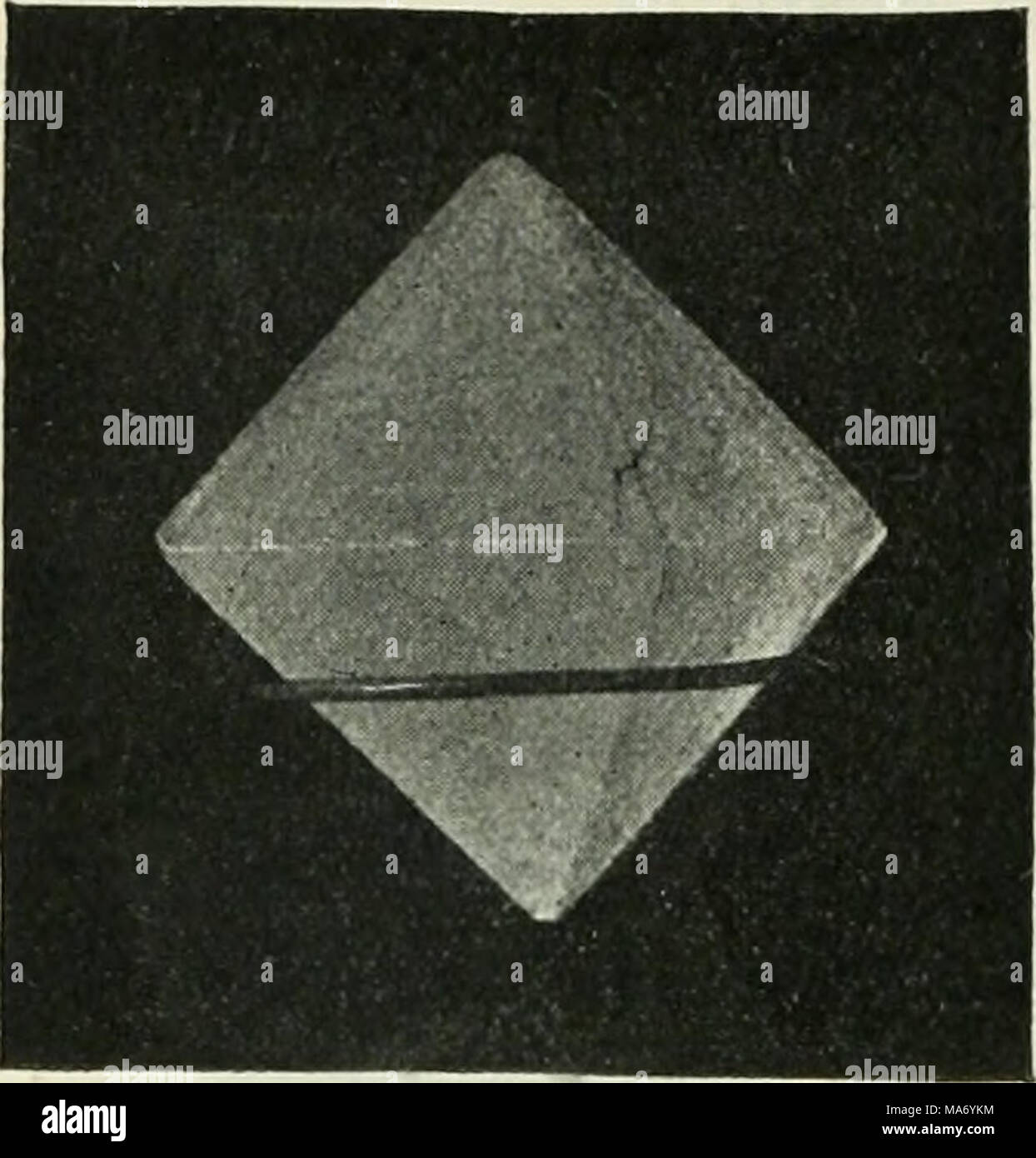
RMMA6YKM–. Elementary physics and chemistry: first stage . Fig. 85.âAn eight-sided crystal of alum. (From a photograph by Mr. H. E. Hadley.) How Crystals can be made.â Warm water when saturated with any soluble substance, as you learnt in a previous lesson, often contains more of the solid dissolved than an equal quantity of a cold saturated solution. The con- sequence of this is, that if you allow a warm saturated solution to get cold, the water can no longer keep all the substance in solution, and it separates out in the solid state, which, under these cir- cumstances, always takes a crystalline char

RF2HXE0D8–Potassium alum stones, or potash alum, called ame-stone, is the double sulfate of aluminum and potassium, widely used to reduce sweating

RM2AGC2PB–. Canadian grocer January-June 1910. CRYSTALS OR POWDER. The Old Refinery, H. W. CARTER & CO., LTD., 46 BRISTOL, EG. THE CANADIAN GROCER ?aMSMaMSMaMajsMajajajaEjaMSJSMMasMS sMEiMSMMaMaajsMaiaMMSiaraisjajQfMMMMSMarajarsMeMasiafajaMaMMa Jy^ WHITEST LiGHTEjI*! Economy Baking^owd€ SiAlNS NOAfci. Economy really means—supply to yourcustomers nothing but the best, because itpays in the long run. Baking Powder con-taining Alum may, and should, cost you afew cents less per dozen than Magic, becauseit costs less to manufacture. Alum is used in baking powder be-cause it is cheap. Dont handle cheap goods

RF2HXE0CP–Potassium alum stones, or potash alum, called ame-stone, is the double sulfate of aluminum and potassium, widely used to reduce sweating
RM2AG4HX9–. Journal. colouringmatters, >o that according to the degree to which the fibrehas been affected the stains appear after dyeing of a lightercolour than the rest of the cloth, or they may remainaltogether undyed. The behaviour of mildew stains towardsvarious dyes was found as follows :— Scarlet, dyed in one bath withcochineal, tin crystals andoxnlie acid. Billiard-cloth green, dyed withacid gn en, sulphuric acid andGlaubers salt. Moss-green, dyed with indigo-carmine, fustic and alum, inone bath. Vat-blue topped ith logwoo 1 .. Vat-blue, medium shade Dove-grey dyed with alizarin-blue sw. Dark

RF2HXE0CT–Potassium alum stones, or potash alum, called ame-stone, is the double sulfate of aluminum and potassium, widely used to reduce sweating

RM2AJEH9K–Field, laboratory, and library manual in physical geography . rble soluble in water? Put some ofthe powdered marble in a small beaker and fill the beaker withwater. Lead a tube from a carbon dioxide generator into thisbeaker of water and let the gas slowly bubble up through Fig. 26. A Beaker ar-ranged, for the Forma-tion of Crystals of Alum VEINS 137 the water. Continue this process for a day or two. Againpour off the water, filter, and evaporate to dryness. Doyou find any residuein the evaporatingdish? Is marble solu-ble in water which hascarbon dioxide in it?Water with carbondioxide is respo

RM2CE1YCB–. A practical handbook of dyeing and calico-printing. With eleven page-plates, forty-seven specimens of dyed and printed fabrics, and thirty-eight woodcuts . iquor) Z in the after-mentioned compounds :— Z.—Alum 10 kilos. Acetate of lead 10 ,, Water 20 litres. We designate as red mordant for brown the following mixture :— Alum 144 kilos. Pyrolignite of lead 144 „ Water . 660 ,, Crystals of soda (carbonate of) .... 4 ,,With these the following brown mordants may be prepared :— A.—Mordant Z 8 litres Pyrolignite of iron at io° Baume (sp. gr. 1*075) .. 4 ,,Extract of quercitron at 200 Baume (sp. gr

RM2CE204H–. A practical handbook of dyeing and calico-printing. With eleven page-plates, forty-seven specimens of dyed and printed fabrics, and thirty-eight woodcuts . lb. oxalic acid ; 2 gallons prussiate tin pulp.. Light Blue. CYANOGEN COLOURS. 165 The tin pulp is prepared from a mixture of the two following solutions, wellstirred, and filtered to a pulp :— a. 1 gallon of hot water ; 1 lb. of yellow prussiate of potash. b. 1 gallon of water; 1 lb. muriate of tin crystals.The cloth when printed is steamed for 40 minutes, aged during one night, andrinsed in a weak solution of bichrome and alum at 120° F

RM2CE1WK7–. A practical handbook of dyeing and calico-printing. With eleven page-plates, forty-seven specimens of dyed and printed fabrics, and thirty-eight woodcuts . Pale Yellow from Persian Berries. No. 49. Dark Yellotv.— Berry liquor, at 4° Tw 1 gal. Flour ii lbs. Boil, and add— Alum g ozs. When cool add— Tin crystals .. ; 3 ozs. When dissolved, strain. After printing, steam for thirty minutes, as in No. 48.. Dark Yellow from Persian Berries. For other patterns, and particulars of various coal-tar colours fixed by thesteam process, the reader is referred to pp. 206, 207, 217, 218, 225, and 226. HOFM
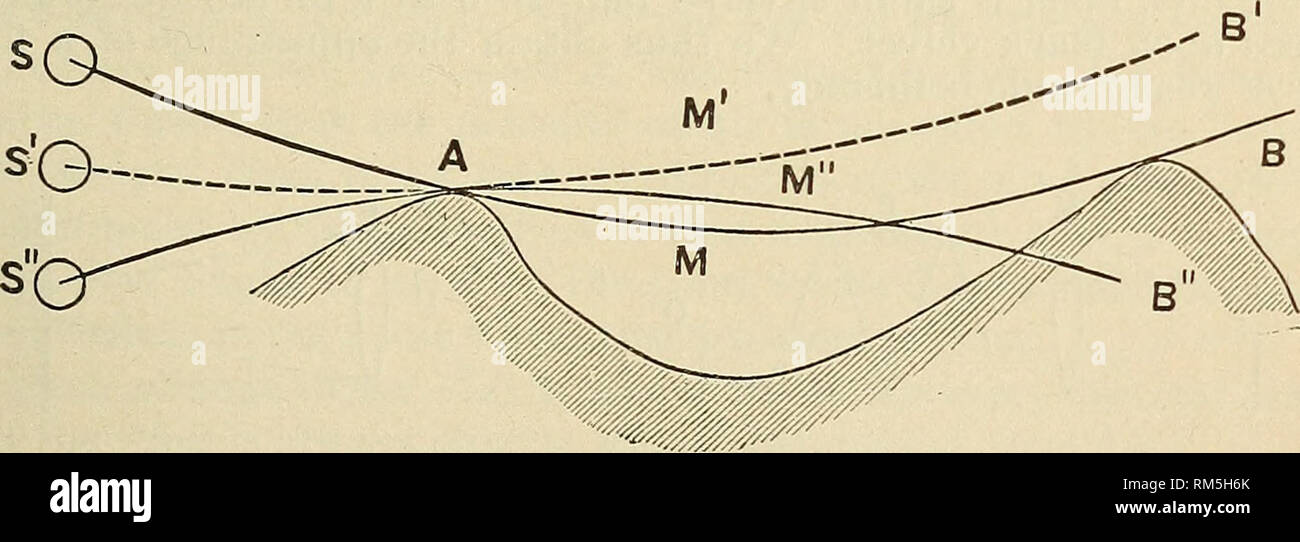
RMRM5H6K–. Annual report of the Board of Regents of the Smithsonian Institution. Smithsonian Institution; Smithsonian Institution. Archives; Discoveries in science. PHYSICAL PHENOMENA OF UPPER REGIONS OF ATMOSPHERE. 127 add to the contents of the jar a quarter of its total volume of rectified spirits; the alum, insoluble in the alcoholic mixture, precipitates in very minute crystals that float within the liquid. The image of the sun first becomes dim as in a fog, but soon a brilliant and slightly iridescent circle is seen, simulating very closely the appearance of a halo. The experiment is brilliant an
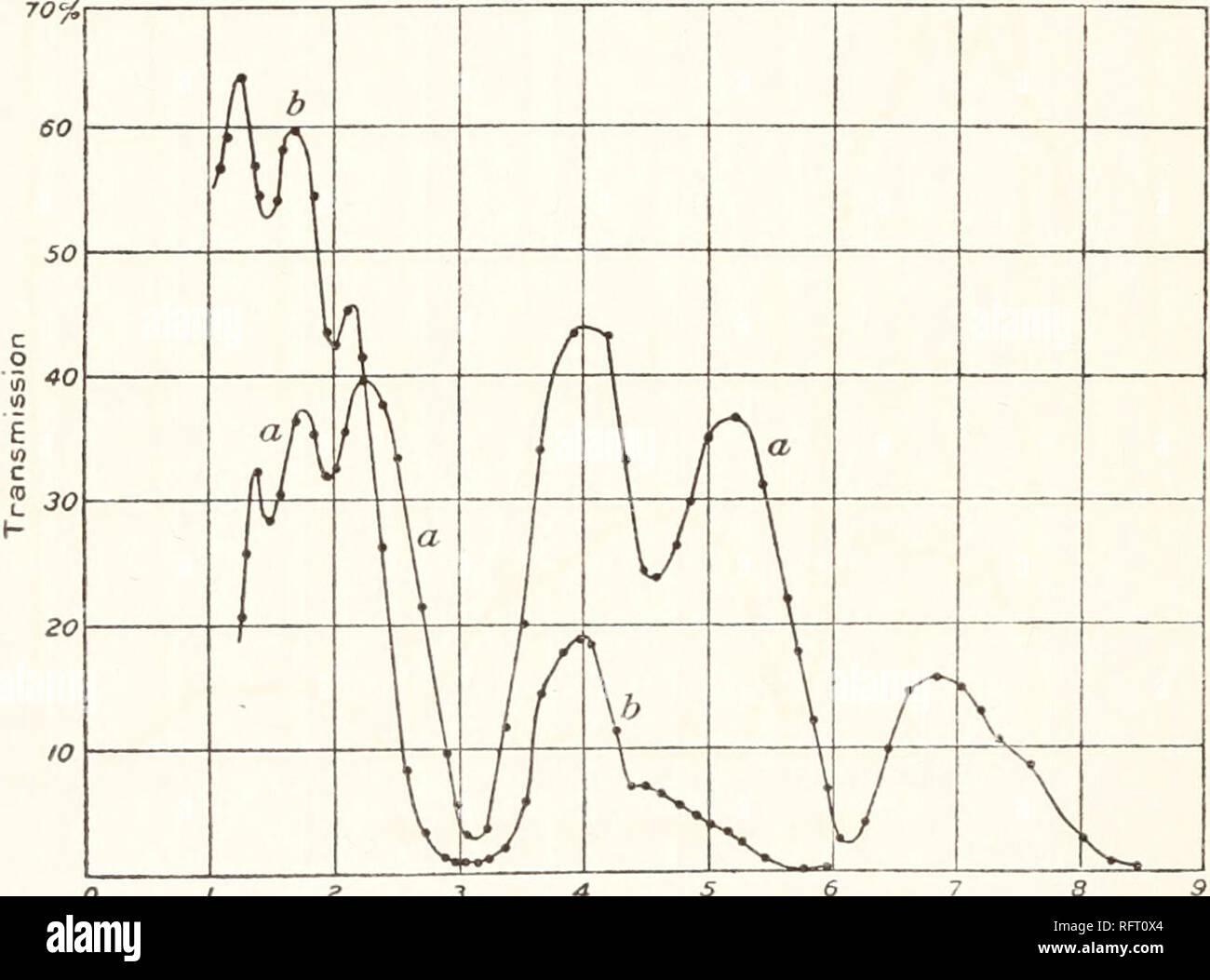
RMRFT0X4–. Carnegie Institution of Washington publication. 24 INFRA-RED TRANSMISSION SPECTRA. 3, and 4.75 /x, beyond which point the opacity becomes too great for further exploration. The crystals obtainable were small, so that the section was not quite long enough to cover the whole slit. STILBITE (CaAl2Si6Ow+6H2O). (Transparent section cut perpendicular to optic axis. f = o.ii. Curve a, fig. 7.) The stilbite curve is to be noticed for its great transparency with all the water bands superposed. There do not appear to be any important bands belonging to the mineral itself. POTASSIUM ALUM (K2SO4Al2(SO4)

RMRM8X7H–. Annual report of the Cornell University Agricultural Experiment Station, Ithaca, N.Y. Cornell University. Agricultural Experiment Station; Agriculture -- New York (State). 7IO Reading-Course for Farmers' Wives. For each gallon of water, use four tablespoonfuls of alum. Pour a half pound of ammonium phosphate into a quart jar and fill with water. Shake and let stand. Keep this saturated solution for use as needed and add more water to any crystals that may be left in the jar. Use a little of this solution in the bluing water or as a part of the li'quid used in making starch. EQUIPMENT A great
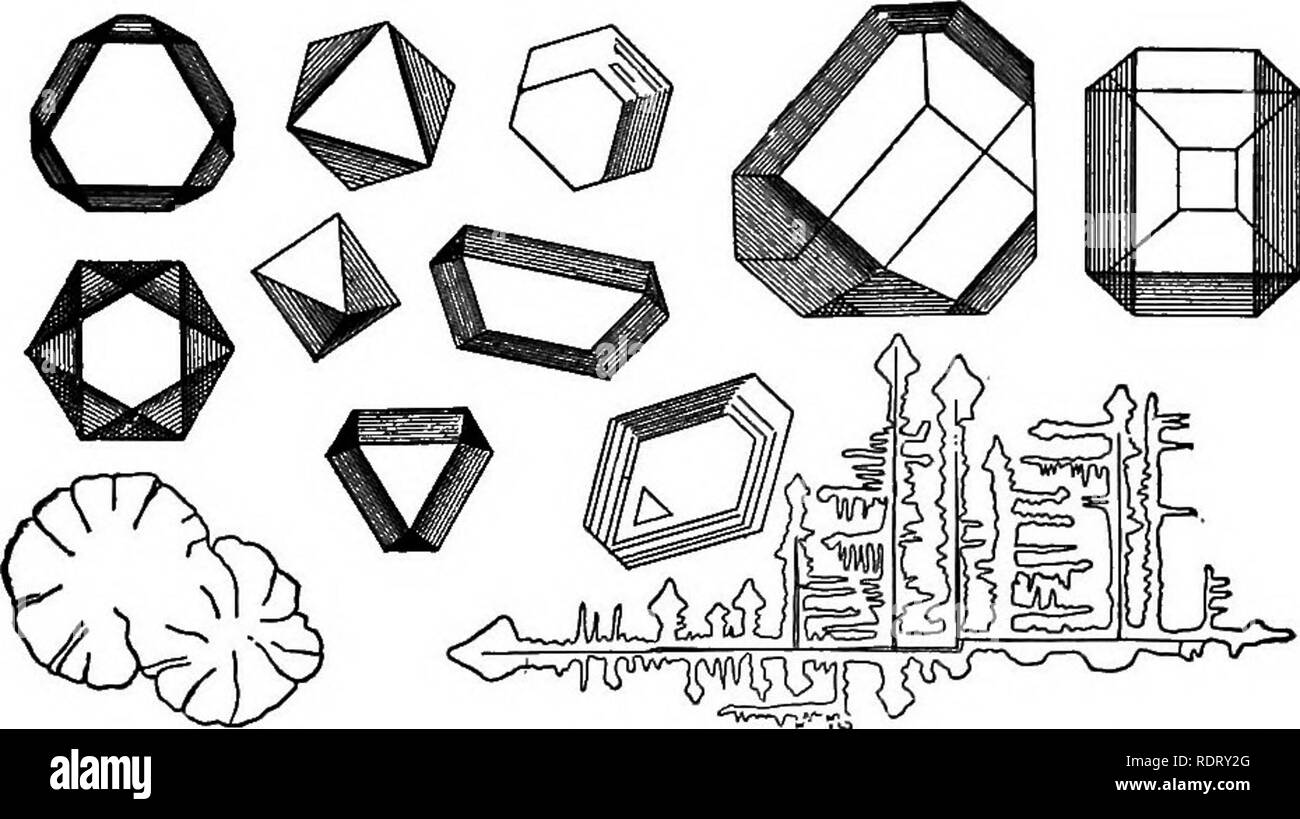
RMRDRY2G–. Elements of applied microscopy. A text-book for beginners. Microscopy. MICROCHEMISTRY. 145 phuric acid, which in concentrated solution throws out short orthorhombic crystals of anhydrous calcium sul-. FiG. 54.—Crystals of Cesium Alum. (After Lehmann.) phate. From dilute acid solutions slender monoclinic prisms of the compound (CaS04+ 2H2O) crystallize out. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations may not perfectly resemble the original work.. Winslow, C. -E. A.

RMRDRY2A–. Elements of applied microscopy. A text-book for beginners. Microscopy. FiG. 54.—Crystals of Cesium Alum. (After Lehmann.) phate. From dilute acid solutions slender monoclinic prisms of the compound (CaS04+ 2H2O) crystallize out. Fig. e,e,.—Crystals of Calcium Sulphate. (After Lehmann.) (Fig. 55), showing numerous double twins, as at C, and in presence of strong acids, masses of minute needle- like crystals, as at .4.. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations ma

RMRD27XA–. Elementary physics and chemistry: first stage. Science. CRYSTALS AND CRYSTALLISATION—Continued. PRACTICAL WORK. Things required.—Crystals of washing-soda, sugar candy, borax, rock-crystal, blue vitriol, rock-salt, and alum. Flasks. Sand-bath. Laboratory burner. Tripod stand. Blotting paper. Magnify- ing glass. Evaporating basin. Sulphur. Iron spoon. Test-tubes. What to do. Evaporate a solu- tion of common salt by gently heating it, and, when the basin is dry, examine a little of the residue. Care- ful inspection will discover small cubes, the shape of some of which can be recognised by the u

RMRDJGXW–. Elements of applied microscopy. A text-book for beginners. Microscopy. FiG. 54.—Crystals of C-esidm Alum. (After Lehmann.) phate. From dilute acid solutions slender monoclinic prisms of the compound (CaS04+ 2H2O) crystallize out. Fig. 55.—Crystals of Calcium Sulphate. (After Lehmann.) (Fig. 55), showing numerous double twins, as at C, and in presence of strong acids, masses of minute needle- like crystals, as at .4.. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations may

RMRD27XC–. Elementary physics and chemistry: first stage. Science. ELEMENTARY PHYSICS AND CHEMISTRY. LESSON XXXIII.. CRYSTALS AND CRYSTALLISATION—Continued. PRACTICAL WORK. Things required.—Crystals of washing-soda, sugar candy, borax, rock-crystal, blue vitriol, rock-salt, and alum. Flasks. Sand-bath. Laboratory burner. Tripod stand. Blotting paper. Magnify- ing glass. Evaporating basin. Sulphur. Iron spoon. Test-tubes. What to do. Evaporate a solu- tion of common salt by gently heating it, and, when the basin is dry, examine a little of the residue. Care- ful inspection will discover small cubes, the
RMRDJGY3–. Elements of applied microscopy. A text-book for beginners. Microscopy. MICROCHEMISTR Y. 145 phuric acid, which in concentrated solution throws out short orthorhombic crystals of anhydrous calcium sul-. FiG. 54.—Crystals of C-esidm Alum. (After Lehmann.) phate. From dilute acid solutions slender monoclinic prisms of the compound (CaS04+ 2H2O) crystallize out. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations may not perfectly resemble the original work.. Winslow, C. -E.
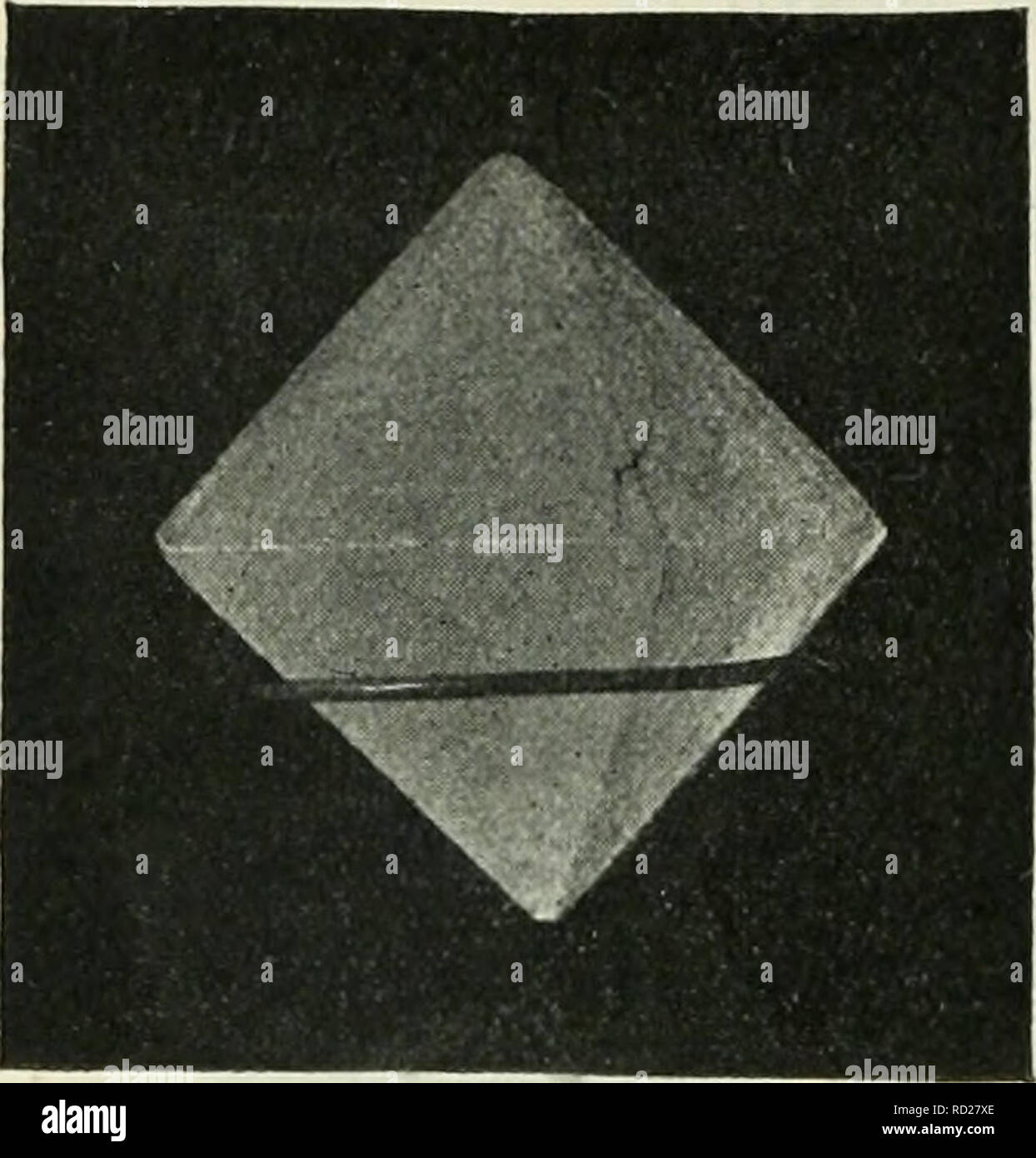
RMRD27XE–. Elementary physics and chemistry: first stage. Science. 126 ELEMENTARY PHYSICS AND CHEMISTRY,. Fig. 85.âAn eight-sided crystal of alum. (From a photograph by Mr. H. E. Hadley.) How Crystals can be made.â Warm water when saturated with any soluble substance, as you learnt in a previous lesson, often contains more of the solid dissolved than an equal quantity of a cold saturated solution. The con- sequence of this is, that if you allow a warm saturated solution to get cold, the water can no longer keep all the substance in solution, and it separates out in the solid state, which, under these c

RMRDXTE0–. Plant physiology. Plant physiology. Fig. S4-—Crystals of ammonium magnesium phosphate. yellow crystals of ammonium phospho-molybdate being formed and gradually becoming bright green (Fig. ss). Upon addition of strontium nitrate sulphur separates out as small rounded crystals of strontium sulphate (Fig. 56). An- i6%tt4''^ h O o^^y^^^q^^^^o. Fig. 56-—Crystals of strontium sulphate. Fig. 57.—Crystals of thallium chloride. Other test for sulphuric acid is the addition of cesium chloride and aluminium chloride, which leads to the formation of large crystals of caesium-alum. Chlor- ides may be ide
Search Results for Alum crystals Stock Photos and Images (77)
Page 1 of 1