On poisons in relation to medical jurisprudence and medicine . Mercury sublimedin globules, in a re-duction-tube. The same, magnified. A small sublimate of mercury,.magnified 124 diameters. In Organic Liquids.—Corrosive sublimate may be sometimes ob-tained by decantation as a heavy sediment from the mucous and bloodycontents of the stomach or the matters vomited. These should be sep-arated, dried, and weighed. Unlike arsenic, corrosive sublimate insolution is precipitated as an insoluble compound by many organicprinciples, such as albumen, fibrin, casein, mucous membrane, also bygluten, tannic
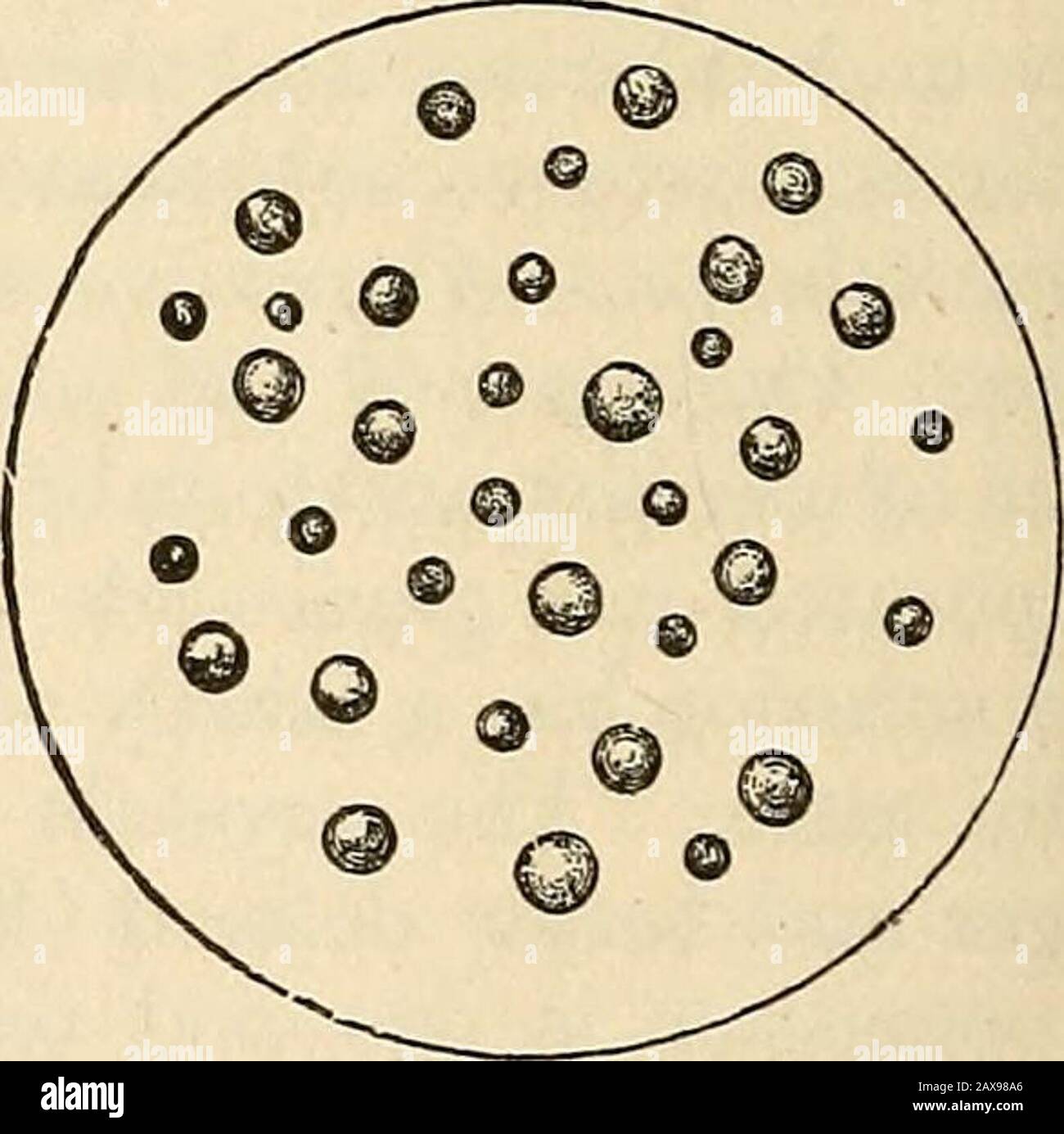
Image details
Contributor:
The Reading Room / Alamy Stock PhotoImage ID:
2AX98A6File size:
7.1 MB (195 KB Compressed download)Releases:
Model - no | Property - noDo I need a release?Dimensions:
1583 x 1578 px | 26.8 x 26.7 cm | 10.6 x 10.5 inches | 150dpiMore information:
This image is a public domain image, which means either that copyright has expired in the image or the copyright holder has waived their copyright. Alamy charges you a fee for access to the high resolution copy of the image.
This image could have imperfections as it’s either historical or reportage.
On poisons in relation to medical jurisprudence and medicine . Mercury sublimedin globules, in a re-duction-tube. The same, magnified. A small sublimate of mercury, .magnified 124 diameters. In Organic Liquids.—Corrosive sublimate may be sometimes ob-tained by decantation as a heavy sediment from the mucous and bloodycontents of the stomach or the matters vomited. These should be sep-arated, dried, and weighed. Unlike arsenic, corrosive sublimate insolution is precipitated as an insoluble compound by many organicprinciples, such as albumen, fibrin, casein, mucous membrane, also bygluten, tannic acid, and other vegetable substances. Thus, then, wemust not expect to find it in all cases in a state of solution in the PROCESS FOR SEPARATING THE POISON. 373 stomach. After removing the mineral sediment, we may separate anyof these insoluble compounds, reserving them for further analysis. As a trial-test for the presence of mercury in solution in the organicliquid, we may employ copper gauze or foil. A small portion of liquidacidulated with one-fourth