Quick filters:
Page 1 of 1
Strontium 85 Stock Photos and Images
 SrCl2 - Strontium Chloride. Chemical compound. CAS number 10476-85-4 Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/srcl2-strontium-chloride-chemical-compound-cas-number-10476-85-4-image595351003.html
SrCl2 - Strontium Chloride. Chemical compound. CAS number 10476-85-4 Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/srcl2-strontium-chloride-chemical-compound-cas-number-10476-85-4-image595351003.htmlRF2WGGG7R–SrCl2 - Strontium Chloride. Chemical compound. CAS number 10476-85-4
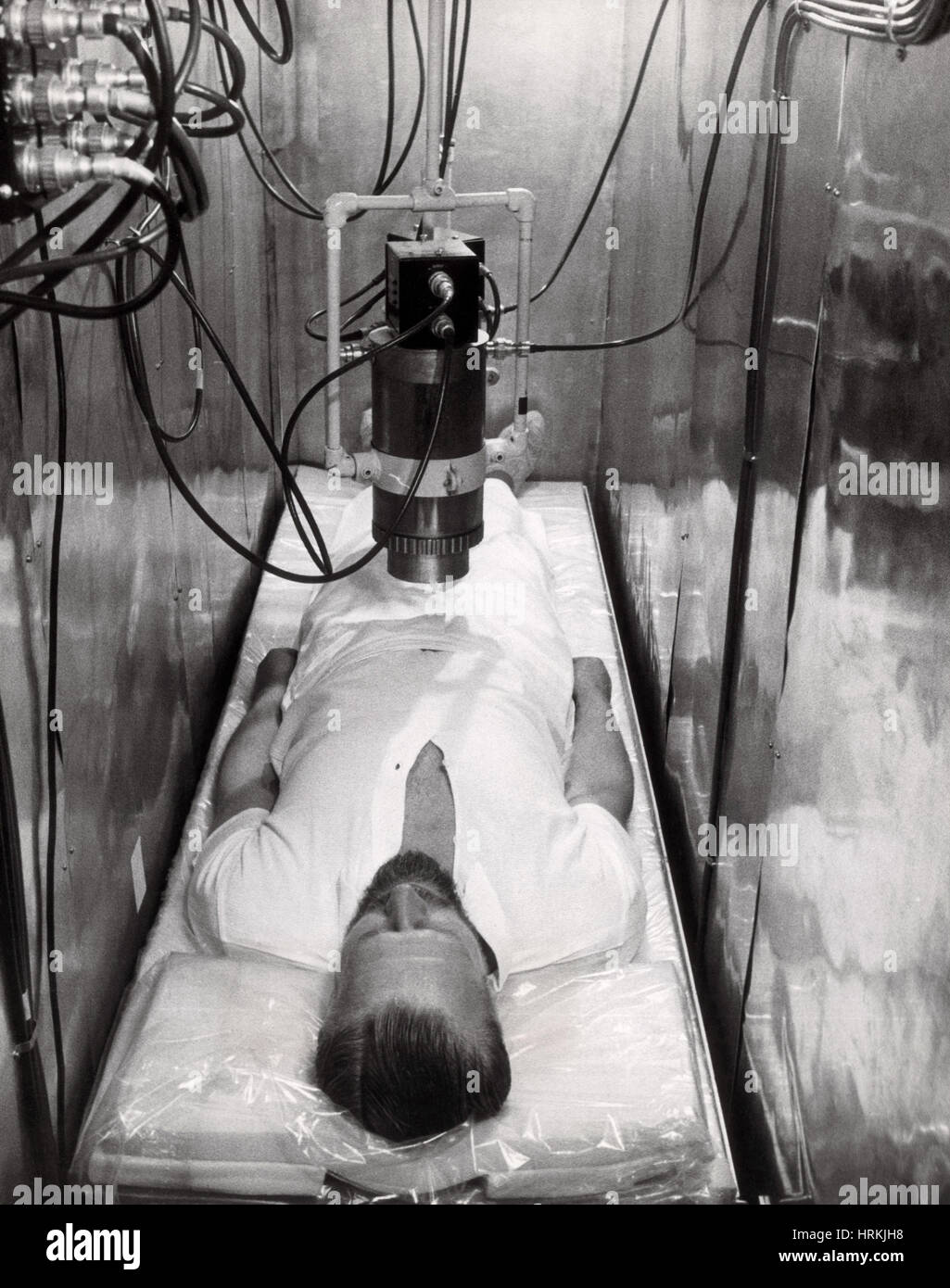 Radiation Experiment Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stock-photo-radiation-experiment-135041348.html
Radiation Experiment Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stock-photo-radiation-experiment-135041348.htmlRMHRKJH8–Radiation Experiment
 . Bulletin - New York State Museum. Science. GUIE>E TO THE MIXEUALOGIC COLLECTIONS 85 Witherite is mined at Fallowfield Eng. Small deposits of the mineral occur near Lexington Ivy. and at Thunder bay, Lake Superior. It is used as an adulterant of white lead and in the refining of beet sugar molasses. Strontianite SrC03 Strontianite, the carbonate of strontium, contains 29.9$ carbon dioxid and 70.1$ strontia. Distinct orthorhombic crystals are quite rare. Eadiated, spear-shaped or acicular crystalline aggregates are common; also columnar, fibrous and granular masses. The luster of strontiani Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/bulletin-new-york-state-museum-science-guiegte-to-the-mixeualogic-collections-85-witherite-is-mined-at-fallowfield-eng-small-deposits-of-the-mineral-occur-near-lexington-ivy-and-at-thunder-bay-lake-superior-it-is-used-as-an-adulterant-of-white-lead-and-in-the-refining-of-beet-sugar-molasses-strontianite-src03-strontianite-the-carbonate-of-strontium-contains-299-carbon-dioxid-and-701-strontia-distinct-orthorhombic-crystals-are-quite-rare-eadiated-spear-shaped-or-acicular-crystalline-aggregates-are-common-also-columnar-fibrous-and-granular-masses-the-luster-of-strontiani-image233989959.html
. Bulletin - New York State Museum. Science. GUIE>E TO THE MIXEUALOGIC COLLECTIONS 85 Witherite is mined at Fallowfield Eng. Small deposits of the mineral occur near Lexington Ivy. and at Thunder bay, Lake Superior. It is used as an adulterant of white lead and in the refining of beet sugar molasses. Strontianite SrC03 Strontianite, the carbonate of strontium, contains 29.9$ carbon dioxid and 70.1$ strontia. Distinct orthorhombic crystals are quite rare. Eadiated, spear-shaped or acicular crystalline aggregates are common; also columnar, fibrous and granular masses. The luster of strontiani Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/bulletin-new-york-state-museum-science-guiegte-to-the-mixeualogic-collections-85-witherite-is-mined-at-fallowfield-eng-small-deposits-of-the-mineral-occur-near-lexington-ivy-and-at-thunder-bay-lake-superior-it-is-used-as-an-adulterant-of-white-lead-and-in-the-refining-of-beet-sugar-molasses-strontianite-src03-strontianite-the-carbonate-of-strontium-contains-299-carbon-dioxid-and-701-strontia-distinct-orthorhombic-crystals-are-quite-rare-eadiated-spear-shaped-or-acicular-crystalline-aggregates-are-common-also-columnar-fibrous-and-granular-masses-the-luster-of-strontiani-image233989959.htmlRMRGK4G7–. Bulletin - New York State Museum. Science. GUIE>E TO THE MIXEUALOGIC COLLECTIONS 85 Witherite is mined at Fallowfield Eng. Small deposits of the mineral occur near Lexington Ivy. and at Thunder bay, Lake Superior. It is used as an adulterant of white lead and in the refining of beet sugar molasses. Strontianite SrC03 Strontianite, the carbonate of strontium, contains 29.9$ carbon dioxid and 70.1$ strontia. Distinct orthorhombic crystals are quite rare. Eadiated, spear-shaped or acicular crystalline aggregates are common; also columnar, fibrous and granular masses. The luster of strontiani
 SrCl2 strontium chloride CAS 10476-85-4 chemical substance in white plastic laboratory packaging Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/srcl2-strontium-chloride-cas-10476-85-4-chemical-substance-in-white-plastic-laboratory-packaging-image545279578.html
SrCl2 strontium chloride CAS 10476-85-4 chemical substance in white plastic laboratory packaging Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/srcl2-strontium-chloride-cas-10476-85-4-chemical-substance-in-white-plastic-laboratory-packaging-image545279578.htmlRF2PK3HJJ–SrCl2 strontium chloride CAS 10476-85-4 chemical substance in white plastic laboratory packaging
 . Carnegie Institution of Washington publication. STRONTIUM IODIDE. 33 Curve I , v = 33 II, v = 64 III, v = 128 IV, V = 256 V , v = 512 VI, v =. 25 $ 50 # 75/o Concentration of Methyl Alcohol FIG. 4. — STRONTIUM IODIDE AT 0°. TABLE 15. — Comparison of the molecular conductivity of strontium iodide. In water, methyl alcohol, and mixtures of these solvents. V AtO°. At 25°. 0 p. ct. 25 p.ct. 50 p. ct. 75 p. ct. 100 p. ct. 0 p. ct. 25 p.ct. 50 p.ct. 75 p. ct. 100 p. ct. 32 113.1 63.06 50.19 55.53 75.82 205.3 131.3 103.8 98.09 101.4 64 117.7 66.05 52.61 59.24 85.01 214.5 138.5 109.9 104.8 115.3 128 Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/carnegie-institution-of-washington-publication-strontium-iodide-33-curve-i-v-=-33-ii-v-=-64-iii-v-=-128-iv-v-=-256-v-v-=-512-vi-v-=-25-50-75o-concentration-of-methyl-alcohol-fig-4-strontium-iodide-at-0-table-15-comparison-of-the-molecular-conductivity-of-strontium-iodide-in-water-methyl-alcohol-and-mixtures-of-these-solvents-v-ato-at-25-0-p-ct-25-pct-50-p-ct-75-p-ct-100-p-ct-0-p-ct-25-pct-50-pct-75-p-ct-100-p-ct-32-1131-6306-5019-5553-7582-2053-1313-1038-9809-1014-64-1177-6605-5261-5924-8501-2145-1385-1099-1048-1153-128-image233478900.html
. Carnegie Institution of Washington publication. STRONTIUM IODIDE. 33 Curve I , v = 33 II, v = 64 III, v = 128 IV, V = 256 V , v = 512 VI, v =. 25 $ 50 # 75/o Concentration of Methyl Alcohol FIG. 4. — STRONTIUM IODIDE AT 0°. TABLE 15. — Comparison of the molecular conductivity of strontium iodide. In water, methyl alcohol, and mixtures of these solvents. V AtO°. At 25°. 0 p. ct. 25 p.ct. 50 p. ct. 75 p. ct. 100 p. ct. 0 p. ct. 25 p.ct. 50 p.ct. 75 p. ct. 100 p. ct. 32 113.1 63.06 50.19 55.53 75.82 205.3 131.3 103.8 98.09 101.4 64 117.7 66.05 52.61 59.24 85.01 214.5 138.5 109.9 104.8 115.3 128 Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/carnegie-institution-of-washington-publication-strontium-iodide-33-curve-i-v-=-33-ii-v-=-64-iii-v-=-128-iv-v-=-256-v-v-=-512-vi-v-=-25-50-75o-concentration-of-methyl-alcohol-fig-4-strontium-iodide-at-0-table-15-comparison-of-the-molecular-conductivity-of-strontium-iodide-in-water-methyl-alcohol-and-mixtures-of-these-solvents-v-ato-at-25-0-p-ct-25-pct-50-p-ct-75-p-ct-100-p-ct-0-p-ct-25-pct-50-pct-75-p-ct-100-p-ct-32-1131-6306-5019-5553-7582-2053-1313-1038-9809-1014-64-1177-6605-5261-5924-8501-2145-1385-1099-1048-1153-128-image233478900.htmlRMRFRTM4–. Carnegie Institution of Washington publication. STRONTIUM IODIDE. 33 Curve I , v = 33 II, v = 64 III, v = 128 IV, V = 256 V , v = 512 VI, v =. 25 $ 50 # 75/o Concentration of Methyl Alcohol FIG. 4. — STRONTIUM IODIDE AT 0°. TABLE 15. — Comparison of the molecular conductivity of strontium iodide. In water, methyl alcohol, and mixtures of these solvents. V AtO°. At 25°. 0 p. ct. 25 p.ct. 50 p. ct. 75 p. ct. 100 p. ct. 0 p. ct. 25 p.ct. 50 p.ct. 75 p. ct. 100 p. ct. 32 113.1 63.06 50.19 55.53 75.82 205.3 131.3 103.8 98.09 101.4 64 117.7 66.05 52.61 59.24 85.01 214.5 138.5 109.9 104.8 115.3 128
Search Results for Strontium 85 Stock Photos and Images (5)
Page 1 of 1