Quick filters:
Page 1 of 1
Solid carbonic acid Stock Photos and Images

RM2AG58D9–. Dental materia medica and therapeutics; with special reference to the rational application of remedial measures to dental diseases ... uce a short superficial anesthesia in afew minutes. Too rapid cooling or prolonged freezing by methylchlorid (boiling point —12° F., —24.5° C), or the various mix-tures thereof, produce deeper anesthesia, but such procedures aredangerous. They frequently cut off circulation in the affected partso completely as to produce sloughing (gangrene). Liquid nitrous PHYSIOLOGIC ACTION OF ANESTHETICS. 453 oxid gas, liquid or solid carbonic acid (recently known as carbo

RM2ABXD3K–AN IMPROVED SAW. Mining Near the Equator. A HUB ATTACHING DEVICE. LEWIS' HUB ATTACHING DEVICE. The Frozen Milk industry. Solid Carbonic Acid. OB. 4 Fast Locomotive. GUSTAVESON'S PNEUMATIC TIRE VALVE. The Siberian Railway. AN IMPROVED TUBE CLEANER. EDLUND'S HAND SAW. THE FARIES B OILER TUBE CLEANER., scientific american, 1895-08-24
RMPG0E5Y–. A manual of veterinary hygiene. Veterinary hygiene. VENTILATION 63 the most favourable circumstances, viz., where the air is obtained through cracks and crannies in doors and windows, diffusion is a slow process, and by itself can never be trusted for the purpose of renovating vitiated air. Diffusion merely causes the various gases present in a building to mix. Eoscoe showed that carbonic acid experi- mentally evolved in a room was reduced to one-half in ninety minutes by this process. This is not sufficiently rapid for the purpose of ventilation, and further, it leaves the solid particles o
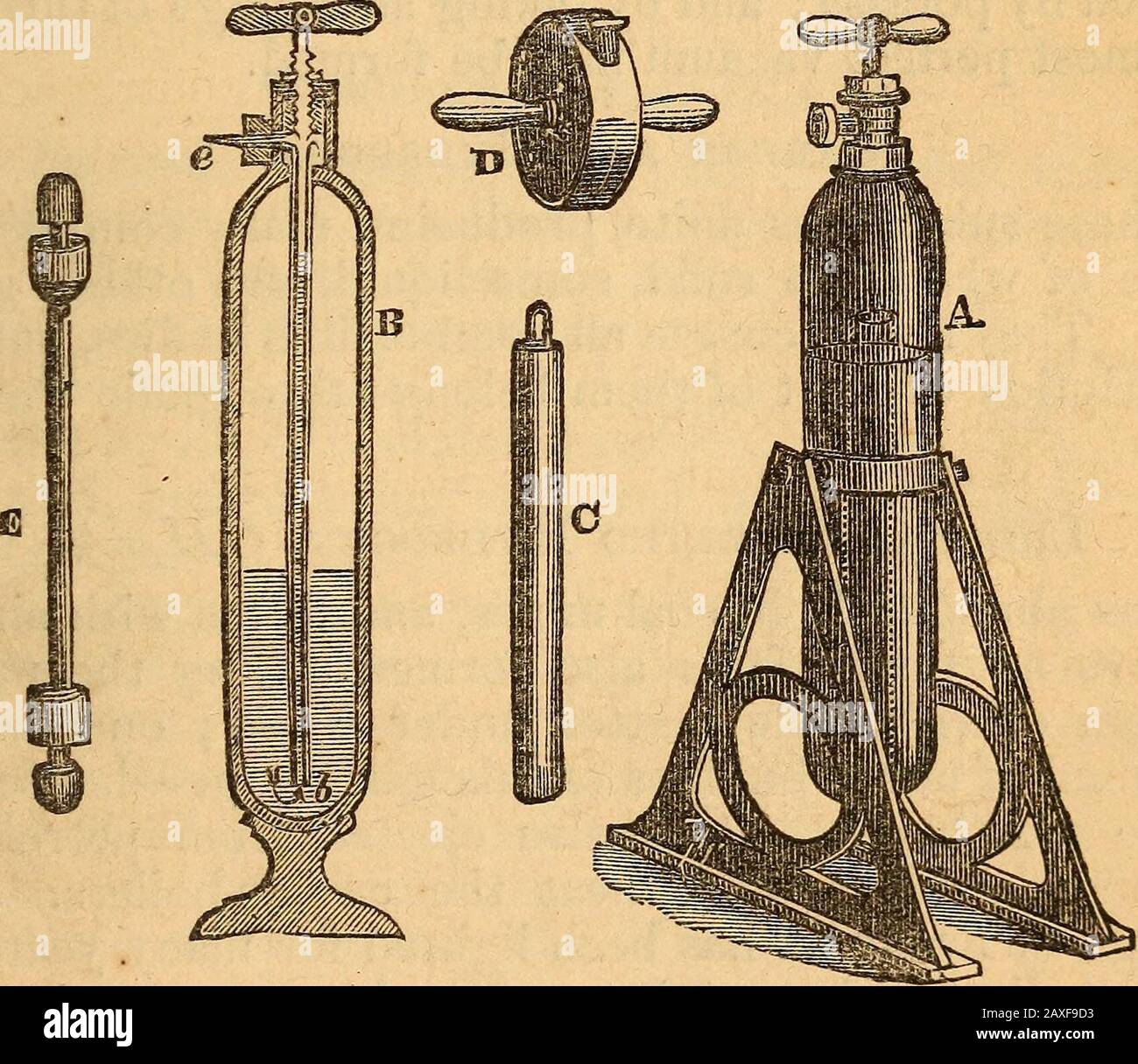
RM2AXF9D3–A text-book on chemistryFor the use of schools and colleges . SOLID CARBONIC ACID. 297 If through a tube the breath be made to pass intolime-water, a deposit of carbonate of lime renders thewater milky; or if the breath be conducted through lit-mus water, the color changes to red; the air thus ex-pired from the lungs contains three or four per cent, ofcarbonic acid. A man throws out about eight ouncesof carbon as carbonic acid every day. Under a pressure of thirty-six atmospheres, or by be-ing cooled to -—106°, carbonic acid condenses into a liq-uid four times more expansible by heat than atmo

RM2ABXP6M–SOLID CARBONIC ACID FROM 'SPARKLETS.' drop of mercury be placed on it it can be frozen in a few seconds. The experiment is SOME DISASTROUS ACETYLENE GAS EXPLOSIONS 25 1899 occurred February 6 at Fort Wayne Ind. in a two-story house occupied by four persons. On the morning of the occurrence. the water-pipes. He light ed a candle and crawled through a small opening which connected the cellar plosion tearing away the section holding the weather boarding apart from the studding and the plaster ing. The inside of the house was a total wreck. The floors of several of the rooms were pushed upward and
RM2CHF36W–. The London, Edinburgh and Dublin philosophical magazine and journal of science . en and W. G. Robson on The former result was therefore due to the proportion ofalcohol being too large. In order to ascertain whether alower critical temperature for the two liquids existed wecooled some methyl alcohol to —78° 0. in solid carbonic acid,and distilled some ethane into it. The two layers appearedagain quite distinctly, without any indication of a criticalpoint. The pressures of the three phases (Table IV.) will be seen tobe smaller than those for pure ethane. The pressures indi-cated by pb are thos

RFK2XHY8–White Mammoth landscape with dead trees in Angel Terrace, Mamoth Hot Springs, Yellowstone National Park, WY
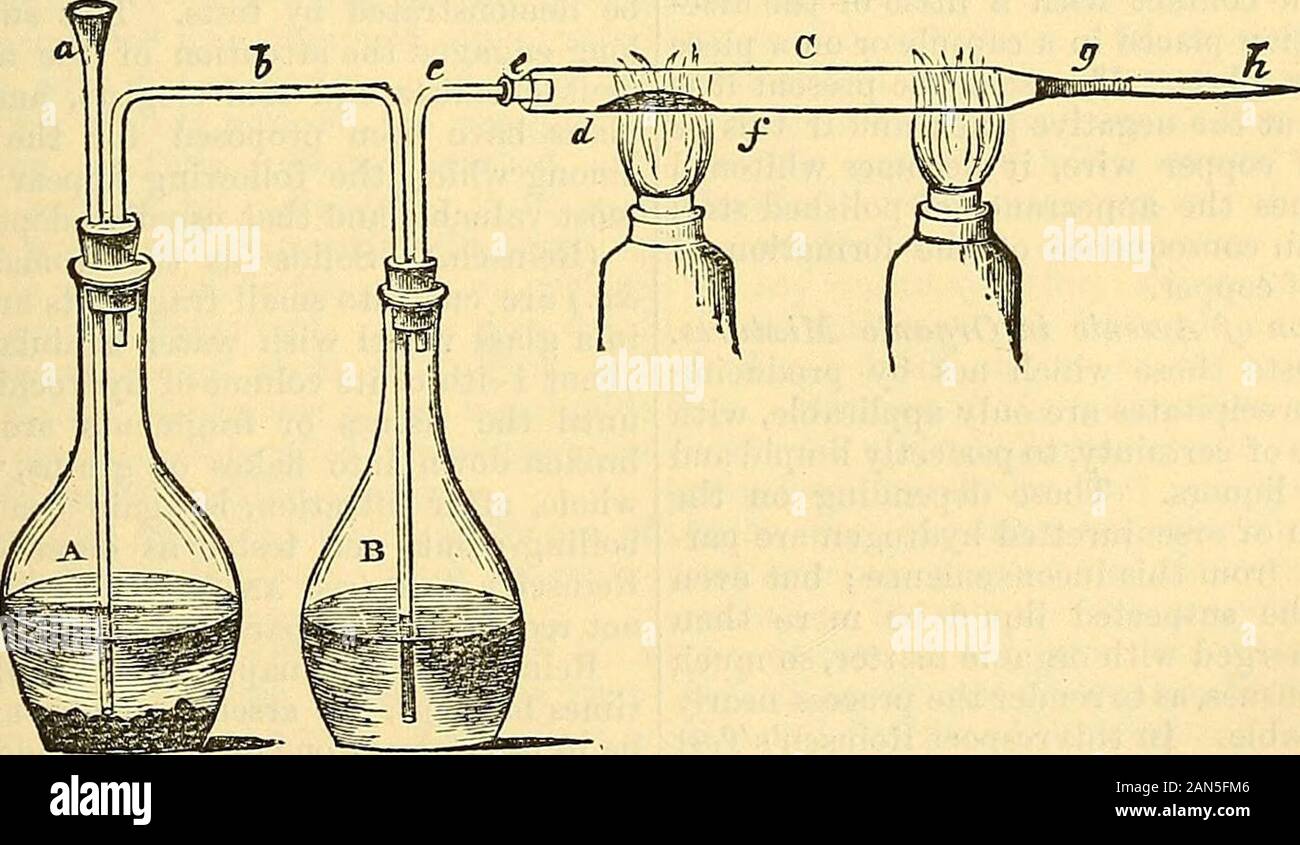
RM2AN5FM6–Cooley's cyclopaedia of practical receipts and collateral information in the arts, manufactures, professions, and trades including medicine, pharmacy, hygiene, and domestic economy : designed as a comprehensive supplement to the Pharmacopoeia and general book of reference for the manufacturer, tradesman, amateur, and heads of families . carbonic acid gas. This methodhas been followed by Drs Babo and Freseniuswith the most satisfactory results, and is thusperformed:—{A) is a capacious flask for theevolution of carbonic acid, half filled withrather large pieces of solid limestone or mar-ble (not
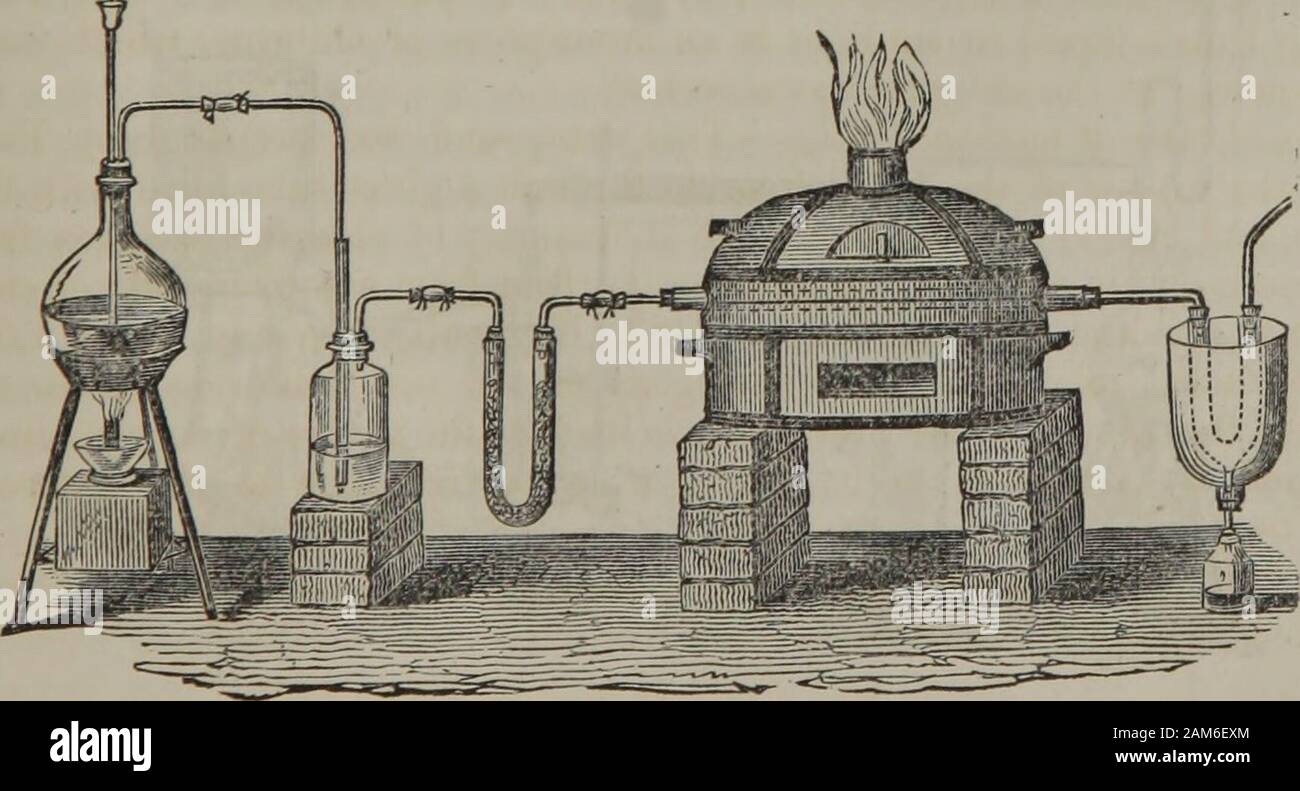
RM2AM6EXM–Elements of chemistry : including the applications of the science in the arts . isomeric withFaradays sesquichloride, but of which the vapour density was 4.082. Kolbeformed a crystallizable compound of perchloride of carbon and sulphurous acid,which has the formula 2(S02) + C2C14. Another chloride of carbon, of the formula C20C18, was obtained by M. Laurent,by the action of chlorine upon naphthaline, C20H8, in the form of a crystalline solid,soluble in boiling petroleum. Chloroxicarbonic gas, CO.C1. — This gas is formed by exposing equal measuresof chlorine and carbonic oxide to sunshine, when

RFK2XJ1P–Bacteria and Algea color the Minerva Terrace in Mammoth Hot Springs, Yellowstone National Park, WY
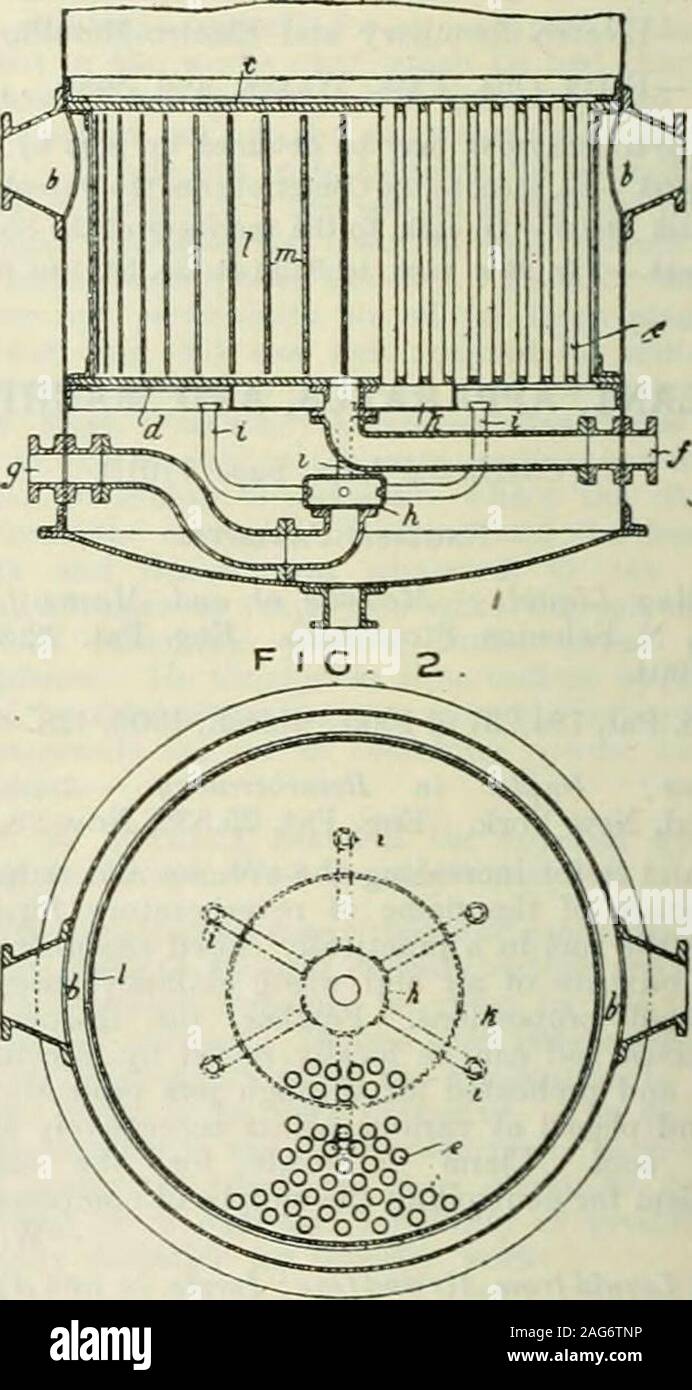
RM2AG6TNP–. Journal. , the latter being either ready madeor produced during the operation of mixing.—C. S. German Patents. Liquids, especially those containing Carbonic Acid; Process and Apparatus for the Purification of . Act.-Ges. Fabr. f. Brauerei-Eiorichtungen vorm. H.Gehrke and Co. Ger. Pat. 161,025, March 17, 1903. The process, which is intended specially for the clari-fication of liquids containing dissolved carbonic acid,consists in passing the liquid under pressure through acentrifugal separator in which filter-elements are disposedparallel to the cover of the apparatus. The solid impuritiesare

RFK2XHWJ–Bacteria and Algea color the Minerva Terrace in Mammoth Hot Springs, Yellowstone National Park, WY
RM2AXBY8R–..Report on the building and ornamental stones of Canada, volI-V . feet—Solid bed. 6 feet—Solid bed. The jointing is not developed sufficiently to interfere with the obtainingof stone of the largest dimensions required. The good stone (88) is muchalike in all the beds. The stone: Xo. 88.—This example is lighter than the Queenston stoneand is shown in Plate LXXVII, Xo. 2. The colour is permanent, as scarcelyany difference is seen after treatment with carbonic acid. The structuie isvery similar to that of the l^lue Queenston stone. The physical propertiesare listed below:— Specific gravity Weigh

RFK2XJ0B–White Mammoth landscape with dead trees in Angel Terrace, Mamoth Hot Springs, Yellowstone National Park, WY
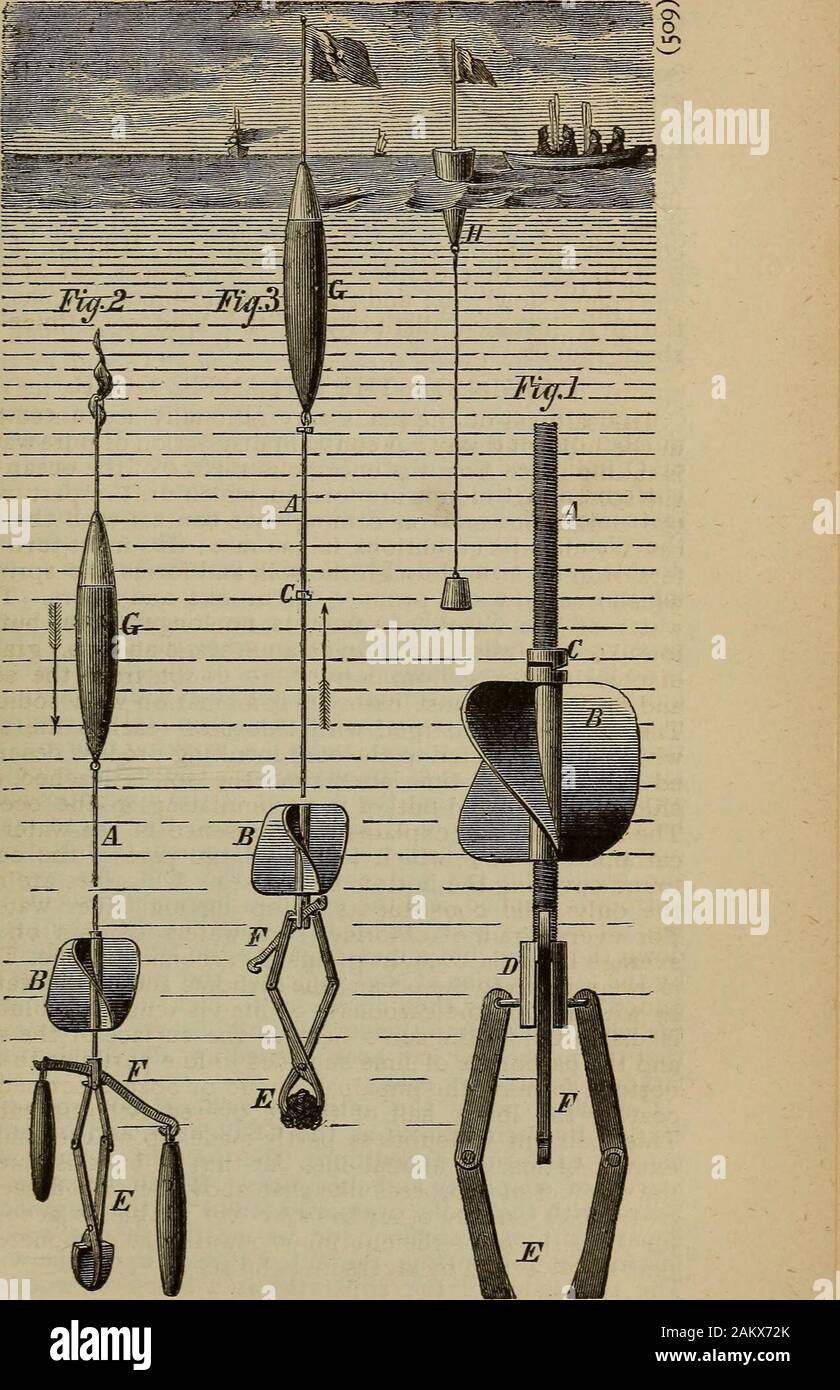
RM2AKX72K–The Science record; a compendium of scientific progress and discovery . he same process explains the presence in sea-water ofcarbonate of lime, which forms a large part of the oozefound covering the bottom of the sea. Chlorides are notthe only solid constituents of spring and river waters.For every grain of chloride, such waters contain on anaverage from six to eight grains of carbonate of lime. Thisis swept down to the ocean ; the carbonic acid in the water,by which alone the carbonate of lime is rendered soluble,is dissipated by the suns heat at the surface of the sea,and the carbonate of li

RFK2XHX3–Bacteria and Algea color the Minerva Terrace in Mammoth Hot Springs, Yellowstone National Park, WY

RM2AXF56W–Scientific amusements . d. The forms assumed bycarbon are very variable, and pervade nature in all itsphases. We have carbon in crystals, in the animal andvegetable kingdoms, and amongst the chief minerals a solid,odourless, tasteless, infusible, and almost insoluble body.In various combinations carbon meets us at every turn ;united with oxygen it forms carbonic acid, which weexhale for the plants to imbibe. We have it in coal, CARBON. 97 with hydrogen and oxygen. We have it building upanimal tissues, and it is never absent in two out of thethree great divisions of nature — the plants and the

RFK2XJ15–White Mammoth landscape with dead trees in Angel Terrace, Mamoth Hot Springs, Yellowstone National Park, WY
RM2AX9735–On poisons in relation to medical jurisprudence and medicine . e, it melts, then becomes solid; again melts,acquiring a dark color, and gives off vapors of acetic acid; a blackmass is left in the tube, consisting of carbon and reduced metallic lead.There is no metallic sublimate formed. 2. It is very soluble in water,even when cold; river or spring water is turned milky by it, chieflyfrom the presence of carbonic acid and sulphates. Goulard water isgenerally opaque for this reason. 3. A small portion of the powderplaced in a saucer, containing a solution of iodide of potassium, acquiresa fine

RM2AND0EP–The history of Banbury : including copious historical and antiquarian notices of the neighborhood . deposit, and a scum of thesame colour appears on the water in parts where stagnant. The spring discharges fromhalf a gallon to one gallon in a minute. In 32 oz. of the water at 60° are Carbonic Acid gas 5 cubic inches Hydrochlorate Magnesia 0.21 grains. Chloride Sodium or common Salt 0.54 Sulphate Lime 1.5 Carbonate Lime 3.8 Protoxide Iron 0.024 Silica a trace Total weigh! of solid contents 6.074 The water of St. Rumbalds Well at Astrop is also slightly chalybeate. (44) Bakers Northamp., p. 720.
RM2CJ9PWX–. Scientific news for general readers; a popular illustrated weekly journal of science . dation the muscularenergy of the body. The solid h3-drocarbons, starch andoil, after digestion and incorporation with the tissues, onbeing converted into carbonic acid and water, supply theheat energy of the body, just as the carbon and hydro-carbon in coal supplies heat energy to a steam engine. Thenitrogenous constituents, however, are used, as we observedabove, to make good the wear and tear of the nitrogenoustissues ; and it must be remembered that the muscles wearout faster when doing work, and that t

RM2CGH5N7–. Book of summer resorts, explaining where to find them, how to find them, and their especial advantages, with details of time tables and prices .. . lid contents 479.191 Carbonic Acid. 320.777 Atmospheric Air 1.461 Gaseous contents 322.233 Temperature 48® The mineral waters, as will be seen, contam large por-tions of chloride of sodium, carbonate of soda, carbonate oflime, carbonate of magnesia, carbonate of iron, iodide ofsoda, and traces of silex, alumina, and bydrobromate ofpotasb. These solid contents passing over the ground, areprecipitated, and form a rock, as is most wonderfullyshown i

RM2CH28T1–. The Pacific tourist : Williams' illustrated trans-continental guide of travel, from the Atlantic to the Pacific Ocean : containing full descriptions of railroad routes ... A complete traveler's guide of the Union and Central Pacific railroads ... . ness in a platine capsule, gave8.25 grains of solid dry saline matter. Carbonate of lime and magnesia, Peroxide of iron, Lime. Chlorine, Soda, Magnesia, Sulphuric Acid, 8.229 43.981 It is slightly charged with hydro-sulphuric acidgas, and with carbonic acid gas, and is a pleas-ant, saline mineral water, having the valuableproperties belonging to a

RM2CH3TNE–. The Pacific tourist . s in a platine capsule, gave8.25 grains of solid dry saline matter. Ca bonate of lime and iiiagnesia, Peroxide of iron, Lime. Chlorine, Soda, Magnesia, Sulphur.c Acid, 8.229 43.981 It is slightly charged with hydro-sulphuric acidgas, and with carbonic acid gas, and is a pleas-ant, saline mineral water, having the valuableproperties belonging to a saline sulphur spring. The temperature is lukewarm, and, being ofa sulphurous nature, the effects are very pene-trating ; at first the sensation is delicious, pro-ducins a delightful feeling of ease and re- 0.240 1.280 0.040 0.

RM2CH2WWX–. The Pacific tourist . n a platine capsule, gave8.25 grains of solid dry saline matter. Carbonate of lime and magnesia, 0.240 1.280 Peroxide of iron, 0.040 0.208 Lime. 0..i45 2.907 Chlorine, 3.4.54 18421 Soda. 2.877 15..344 Magnesia, 0.370 2.073 Suiplmr.c Acid, 0.703 3.748 8.229 4.3.981 It is slightly charged with hydro-sulphuric acidgas, and with carbonic acid gas, and is a plea.s-ant, saline mineral water, having the valuableproperties belonging to a saline sulphur spring. The temperature is lukewarm, and, being ofa sulphurous nature, the effects are very pene-trating ; at first the sensati

RM2CGYJK2–. The Pacific tourist : Adams & Bishop's illustrated trans-continental guide of travel, from the Atlantic to the Pacific Ocean ... : a complete traveler's guide of the Union and Central Pacific railroads ... . ss in a platine capsule, gave8.25 grains of solid dry saline matter. Catbonate of lime and iiiagnesia, 0.240 1.280 Peroxide of iron, 0.040 0.208 Lime. 0..145 2.907 Chlorine, 3.454 18421 Soda. 2.877 15.344 Magnesia, 0.370 2.073 Sulphur.c Acid, 0.703 3.748 8.229 43.981 It is slightly charged with hydro-sulphuric acidgas, and with carbonic acid gas, and is a pleas-ant, saline mineral water,

RM2CR2KWC–. The Pacific tourist . yness in a platine capsule, gave8.25 grains of solid dry saline matter. Caibonateof lime and magm sia, 0.240 1.280 Peroxide of iron, 0.040 0.20S LErae, 0.M5 2.007 Chlorine, 3.454 18421 SoJa. 2.877 15.344 Magnesia, 0.370 2.073 Sulphuric Acid, 0.703 3.748 8.229 43.981 It is slightly charged with hydro-sulphuric acidgas, and with carbonic acid gas, and is a pleas-ant, saline mineral water, having the valuableproperties belonging to a saline sulphur spring. The temperature is lukewarm, ai*l, being ofa sulphurous nature, the effects are very pene-trating ; at first the sensa

RM2CGYDH2–. The Pacific tourist . .25 grains of solid dry saline matter. Cat>on;te of lime and luagui^aia, 0.240 1.280 Peroxide of iron. 0.040 0-.08 Lime. 0.--t5 2.;x»7 Clilorino, 3.4.V» 18 421 .S.kIh. 2.877 l.5..U4 Majjiiesia, c-no 2.07.J Sulphur.c Acid, 0.703 3.T48 8.229 4.3.981 It is .slightly charged with hydro-sulphuric acidgas, and with carbonic aciil gas, and is a ple.a.s-ant, saline mineral water, having the valualileproperties be|oni,ing to a saline sidihur spring. The temperature is lukewarm. uimI, being ofa sulphurous nature, the etTects are very pene-trating ; at first the .sensation is

RMRDPP5H–. My garden, its plan and culture together with a general description of its geology, botany, and natural history. Gardening. GENERAL PLAN OF THE GARDEN. 45 mill partakes of the character of a rapid, and here the water rising in the grounds of Wallington Flouse flows into the river by the Crystal Waterfall (fig. 42). The water falls over an edge of moss-grown slate, looking more like a sheet of glass than of water. This peculiar effect is due to the presence of chalk dissolved in carbonic acid, and to the total absence of any solid particles, as is usual in chalk waters coming from the depths
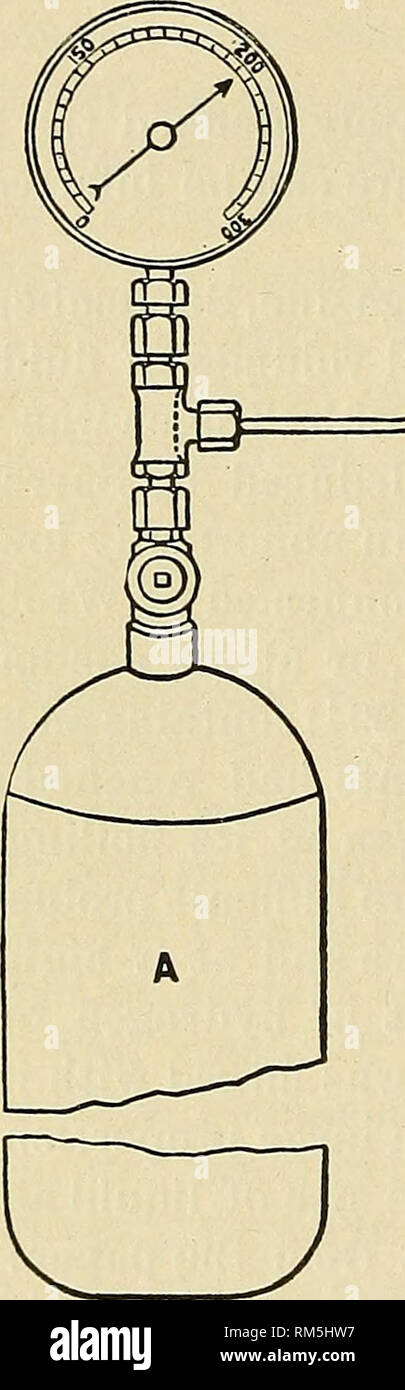
RMRM5HW7–. Annual report of the Board of Regents of the Smithsonian Institution. Smithsonian Institution; Smithsonian Institution. Archives; Discoveries in science. 262 LIQUEFACTIOM OF HYDROGEN AND HELIUM. of a jet of hydrogen containing visible liquid. This is reproduced in the figure. A represents one of the hydrogen cylinders; B and C, vacuum vessels containing carbonic acid under exhaustion and liquid air, respectively; D is the coil, G the pin-hole nozzle, and F the valve. By means of this jet, liquid air can be quickly transformed into a hard solid. It was shown that such a jet could be used to c
RMRDCH4P–. A manual of veterinary hygiene. Veterinary hygiene. VENTILATION 63 the most favourable circumstances, viz., where the air is obtained through cracks and crannies in doors and windows, diffusion is a slow process, and by itself can never be trusted for the purpose of renovating vitiated air. Diffusion merely causes the various gases present in a building to mix. Eoscoe showed that carbonic acid experi- mentally evolved in a room was reduced to one-half in ninety minutes by this process. This is not sufficiently rapid for the purpose of ventilation, and further, it leaves the solid particles o

RMRJD1AY–. The Central Rio Grande Valley of New Mexico. Agriculture. 46 THE CENTRAL RIO GRANDE VALLEY. Valle Grande, the Crown of Uie Jeniez Range, Sandoval County with temperature of 103 degrees Fahrenheit, carries free carbonic acid gas and its deposit is a reddish brown, while a third spring, temperature 119 degrees Fahrenheit, is impregnated with sulphurated hydrogen and iron. The other springs carry sodium, lime and magnesium. The solid constituents are about .24 per one one hundred parts of water. The upper group, or Sulphur springs ,are two miles above the lower group at an altitude 'of 6,740 fe
Search Results for Solid carbonic acid Stock Photos and Images (49)
Page 1 of 1
















