Quick filters:
Page 1 of 2
Potassium bromide Stock Photos and Images
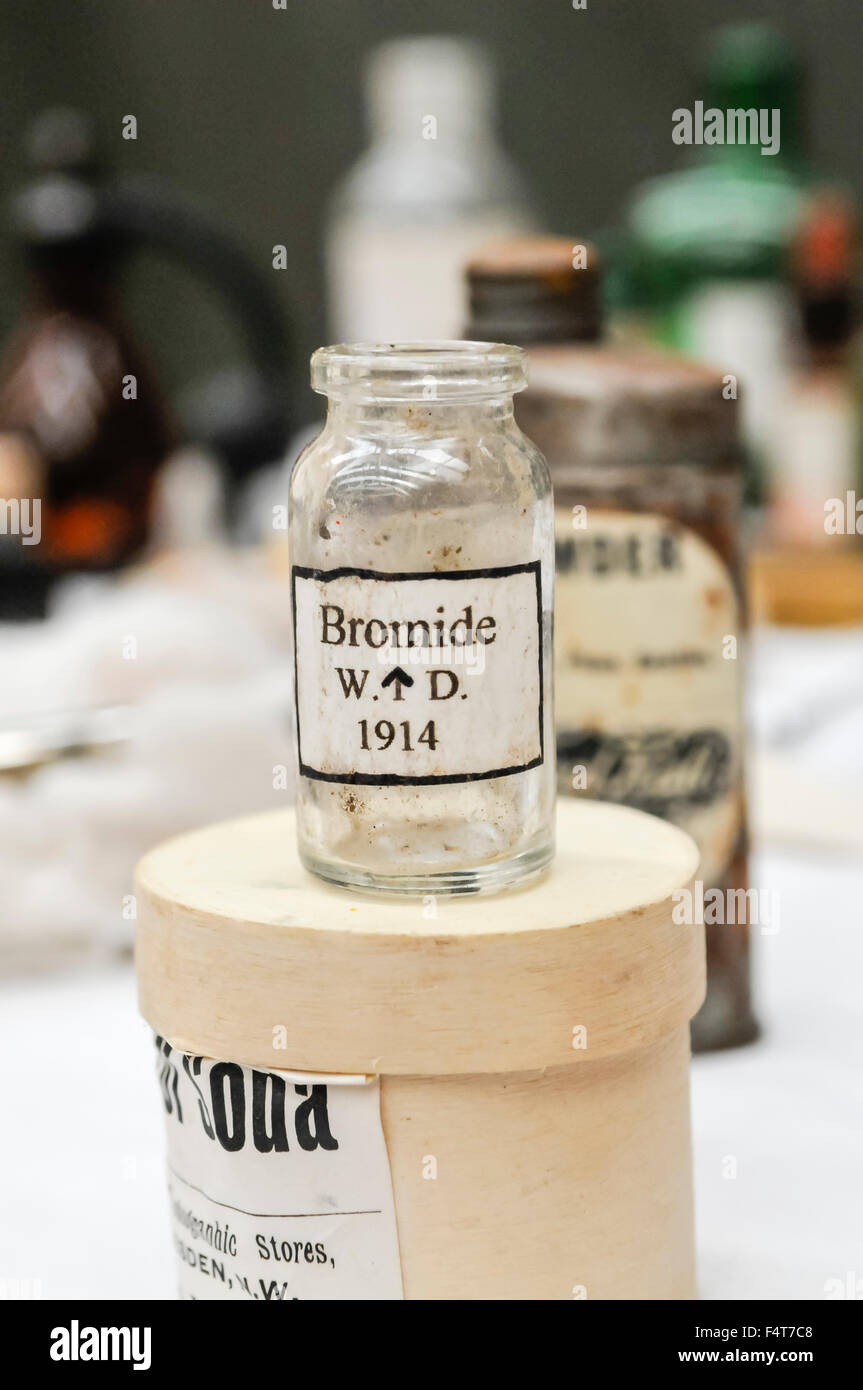 Bromide bottle dating from 1914. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stock-photo-bromide-bottle-dating-from-1914-89043144.html
Bromide bottle dating from 1914. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stock-photo-bromide-bottle-dating-from-1914-89043144.htmlRMF4T7C8–Bromide bottle dating from 1914.
 Bromism is the syndrome which results from the long-term consumption of bromine, usually through bromide-based sedatives such as potassium bromide and lithium bromide. Bromism was once a very common disorder, being responsible for 5 to 10% of psychiatric hospital admissions, but is now uncommon since bromide was withdrawn from clinical use in many countries and was severely restricted in others. Dermatological effects include cherry angiomas, acneiform, pustular and erythematous rashes. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/bromism-is-the-syndrome-which-results-from-the-long-term-consumption-of-bromine-usually-through-bromide-based-sedatives-such-as-potassium-bromide-and-lithium-bromide-bromism-was-once-a-very-common-disorder-being-responsible-for-5-to-10-of-psychiatric-hospital-admissions-but-is-now-uncommon-since-bromide-was-withdrawn-from-clinical-use-in-many-countries-and-was-severely-restricted-in-others-dermatological-effects-include-cherry-angiomas-acneiform-pustular-and-erythematous-rashes-image458814117.html
Bromism is the syndrome which results from the long-term consumption of bromine, usually through bromide-based sedatives such as potassium bromide and lithium bromide. Bromism was once a very common disorder, being responsible for 5 to 10% of psychiatric hospital admissions, but is now uncommon since bromide was withdrawn from clinical use in many countries and was severely restricted in others. Dermatological effects include cherry angiomas, acneiform, pustular and erythematous rashes. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/bromism-is-the-syndrome-which-results-from-the-long-term-consumption-of-bromine-usually-through-bromide-based-sedatives-such-as-potassium-bromide-and-lithium-bromide-bromism-was-once-a-very-common-disorder-being-responsible-for-5-to-10-of-psychiatric-hospital-admissions-but-is-now-uncommon-since-bromide-was-withdrawn-from-clinical-use-in-many-countries-and-was-severely-restricted-in-others-dermatological-effects-include-cherry-angiomas-acneiform-pustular-and-erythematous-rashes-image458814117.htmlRM2HJCP2D–Bromism is the syndrome which results from the long-term consumption of bromine, usually through bromide-based sedatives such as potassium bromide and lithium bromide. Bromism was once a very common disorder, being responsible for 5 to 10% of psychiatric hospital admissions, but is now uncommon since bromide was withdrawn from clinical use in many countries and was severely restricted in others. Dermatological effects include cherry angiomas, acneiform, pustular and erythematous rashes.
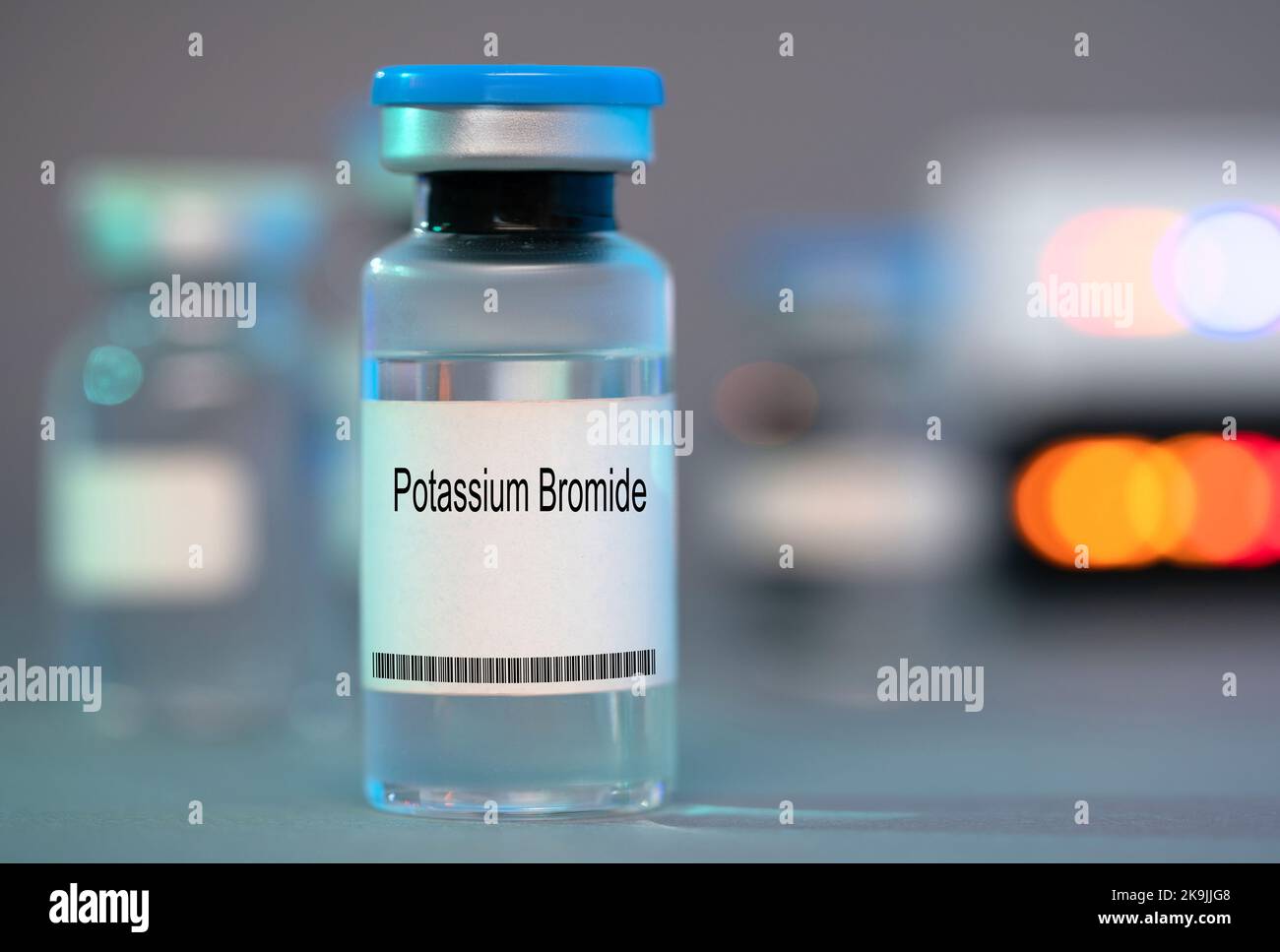 Vial of potassium bromide Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/vial-of-potassium-bromide-image487831912.html
Vial of potassium bromide Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/vial-of-potassium-bromide-image487831912.htmlRF2K9JJG8–Vial of potassium bromide
 Bottled crystals of potassium bromide Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/bottled-crystals-of-potassium-bromide-image216090741.html
Bottled crystals of potassium bromide Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/bottled-crystals-of-potassium-bromide-image216090741.htmlRMPFFNXD–Bottled crystals of potassium bromide
 Stem of Leaves and Flowers, c. 1835/37, William Henry Fox Talbot, English, 1800–1877, England, Photogenic drawing, stabilized (fixed) in potassium iodide, potassium bromide or ammonia water, 11.3 × 8.5 cm (image/paper Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stem-of-leaves-and-flowers-c-183537-william-henry-fox-talbot-english-18001877-england-photogenic-drawing-stabilized-fixed-in-potassium-iodide-potassium-bromide-or-ammonia-water-113-85-cm-imagepaper-image328647505.html
Stem of Leaves and Flowers, c. 1835/37, William Henry Fox Talbot, English, 1800–1877, England, Photogenic drawing, stabilized (fixed) in potassium iodide, potassium bromide or ammonia water, 11.3 × 8.5 cm (image/paper Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stem-of-leaves-and-flowers-c-183537-william-henry-fox-talbot-english-18001877-england-photogenic-drawing-stabilized-fixed-in-potassium-iodide-potassium-bromide-or-ammonia-water-113-85-cm-imagepaper-image328647505.htmlRM2A2K56W–Stem of Leaves and Flowers, c. 1835/37, William Henry Fox Talbot, English, 1800–1877, England, Photogenic drawing, stabilized (fixed) in potassium iodide, potassium bromide or ammonia water, 11.3 × 8.5 cm (image/paper
 Stem of Leaves and Flowers. William Henry Fox Talbot; English, 1800-1877. Date: 1830-1842. Dimensions: 11.3 × 8.5 cm (image/paper). Photogenic drawing, stabilized (fixed) in potassium iodide, potassium bromide or ammonia water. Origin: England. Museum: The Chicago Art Institute. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stem-of-leaves-and-flowers-william-henry-fox-talbot-english-1800-1877-date-1830-1842-dimensions-113-85-cm-imagepaper-photogenic-drawing-stabilized-fixed-in-potassium-iodide-potassium-bromide-or-ammonia-water-origin-england-museum-the-chicago-art-institute-image240139829.html
Stem of Leaves and Flowers. William Henry Fox Talbot; English, 1800-1877. Date: 1830-1842. Dimensions: 11.3 × 8.5 cm (image/paper). Photogenic drawing, stabilized (fixed) in potassium iodide, potassium bromide or ammonia water. Origin: England. Museum: The Chicago Art Institute. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stem-of-leaves-and-flowers-william-henry-fox-talbot-english-1800-1877-date-1830-1842-dimensions-113-85-cm-imagepaper-photogenic-drawing-stabilized-fixed-in-potassium-iodide-potassium-bromide-or-ammonia-water-origin-england-museum-the-chicago-art-institute-image240139829.htmlRMRXK8PD–Stem of Leaves and Flowers. William Henry Fox Talbot; English, 1800-1877. Date: 1830-1842. Dimensions: 11.3 × 8.5 cm (image/paper). Photogenic drawing, stabilized (fixed) in potassium iodide, potassium bromide or ammonia water. Origin: England. Museum: The Chicago Art Institute.
 KBr - Potassium Bromide. Chemical compound. CAS number 7758-02-3 Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/kbr-potassium-bromide-chemical-compound-cas-number-7758-02-3-image595350915.html
KBr - Potassium Bromide. Chemical compound. CAS number 7758-02-3 Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/kbr-potassium-bromide-chemical-compound-cas-number-7758-02-3-image595350915.htmlRF2WGGG4K–KBr - Potassium Bromide. Chemical compound. CAS number 7758-02-3
RM2F77BEN–Skiopticon image from the Department of Photography at the Royal Institute of Technology. Use by Professor Helmer Bäckström as lecture material. Bäckström was Sweden's first professor in photography at the Royal Institute of Technology in Stockholm 1948-1958.Typical curve families, obtained with various developing times for deep developing with potassium bromide additive. The straight parts of the curves have been pulled out to their intersection. For more info see: Bäckström, Helmer. Photographic manual. Nature and Culture. Stockholm. 1942. p. 272.
 William Henry Fox Talbot. Stem of Leaves and Flowers. 1830–1842. England. Photogenic drawing, stabilized (fixed) in potassium iodide, potassium bromide or ammonia water Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/william-henry-fox-talbot-stem-of-leaves-and-flowers-18301842-england-photogenic-drawing-stabilized-fixed-in-potassium-iodide-potassium-bromide-or-ammonia-water-image337975192.html
William Henry Fox Talbot. Stem of Leaves and Flowers. 1830–1842. England. Photogenic drawing, stabilized (fixed) in potassium iodide, potassium bromide or ammonia water Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/william-henry-fox-talbot-stem-of-leaves-and-flowers-18301842-england-photogenic-drawing-stabilized-fixed-in-potassium-iodide-potassium-bromide-or-ammonia-water-image337975192.htmlRM2AHT2PG–William Henry Fox Talbot. Stem of Leaves and Flowers. 1830–1842. England. Photogenic drawing, stabilized (fixed) in potassium iodide, potassium bromide or ammonia water
 Stem of Leaves and Flowers, c. 1835/37. A work made of photogenic drawing, stabilized (fixed) in potassium iodide, potassium bromide or ammonia water. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stem-of-leaves-and-flowers-c-183537-a-work-made-of-photogenic-drawing-stabilized-fixed-in-potassium-iodide-potassium-bromide-or-ammonia-water-image440345464.html
Stem of Leaves and Flowers, c. 1835/37. A work made of photogenic drawing, stabilized (fixed) in potassium iodide, potassium bromide or ammonia water. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stem-of-leaves-and-flowers-c-183537-a-work-made-of-photogenic-drawing-stabilized-fixed-in-potassium-iodide-potassium-bromide-or-ammonia-water-image440345464.htmlRM2GGBD3M–Stem of Leaves and Flowers, c. 1835/37. A work made of photogenic drawing, stabilized (fixed) in potassium iodide, potassium bromide or ammonia water.
 Bottles of chemcicals on shelves in a Science laboratory including Potassium Iodide, Potassium Chloride, Potassium bromide, Sodium nitrate, Chemicals Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/bottles-of-chemcicals-on-shelves-in-a-science-laboratory-including-potassium-iodide-potassium-chloride-potassium-bromide-sodium-nitrate-chemicals-image220391317.html
Bottles of chemcicals on shelves in a Science laboratory including Potassium Iodide, Potassium Chloride, Potassium bromide, Sodium nitrate, Chemicals Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/bottles-of-chemcicals-on-shelves-in-a-science-laboratory-including-potassium-iodide-potassium-chloride-potassium-bromide-sodium-nitrate-chemicals-image220391317.htmlRFPPFKAD–Bottles of chemcicals on shelves in a Science laboratory including Potassium Iodide, Potassium Chloride, Potassium bromide, Sodium nitrate, Chemicals
 War surgery of the faceA treatise on plastic restoration after facial injury by John BRoberts ..Prepared at the suggestion of the subsection on plastic and oral surgery connected with the office of the surgeon generalIllustrated with 256 figures . Fig. 83.— In this specimen the sinus extends nearly to region of caninetooth. (From M. H. Cryer.) 200 WAR SURGERY OF THE FACE. tympanum, and tests of the nerve distributions should be part ofthe surgeons routine investigation. Purgation, elevation of the head, administration of hexa-methylenamin and potassium bromide are advantageous at suchtimes. Tr Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/war-surgery-of-the-facea-treatise-on-plastic-restoration-after-facial-injury-by-john-broberts-prepared-at-the-suggestion-of-the-subsection-on-plastic-and-oral-surgery-connected-with-the-office-of-the-surgeon-generalillustrated-with-256-figures-fig-83-in-this-specimen-the-sinus-extends-nearly-to-region-of-caninetooth-from-m-h-cryer-200-war-surgery-of-the-face-tympanum-and-tests-of-the-nerve-distributions-should-be-part-ofthe-surgeons-routine-investigation-purgation-elevation-of-the-head-administration-of-hexa-methylenamin-and-potassium-bromide-are-advantageous-at-suchtimes-tr-image339205308.html
War surgery of the faceA treatise on plastic restoration after facial injury by John BRoberts ..Prepared at the suggestion of the subsection on plastic and oral surgery connected with the office of the surgeon generalIllustrated with 256 figures . Fig. 83.— In this specimen the sinus extends nearly to region of caninetooth. (From M. H. Cryer.) 200 WAR SURGERY OF THE FACE. tympanum, and tests of the nerve distributions should be part ofthe surgeons routine investigation. Purgation, elevation of the head, administration of hexa-methylenamin and potassium bromide are advantageous at suchtimes. Tr Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/war-surgery-of-the-facea-treatise-on-plastic-restoration-after-facial-injury-by-john-broberts-prepared-at-the-suggestion-of-the-subsection-on-plastic-and-oral-surgery-connected-with-the-office-of-the-surgeon-generalillustrated-with-256-figures-fig-83-in-this-specimen-the-sinus-extends-nearly-to-region-of-caninetooth-from-m-h-cryer-200-war-surgery-of-the-face-tympanum-and-tests-of-the-nerve-distributions-should-be-part-ofthe-surgeons-routine-investigation-purgation-elevation-of-the-head-administration-of-hexa-methylenamin-and-potassium-bromide-are-advantageous-at-suchtimes-tr-image339205308.htmlRM2AKT3R8–War surgery of the faceA treatise on plastic restoration after facial injury by John BRoberts ..Prepared at the suggestion of the subsection on plastic and oral surgery connected with the office of the surgeon generalIllustrated with 256 figures . Fig. 83.— In this specimen the sinus extends nearly to region of caninetooth. (From M. H. Cryer.) 200 WAR SURGERY OF THE FACE. tympanum, and tests of the nerve distributions should be part ofthe surgeons routine investigation. Purgation, elevation of the head, administration of hexa-methylenamin and potassium bromide are advantageous at suchtimes. Tr
 Stem of Leaves and Flowers, c. 1835/37, William Henry Fox Talbot, English, 1800–1877, England, Photogenic drawing, stabilized (fixed) in potassium iodide, potassium bromide or ammonia water, 11.3 × 8.5 cm (image/paper), Reimagined by Gibon, design of warm cheerful glowing of brightness and light rays radiance. Classic art reinvented with a modern twist. Photography inspired by futurism, embracing dynamic energy of modern technology, movement, speed and revolutionize culture. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stem-of-leaves-and-flowers-c-183537-william-henry-fox-talbot-english-18001877-england-photogenic-drawing-stabilized-fixed-in-potassium-iodide-potassium-bromide-or-ammonia-water-113-85-cm-imagepaper-reimagined-by-gibon-design-of-warm-cheerful-glowing-of-brightness-and-light-rays-radiance-classic-art-reinvented-with-a-modern-twist-photography-inspired-by-futurism-embracing-dynamic-energy-of-modern-technology-movement-speed-and-revolutionize-culture-image349725221.html
Stem of Leaves and Flowers, c. 1835/37, William Henry Fox Talbot, English, 1800–1877, England, Photogenic drawing, stabilized (fixed) in potassium iodide, potassium bromide or ammonia water, 11.3 × 8.5 cm (image/paper), Reimagined by Gibon, design of warm cheerful glowing of brightness and light rays radiance. Classic art reinvented with a modern twist. Photography inspired by futurism, embracing dynamic energy of modern technology, movement, speed and revolutionize culture. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stem-of-leaves-and-flowers-c-183537-william-henry-fox-talbot-english-18001877-england-photogenic-drawing-stabilized-fixed-in-potassium-iodide-potassium-bromide-or-ammonia-water-113-85-cm-imagepaper-reimagined-by-gibon-design-of-warm-cheerful-glowing-of-brightness-and-light-rays-radiance-classic-art-reinvented-with-a-modern-twist-photography-inspired-by-futurism-embracing-dynamic-energy-of-modern-technology-movement-speed-and-revolutionize-culture-image349725221.htmlRF2B8YA2D–Stem of Leaves and Flowers, c. 1835/37, William Henry Fox Talbot, English, 1800–1877, England, Photogenic drawing, stabilized (fixed) in potassium iodide, potassium bromide or ammonia water, 11.3 × 8.5 cm (image/paper), Reimagined by Gibon, design of warm cheerful glowing of brightness and light rays radiance. Classic art reinvented with a modern twist. Photography inspired by futurism, embracing dynamic energy of modern technology, movement, speed and revolutionize culture.
 Lab prep of Bromine Manganese dioxide plus HCl remove xs HCl pass through water & conc sulphuric then bubble through KBr soln Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/lab-prep-of-bromine-manganese-dioxide-plus-hcl-remove-xs-hcl-pass-image1417773.html
Lab prep of Bromine Manganese dioxide plus HCl remove xs HCl pass through water & conc sulphuric then bubble through KBr soln Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/lab-prep-of-bromine-manganese-dioxide-plus-hcl-remove-xs-hcl-pass-image1417773.htmlRMANA22E–Lab prep of Bromine Manganese dioxide plus HCl remove xs HCl pass through water & conc sulphuric then bubble through KBr soln
 Old commercial advertisements. Medicines and medical remedies. La Ilustracion Artitsica, January 1893. Spain. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stock-photo-old-commercial-advertisements-medicines-and-medical-remedies-la-ilustracion-176787371.html
Old commercial advertisements. Medicines and medical remedies. La Ilustracion Artitsica, January 1893. Spain. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stock-photo-old-commercial-advertisements-medicines-and-medical-remedies-la-ilustracion-176787371.htmlRMM7HA2K–Old commercial advertisements. Medicines and medical remedies. La Ilustracion Artitsica, January 1893. Spain.
 Overlooking the Dead Sea's emerald green water and rugged, salt crusted bank. The white and tan salt crystals are bumpy and rough. From above. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stock-photo-overlooking-the-dead-seas-emerald-green-water-and-rugged-salt-crusted-140752077.html
Overlooking the Dead Sea's emerald green water and rugged, salt crusted bank. The white and tan salt crystals are bumpy and rough. From above. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stock-photo-overlooking-the-dead-seas-emerald-green-water-and-rugged-salt-crusted-140752077.htmlRFJ4YPKW–Overlooking the Dead Sea's emerald green water and rugged, salt crusted bank. The white and tan salt crystals are bumpy and rough. From above.
 Sodium molybdate is an inorganic compound. It is a source of molybdenum, foliar fertilizer applied both in seed treatment and foliar application Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-molybdate-is-an-inorganic-compound-it-is-a-source-of-molybdenum-foliar-fertilizer-applied-both-in-seed-treatment-and-foliar-application-image561540960.html
Sodium molybdate is an inorganic compound. It is a source of molybdenum, foliar fertilizer applied both in seed treatment and foliar application Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-molybdate-is-an-inorganic-compound-it-is-a-source-of-molybdenum-foliar-fertilizer-applied-both-in-seed-treatment-and-foliar-application-image561540960.htmlRF2RHGB68–Sodium molybdate is an inorganic compound. It is a source of molybdenum, foliar fertilizer applied both in seed treatment and foliar application
 Close up of Dead Sea salt covering a piece of driftwood in salt saturated sand with the aquamarine colored Dead Sea in background and blue sky above. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stock-photo-close-up-of-dead-sea-salt-covering-a-piece-of-driftwood-in-salt-saturated-140752278.html
Close up of Dead Sea salt covering a piece of driftwood in salt saturated sand with the aquamarine colored Dead Sea in background and blue sky above. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stock-photo-close-up-of-dead-sea-salt-covering-a-piece-of-driftwood-in-salt-saturated-140752278.htmlRFJ4YPY2–Close up of Dead Sea salt covering a piece of driftwood in salt saturated sand with the aquamarine colored Dead Sea in background and blue sky above.
 Sodium molybdate is an inorganic compound. It is a source of molybdenum, foliar fertilizer applied both in seed treatment and foliar application Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-molybdate-is-an-inorganic-compound-it-is-a-source-of-molybdenum-foliar-fertilizer-applied-both-in-seed-treatment-and-foliar-application-image561540949.html
Sodium molybdate is an inorganic compound. It is a source of molybdenum, foliar fertilizer applied both in seed treatment and foliar application Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-molybdate-is-an-inorganic-compound-it-is-a-source-of-molybdenum-foliar-fertilizer-applied-both-in-seed-treatment-and-foliar-application-image561540949.htmlRF2RHGB5W–Sodium molybdate is an inorganic compound. It is a source of molybdenum, foliar fertilizer applied both in seed treatment and foliar application
 Selection of medicine bottles including Epsom salts, Bromide and Paregoric (an opiate used to treat diarrhoea) from 1914 Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stock-photo-selection-of-medicine-bottles-including-epsom-salts-bromide-and-paregoric-89043128.html
Selection of medicine bottles including Epsom salts, Bromide and Paregoric (an opiate used to treat diarrhoea) from 1914 Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stock-photo-selection-of-medicine-bottles-including-epsom-salts-bromide-and-paregoric-89043128.htmlRMF4T7BM–Selection of medicine bottles including Epsom salts, Bromide and Paregoric (an opiate used to treat diarrhoea) from 1914
 A WEEKLY JOURNAL OF PRACTICAL INFORMATION ART SCIENCE MECHANICS CHEMISTRY AND MANUFACTURES ifils per Annum 4.-41 1 Improvement in Malt and Grain Kilns. ADJUSTABLE INDICATOR FOR ROUNDING SAWS. An Alarmipg Theory. Stub Twist Gun Barrels. Bromide of Potassium., scientific american, 1868-06-06 Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/a-weekly-journal-of-practical-information-art-science-mechanics-chemistry-and-manufactures-ifils-per-annum-4-41-1-improvement-in-malt-and-grain-kilns-adjustable-indicator-for-rounding-saws-an-alarmipg-theory-stub-twist-gun-barrels-bromide-of-potassium-scientific-american-1868-06-06-image334309433.html
A WEEKLY JOURNAL OF PRACTICAL INFORMATION ART SCIENCE MECHANICS CHEMISTRY AND MANUFACTURES ifils per Annum 4.-41 1 Improvement in Malt and Grain Kilns. ADJUSTABLE INDICATOR FOR ROUNDING SAWS. An Alarmipg Theory. Stub Twist Gun Barrels. Bromide of Potassium., scientific american, 1868-06-06 Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/a-weekly-journal-of-practical-information-art-science-mechanics-chemistry-and-manufactures-ifils-per-annum-4-41-1-improvement-in-malt-and-grain-kilns-adjustable-indicator-for-rounding-saws-an-alarmipg-theory-stub-twist-gun-barrels-bromide-of-potassium-scientific-american-1868-06-06-image334309433.htmlRM2ABW32H–A WEEKLY JOURNAL OF PRACTICAL INFORMATION ART SCIENCE MECHANICS CHEMISTRY AND MANUFACTURES ifils per Annum 4.-41 1 Improvement in Malt and Grain Kilns. ADJUSTABLE INDICATOR FOR ROUNDING SAWS. An Alarmipg Theory. Stub Twist Gun Barrels. Bromide of Potassium., scientific american, 1868-06-06
 3D illustration of 'Sodium Fluoride ' title on pill bottle, isolated on white. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/3d-illustration-of-sodium-fluoride-title-on-pill-bottle-isolated-on-image155665165.html
3D illustration of 'Sodium Fluoride ' title on pill bottle, isolated on white. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/3d-illustration-of-sodium-fluoride-title-on-pill-bottle-isolated-on-image155665165.htmlRFK174E5–3D illustration of 'Sodium Fluoride ' title on pill bottle, isolated on white.
 Table Set For Tea, c. 1840, William Henry Fox Talbot, English, 1800–1877, England, Photogenic drawing print from photogenic drawing negative, stabilized (fixed) in potassium iodide or potassium bromide, 7.7 × 16.1 cm (image), 11.1 × 18.7 cm (paper Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/table-set-for-tea-c-1840-william-henry-fox-talbot-english-18001877-england-photogenic-drawing-print-from-photogenic-drawing-negative-stabilized-fixed-in-potassium-iodide-or-potassium-bromide-77-161-cm-image-111-187-cm-paper-image328647497.html
Table Set For Tea, c. 1840, William Henry Fox Talbot, English, 1800–1877, England, Photogenic drawing print from photogenic drawing negative, stabilized (fixed) in potassium iodide or potassium bromide, 7.7 × 16.1 cm (image), 11.1 × 18.7 cm (paper Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/table-set-for-tea-c-1840-william-henry-fox-talbot-english-18001877-england-photogenic-drawing-print-from-photogenic-drawing-negative-stabilized-fixed-in-potassium-iodide-or-potassium-bromide-77-161-cm-image-111-187-cm-paper-image328647497.htmlRM2A2K56H–Table Set For Tea, c. 1840, William Henry Fox Talbot, English, 1800–1877, England, Photogenic drawing print from photogenic drawing negative, stabilized (fixed) in potassium iodide or potassium bromide, 7.7 × 16.1 cm (image), 11.1 × 18.7 cm (paper
 Table Set For Tea. William Henry Fox Talbot; English, 1800-1877. Date: 1835-1845. Dimensions: 7.7 × 16.1 cm (image); 11.1 × 18.7 cm (paper). Photogenic drawing print from photogenic drawing negative, stabilized (fixed) in potassium iodide or potassium bromide. Origin: England. Museum: The Chicago Art Institute. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/table-set-for-tea-william-henry-fox-talbot-english-1800-1877-date-1835-1845-dimensions-77-161-cm-image-111-187-cm-paper-photogenic-drawing-print-from-photogenic-drawing-negative-stabilized-fixed-in-potassium-iodide-or-potassium-bromide-origin-england-museum-the-chicago-art-institute-image240137324.html
Table Set For Tea. William Henry Fox Talbot; English, 1800-1877. Date: 1835-1845. Dimensions: 7.7 × 16.1 cm (image); 11.1 × 18.7 cm (paper). Photogenic drawing print from photogenic drawing negative, stabilized (fixed) in potassium iodide or potassium bromide. Origin: England. Museum: The Chicago Art Institute. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/table-set-for-tea-william-henry-fox-talbot-english-1800-1877-date-1835-1845-dimensions-77-161-cm-image-111-187-cm-paper-photogenic-drawing-print-from-photogenic-drawing-negative-stabilized-fixed-in-potassium-iodide-or-potassium-bromide-origin-england-museum-the-chicago-art-institute-image240137324.htmlRMRXK5H0–Table Set For Tea. William Henry Fox Talbot; English, 1800-1877. Date: 1835-1845. Dimensions: 7.7 × 16.1 cm (image); 11.1 × 18.7 cm (paper). Photogenic drawing print from photogenic drawing negative, stabilized (fixed) in potassium iodide or potassium bromide. Origin: England. Museum: The Chicago Art Institute.
 KBr potassium bromide CAS 7758-02-3 chemical substance in white plastic laboratory packaging Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/kbr-potassium-bromide-cas-7758-02-3-chemical-substance-in-white-plastic-laboratory-packaging-image542667037.html
KBr potassium bromide CAS 7758-02-3 chemical substance in white plastic laboratory packaging Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/kbr-potassium-bromide-cas-7758-02-3-chemical-substance-in-white-plastic-laboratory-packaging-image542667037.htmlRF2PETH9H–KBr potassium bromide CAS 7758-02-3 chemical substance in white plastic laboratory packaging
 William Henry Fox Talbot. Table Set For Tea. 1835–1845. England. Photogenic drawing print from photogenic drawing negative, stabilized (fixed) in potassium iodide or potassium bromide Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/william-henry-fox-talbot-table-set-for-tea-18351845-england-photogenic-drawing-print-from-photogenic-drawing-negative-stabilized-fixed-in-potassium-iodide-or-potassium-bromide-image337975148.html
William Henry Fox Talbot. Table Set For Tea. 1835–1845. England. Photogenic drawing print from photogenic drawing negative, stabilized (fixed) in potassium iodide or potassium bromide Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/william-henry-fox-talbot-table-set-for-tea-18351845-england-photogenic-drawing-print-from-photogenic-drawing-negative-stabilized-fixed-in-potassium-iodide-or-potassium-bromide-image337975148.htmlRM2AHT2N0–William Henry Fox Talbot. Table Set For Tea. 1835–1845. England. Photogenic drawing print from photogenic drawing negative, stabilized (fixed) in potassium iodide or potassium bromide
 Bottles of chemcicals on shelves in a Science laboratory including Potassium Iodide, Potassium Chloride, Potassium bromide, Sodium nitrate, Chemicals Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/bottles-of-chemcicals-on-shelves-in-a-science-laboratory-including-potassium-iodide-potassium-chloride-potassium-bromide-sodium-nitrate-chemicals-image220391316.html
Bottles of chemcicals on shelves in a Science laboratory including Potassium Iodide, Potassium Chloride, Potassium bromide, Sodium nitrate, Chemicals Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/bottles-of-chemcicals-on-shelves-in-a-science-laboratory-including-potassium-iodide-potassium-chloride-potassium-bromide-sodium-nitrate-chemicals-image220391316.htmlRFPPFKAC–Bottles of chemcicals on shelves in a Science laboratory including Potassium Iodide, Potassium Chloride, Potassium bromide, Sodium nitrate, Chemicals
 The American annual of photography . e, which is very sensitive to bromide, shadow detail inthe negative is lost. Pyro, Elon, and Ortol are less sensitiveto bromide and should therefore predominate in a negativedeveloper. Pyro oxidizes rapidly and gives stain unless anexcess of sulphite is added, which in turn tends to producefog. For motion picture positive work the effect of the ac-cumulated potassium bromide is of less importance, since thiscan be cornpensated for by increasing the printing exposure. In tank work, as the developer becomes exhausted as a resultof performing useful work, it i Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-american-annual-of-photography-e-which-is-very-sensitive-to-bromide-shadow-detail-inthe-negative-is-lost-pyro-elon-and-ortol-are-less-sensitiveto-bromide-and-should-therefore-predominate-in-a-negativedeveloper-pyro-oxidizes-rapidly-and-gives-stain-unless-anexcess-of-sulphite-is-added-which-in-turn-tends-to-producefog-for-motion-picture-positive-work-the-effect-of-the-ac-cumulated-potassium-bromide-is-of-less-importance-since-thiscan-be-cornpensated-for-by-increasing-the-printing-exposure-in-tank-work-as-the-developer-becomes-exhausted-as-a-resultof-performing-useful-work-it-i-image339975211.html
The American annual of photography . e, which is very sensitive to bromide, shadow detail inthe negative is lost. Pyro, Elon, and Ortol are less sensitiveto bromide and should therefore predominate in a negativedeveloper. Pyro oxidizes rapidly and gives stain unless anexcess of sulphite is added, which in turn tends to producefog. For motion picture positive work the effect of the ac-cumulated potassium bromide is of less importance, since thiscan be cornpensated for by increasing the printing exposure. In tank work, as the developer becomes exhausted as a resultof performing useful work, it i Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-american-annual-of-photography-e-which-is-very-sensitive-to-bromide-shadow-detail-inthe-negative-is-lost-pyro-elon-and-ortol-are-less-sensitiveto-bromide-and-should-therefore-predominate-in-a-negativedeveloper-pyro-oxidizes-rapidly-and-gives-stain-unless-anexcess-of-sulphite-is-added-which-in-turn-tends-to-producefog-for-motion-picture-positive-work-the-effect-of-the-ac-cumulated-potassium-bromide-is-of-less-importance-since-thiscan-be-cornpensated-for-by-increasing-the-printing-exposure-in-tank-work-as-the-developer-becomes-exhausted-as-a-resultof-performing-useful-work-it-i-image339975211.htmlRM2AN35RR–The American annual of photography . e, which is very sensitive to bromide, shadow detail inthe negative is lost. Pyro, Elon, and Ortol are less sensitiveto bromide and should therefore predominate in a negativedeveloper. Pyro oxidizes rapidly and gives stain unless anexcess of sulphite is added, which in turn tends to producefog. For motion picture positive work the effect of the ac-cumulated potassium bromide is of less importance, since thiscan be cornpensated for by increasing the printing exposure. In tank work, as the developer becomes exhausted as a resultof performing useful work, it i
 Table Set For Tea, c. 1840, William Henry Fox Talbot, English, 1800–1877, England, Photogenic drawing print from photogenic drawing negative, stabilized (fixed) in potassium iodide or potassium bromide, 7.7 × 16.1 cm (image), 11.1 × 18.7 cm (paper), Reimagined by Gibon, design of warm cheerful glowing of brightness and light rays radiance. Classic art reinvented with a modern twist. Photography inspired by futurism, embracing dynamic energy of modern technology, movement, speed and revolutionize culture. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/table-set-for-tea-c-1840-william-henry-fox-talbot-english-18001877-england-photogenic-drawing-print-from-photogenic-drawing-negative-stabilized-fixed-in-potassium-iodide-or-potassium-bromide-77-161-cm-image-111-187-cm-paper-reimagined-by-gibon-design-of-warm-cheerful-glowing-of-brightness-and-light-rays-radiance-classic-art-reinvented-with-a-modern-twist-photography-inspired-by-futurism-embracing-dynamic-energy-of-modern-technology-movement-speed-and-revolutionize-culture-image349725211.html
Table Set For Tea, c. 1840, William Henry Fox Talbot, English, 1800–1877, England, Photogenic drawing print from photogenic drawing negative, stabilized (fixed) in potassium iodide or potassium bromide, 7.7 × 16.1 cm (image), 11.1 × 18.7 cm (paper), Reimagined by Gibon, design of warm cheerful glowing of brightness and light rays radiance. Classic art reinvented with a modern twist. Photography inspired by futurism, embracing dynamic energy of modern technology, movement, speed and revolutionize culture. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/table-set-for-tea-c-1840-william-henry-fox-talbot-english-18001877-england-photogenic-drawing-print-from-photogenic-drawing-negative-stabilized-fixed-in-potassium-iodide-or-potassium-bromide-77-161-cm-image-111-187-cm-paper-reimagined-by-gibon-design-of-warm-cheerful-glowing-of-brightness-and-light-rays-radiance-classic-art-reinvented-with-a-modern-twist-photography-inspired-by-futurism-embracing-dynamic-energy-of-modern-technology-movement-speed-and-revolutionize-culture-image349725211.htmlRF2B8YA23–Table Set For Tea, c. 1840, William Henry Fox Talbot, English, 1800–1877, England, Photogenic drawing print from photogenic drawing negative, stabilized (fixed) in potassium iodide or potassium bromide, 7.7 × 16.1 cm (image), 11.1 × 18.7 cm (paper), Reimagined by Gibon, design of warm cheerful glowing of brightness and light rays radiance. Classic art reinvented with a modern twist. Photography inspired by futurism, embracing dynamic energy of modern technology, movement, speed and revolutionize culture.
 Close up of Dead Sea salt covering a piece of driftwood in salt saturated sand with the aquamarine colored Dead Sea in background and blue sky above. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stock-photo-close-up-of-dead-sea-salt-covering-a-piece-of-driftwood-in-salt-saturated-140688216.html
Close up of Dead Sea salt covering a piece of driftwood in salt saturated sand with the aquamarine colored Dead Sea in background and blue sky above. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stock-photo-close-up-of-dead-sea-salt-covering-a-piece-of-driftwood-in-salt-saturated-140688216.htmlRFJ4TW74–Close up of Dead Sea salt covering a piece of driftwood in salt saturated sand with the aquamarine colored Dead Sea in background and blue sky above.
 Sodium molybdate is an inorganic compound. It is a source of molybdenum, foliar fertilizer applied both in seed treatment and foliar application Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-molybdate-is-an-inorganic-compound-it-is-a-source-of-molybdenum-foliar-fertilizer-applied-both-in-seed-treatment-and-foliar-application-image561540943.html
Sodium molybdate is an inorganic compound. It is a source of molybdenum, foliar fertilizer applied both in seed treatment and foliar application Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-molybdate-is-an-inorganic-compound-it-is-a-source-of-molybdenum-foliar-fertilizer-applied-both-in-seed-treatment-and-foliar-application-image561540943.htmlRF2RHGB5K–Sodium molybdate is an inorganic compound. It is a source of molybdenum, foliar fertilizer applied both in seed treatment and foliar application
 LTANUAR.Y 27 1883. PHOTOGRAPHIC NOTES. IMPROVED DEVELOPERS. Water 10 ounces. The bromine solution is first mixed with the water and then Pyrogallic acid 120 grains is added. The solution is now ready for use. Care should be taken to avoid inhaling the fumes of bromine when mixing it with water. Water . 2 ounces. Bromide of potassium . .180 grains. This solution must be kept in a tightly corked bottle. Water 20 ounces. Stock solution No. 2 3 drachms. To develop a quarter plate take seven drachms of No. 3 to which add sixty minims of No. 1; flow this over the plate; and the image will develop Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/ltanuary-27-1883-photographic-notes-improved-developers-water-10-ounces-the-bromine-solution-is-first-mixed-with-the-water-and-then-pyrogallic-acid-120-grains-is-added-the-solution-is-now-ready-for-use-care-should-be-taken-to-avoid-inhaling-the-fumes-of-bromine-when-mixing-it-with-water-water-2-ounces-bromide-of-potassium-180-grains-this-solution-must-be-kept-in-a-tightly-corked-bottle-water-20-ounces-stock-solution-no-2-3-drachms-to-develop-a-quarter-plate-take-seven-drachms-of-no-3-to-which-add-sixty-minims-of-no-1-flow-this-over-the-plate-and-the-image-will-develop-image334326197.html
LTANUAR.Y 27 1883. PHOTOGRAPHIC NOTES. IMPROVED DEVELOPERS. Water 10 ounces. The bromine solution is first mixed with the water and then Pyrogallic acid 120 grains is added. The solution is now ready for use. Care should be taken to avoid inhaling the fumes of bromine when mixing it with water. Water . 2 ounces. Bromide of potassium . .180 grains. This solution must be kept in a tightly corked bottle. Water 20 ounces. Stock solution No. 2 3 drachms. To develop a quarter plate take seven drachms of No. 3 to which add sixty minims of No. 1; flow this over the plate; and the image will develop Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/ltanuary-27-1883-photographic-notes-improved-developers-water-10-ounces-the-bromine-solution-is-first-mixed-with-the-water-and-then-pyrogallic-acid-120-grains-is-added-the-solution-is-now-ready-for-use-care-should-be-taken-to-avoid-inhaling-the-fumes-of-bromine-when-mixing-it-with-water-water-2-ounces-bromide-of-potassium-180-grains-this-solution-must-be-kept-in-a-tightly-corked-bottle-water-20-ounces-stock-solution-no-2-3-drachms-to-develop-a-quarter-plate-take-seven-drachms-of-no-3-to-which-add-sixty-minims-of-no-1-flow-this-over-the-plate-and-the-image-will-develop-image334326197.htmlRM2ABWTD9–LTANUAR.Y 27 1883. PHOTOGRAPHIC NOTES. IMPROVED DEVELOPERS. Water 10 ounces. The bromine solution is first mixed with the water and then Pyrogallic acid 120 grains is added. The solution is now ready for use. Care should be taken to avoid inhaling the fumes of bromine when mixing it with water. Water . 2 ounces. Bromide of potassium . .180 grains. This solution must be kept in a tightly corked bottle. Water 20 ounces. Stock solution No. 2 3 drachms. To develop a quarter plate take seven drachms of No. 3 to which add sixty minims of No. 1; flow this over the plate; and the image will develop
 Academy Secondary school science equipment Chemicals on a shelf Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/academy-secondary-school-science-equipment-chemicals-on-a-shelf-image220391333.html
Academy Secondary school science equipment Chemicals on a shelf Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/academy-secondary-school-science-equipment-chemicals-on-a-shelf-image220391333.htmlRFPPFKB1–Academy Secondary school science equipment Chemicals on a shelf
![American journal of pharmacy . Successors to J AS. R. NICHOLS & CO. MAJ^UFACTURIJVG CHEMISTS, • AND Standard Pharmacsutical Prsparations. BOSTON, MASS., «fe 20 I^IBERTY STREET,;]V. Y. We solicit correspondence upon the following specialties: AMMONIUM BROMIDE, CARBOLIC ACID, POTASSIUM BROMIDE,BROMINE, POTASSIUM ACETATE, CALCIUM SULPHITE (suiphite Lime),PROPYLAMINE, PROPYLAMINE CHLORIDE, SODIUM SULPHITE,SODIUM ACETATE, SCALE IRONS, ETC., ETC. MANUFACTURERS OF Elixir Peruvian Bark with Protoxide of Iron. AN OLD AND VALUABLE AGENT. Since 1856 we have supplied to Physicians this preparation, and it Stock Photo American journal of pharmacy . Successors to J AS. R. NICHOLS & CO. MAJ^UFACTURIJVG CHEMISTS, • AND Standard Pharmacsutical Prsparations. BOSTON, MASS., «fe 20 I^IBERTY STREET,;]V. Y. We solicit correspondence upon the following specialties: AMMONIUM BROMIDE, CARBOLIC ACID, POTASSIUM BROMIDE,BROMINE, POTASSIUM ACETATE, CALCIUM SULPHITE (suiphite Lime),PROPYLAMINE, PROPYLAMINE CHLORIDE, SODIUM SULPHITE,SODIUM ACETATE, SCALE IRONS, ETC., ETC. MANUFACTURERS OF Elixir Peruvian Bark with Protoxide of Iron. AN OLD AND VALUABLE AGENT. Since 1856 we have supplied to Physicians this preparation, and it Stock Photo](https://c8.alamy.com/comp/2AJ7708/american-journal-of-pharmacy-successors-to-j-as-r-nichols-co-majufacturijvg-chemists-and-standard-pharmacsutical-prsparations-boston-mass-fe-20-iiberty-street-v-y-we-solicit-correspondence-upon-the-following-specialties-ammonium-bromide-carbolic-acid-potassium-bromidebromine-potassium-acetate-calcium-sulphite-suiphite-limepropylamine-propylamine-chloride-sodium-sulphitesodium-acetate-scale-irons-etc-etc-manufacturers-of-elixir-peruvian-bark-with-protoxide-of-iron-an-old-and-valuable-agent-since-1856-we-have-supplied-to-physicians-this-preparation-and-it-2AJ7708.jpg) American journal of pharmacy . Successors to J AS. R. NICHOLS & CO. MAJ^UFACTURIJVG CHEMISTS, • AND Standard Pharmacsutical Prsparations. BOSTON, MASS., «fe 20 I^IBERTY STREET,;]V. Y. We solicit correspondence upon the following specialties: AMMONIUM BROMIDE, CARBOLIC ACID, POTASSIUM BROMIDE,BROMINE, POTASSIUM ACETATE, CALCIUM SULPHITE (suiphite Lime),PROPYLAMINE, PROPYLAMINE CHLORIDE, SODIUM SULPHITE,SODIUM ACETATE, SCALE IRONS, ETC., ETC. MANUFACTURERS OF Elixir Peruvian Bark with Protoxide of Iron. AN OLD AND VALUABLE AGENT. Since 1856 we have supplied to Physicians this preparation, and it Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/american-journal-of-pharmacy-successors-to-j-as-r-nichols-co-majufacturijvg-chemists-and-standard-pharmacsutical-prsparations-boston-mass-fe-20-iiberty-street-v-y-we-solicit-correspondence-upon-the-following-specialties-ammonium-bromide-carbolic-acid-potassium-bromidebromine-potassium-acetate-calcium-sulphite-suiphite-limepropylamine-propylamine-chloride-sodium-sulphitesodium-acetate-scale-irons-etc-etc-manufacturers-of-elixir-peruvian-bark-with-protoxide-of-iron-an-old-and-valuable-agent-since-1856-we-have-supplied-to-physicians-this-preparation-and-it-image338219960.html
American journal of pharmacy . Successors to J AS. R. NICHOLS & CO. MAJ^UFACTURIJVG CHEMISTS, • AND Standard Pharmacsutical Prsparations. BOSTON, MASS., «fe 20 I^IBERTY STREET,;]V. Y. We solicit correspondence upon the following specialties: AMMONIUM BROMIDE, CARBOLIC ACID, POTASSIUM BROMIDE,BROMINE, POTASSIUM ACETATE, CALCIUM SULPHITE (suiphite Lime),PROPYLAMINE, PROPYLAMINE CHLORIDE, SODIUM SULPHITE,SODIUM ACETATE, SCALE IRONS, ETC., ETC. MANUFACTURERS OF Elixir Peruvian Bark with Protoxide of Iron. AN OLD AND VALUABLE AGENT. Since 1856 we have supplied to Physicians this preparation, and it Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/american-journal-of-pharmacy-successors-to-j-as-r-nichols-co-majufacturijvg-chemists-and-standard-pharmacsutical-prsparations-boston-mass-fe-20-iiberty-street-v-y-we-solicit-correspondence-upon-the-following-specialties-ammonium-bromide-carbolic-acid-potassium-bromidebromine-potassium-acetate-calcium-sulphite-suiphite-limepropylamine-propylamine-chloride-sodium-sulphitesodium-acetate-scale-irons-etc-etc-manufacturers-of-elixir-peruvian-bark-with-protoxide-of-iron-an-old-and-valuable-agent-since-1856-we-have-supplied-to-physicians-this-preparation-and-it-image338219960.htmlRM2AJ7708–American journal of pharmacy . Successors to J AS. R. NICHOLS & CO. MAJ^UFACTURIJVG CHEMISTS, • AND Standard Pharmacsutical Prsparations. BOSTON, MASS., «fe 20 I^IBERTY STREET,;]V. Y. We solicit correspondence upon the following specialties: AMMONIUM BROMIDE, CARBOLIC ACID, POTASSIUM BROMIDE,BROMINE, POTASSIUM ACETATE, CALCIUM SULPHITE (suiphite Lime),PROPYLAMINE, PROPYLAMINE CHLORIDE, SODIUM SULPHITE,SODIUM ACETATE, SCALE IRONS, ETC., ETC. MANUFACTURERS OF Elixir Peruvian Bark with Protoxide of Iron. AN OLD AND VALUABLE AGENT. Since 1856 we have supplied to Physicians this preparation, and it
 Sodium molybdate is an inorganic compound. It is a source of molybdenum, foliar fertilizer applied both in seed treatment and foliar application Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-molybdate-is-an-inorganic-compound-it-is-a-source-of-molybdenum-foliar-fertilizer-applied-both-in-seed-treatment-and-foliar-application-image561540954.html
Sodium molybdate is an inorganic compound. It is a source of molybdenum, foliar fertilizer applied both in seed treatment and foliar application Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-molybdate-is-an-inorganic-compound-it-is-a-source-of-molybdenum-foliar-fertilizer-applied-both-in-seed-treatment-and-foliar-application-image561540954.htmlRF2RHGB62–Sodium molybdate is an inorganic compound. It is a source of molybdenum, foliar fertilizer applied both in seed treatment and foliar application
 American journal of pharmacy . Snccmom to J AS. R. XICHOLS A CO. MAJVUFACTUErJVG CHEMISTS, AND Standard Pharmacsutical Prsparations. BOSTON, MASS., & 20 IBERTY STREET, N. Y. We solicit correspondence upon the following specialties: AMMONIUM BROMIDE, CARBOLIC ACID, POTASSIUM BROMIDE,.BROMINE, POTASSIUM ACETATE, CALCIUM SULPHITE (suiphite li™,),PROPYLiMINE, PROPYLAMINE CHLORIDE, S0DIU3I SULPHITE,SODIUM ACETATE, SCALE IRONS, ETC., ETC. MANUFACTURERS OF Elixir Peruvian Bark with Protoxide of Iron, AN OLD AND VALUABLE AGENT. Since 1850 we have supplied to Physicians this preparation, and it lias Le Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/american-journal-of-pharmacy-snccmom-to-j-as-r-xichols-a-co-majvufactuerjvg-chemists-and-standard-pharmacsutical-prsparations-boston-mass-20-iberty-street-n-y-we-solicit-correspondence-upon-the-following-specialties-ammonium-bromide-carbolic-acid-potassium-bromidebromine-potassium-acetate-calcium-sulphite-suiphite-lipropylimine-propylamine-chloride-s0diu3i-sulphitesodium-acetate-scale-irons-etc-etc-manufacturers-of-elixir-peruvian-bark-with-protoxide-of-iron-an-old-and-valuable-agent-since-1850-we-have-supplied-to-physicians-this-preparation-and-it-lias-le-image338227079.html
American journal of pharmacy . Snccmom to J AS. R. XICHOLS A CO. MAJVUFACTUErJVG CHEMISTS, AND Standard Pharmacsutical Prsparations. BOSTON, MASS., & 20 IBERTY STREET, N. Y. We solicit correspondence upon the following specialties: AMMONIUM BROMIDE, CARBOLIC ACID, POTASSIUM BROMIDE,.BROMINE, POTASSIUM ACETATE, CALCIUM SULPHITE (suiphite li™,),PROPYLiMINE, PROPYLAMINE CHLORIDE, S0DIU3I SULPHITE,SODIUM ACETATE, SCALE IRONS, ETC., ETC. MANUFACTURERS OF Elixir Peruvian Bark with Protoxide of Iron, AN OLD AND VALUABLE AGENT. Since 1850 we have supplied to Physicians this preparation, and it lias Le Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/american-journal-of-pharmacy-snccmom-to-j-as-r-xichols-a-co-majvufactuerjvg-chemists-and-standard-pharmacsutical-prsparations-boston-mass-20-iberty-street-n-y-we-solicit-correspondence-upon-the-following-specialties-ammonium-bromide-carbolic-acid-potassium-bromidebromine-potassium-acetate-calcium-sulphite-suiphite-lipropylimine-propylamine-chloride-s0diu3i-sulphitesodium-acetate-scale-irons-etc-etc-manufacturers-of-elixir-peruvian-bark-with-protoxide-of-iron-an-old-and-valuable-agent-since-1850-we-have-supplied-to-physicians-this-preparation-and-it-lias-le-image338227079.htmlRM2AJ7G2F–American journal of pharmacy . Snccmom to J AS. R. XICHOLS A CO. MAJVUFACTUErJVG CHEMISTS, AND Standard Pharmacsutical Prsparations. BOSTON, MASS., & 20 IBERTY STREET, N. Y. We solicit correspondence upon the following specialties: AMMONIUM BROMIDE, CARBOLIC ACID, POTASSIUM BROMIDE,.BROMINE, POTASSIUM ACETATE, CALCIUM SULPHITE (suiphite li™,),PROPYLiMINE, PROPYLAMINE CHLORIDE, S0DIU3I SULPHITE,SODIUM ACETATE, SCALE IRONS, ETC., ETC. MANUFACTURERS OF Elixir Peruvian Bark with Protoxide of Iron, AN OLD AND VALUABLE AGENT. Since 1850 we have supplied to Physicians this preparation, and it lias Le
 Sodium molybdate is an inorganic compound. It is a source of molybdenum, foliar fertilizer applied both in seed treatment and foliar application Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-molybdate-is-an-inorganic-compound-it-is-a-source-of-molybdenum-foliar-fertilizer-applied-both-in-seed-treatment-and-foliar-application-image561540948.html
Sodium molybdate is an inorganic compound. It is a source of molybdenum, foliar fertilizer applied both in seed treatment and foliar application Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-molybdate-is-an-inorganic-compound-it-is-a-source-of-molybdenum-foliar-fertilizer-applied-both-in-seed-treatment-and-foliar-application-image561540948.htmlRF2RHGB5T–Sodium molybdate is an inorganic compound. It is a source of molybdenum, foliar fertilizer applied both in seed treatment and foliar application
 American journal of pharmacy . , Suecesson to J AS. R: NICHOLS & CO. MAJVUFJCTURIJVG CHEMISTS, AND Standard Pharmacsutlcal Preparations. BOSTON, MASS., & 20 LIBERTY STREET, N. Y. We solicit correspondence upon the following specialties: AMMONIUM BROMIDE, CARBOLIC ACID, POTASSIUM BROMIDE,BROMINE, POTASSIUM ACETATE, CALCIUM SULPHITE (Suiphite nme),PROPYL AMINE, PROPYLAMINE CHLORIDE, SODIUM SULPHITE,SODIUM ACETATE, SCALE IRONS, ETC., ETC. MANUFACTURERS OF Elixir Peruvian Bark with Protoxide of Iron. AN OLD AND VALUABLE AGENT. Since 1856 we have supplied to Physicians this preparation, and it has Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/american-journal-of-pharmacy-suecesson-to-j-as-r-nichols-co-majvufjcturijvg-chemists-and-standard-pharmacsutlcal-preparations-boston-mass-20-liberty-street-n-y-we-solicit-correspondence-upon-the-following-specialties-ammonium-bromide-carbolic-acid-potassium-bromidebromine-potassium-acetate-calcium-sulphite-suiphite-nmepropyl-amine-propylamine-chloride-sodium-sulphitesodium-acetate-scale-irons-etc-etc-manufacturers-of-elixir-peruvian-bark-with-protoxide-of-iron-an-old-and-valuable-agent-since-1856-we-have-supplied-to-physicians-this-preparation-and-it-has-image338217261.html
American journal of pharmacy . , Suecesson to J AS. R: NICHOLS & CO. MAJVUFJCTURIJVG CHEMISTS, AND Standard Pharmacsutlcal Preparations. BOSTON, MASS., & 20 LIBERTY STREET, N. Y. We solicit correspondence upon the following specialties: AMMONIUM BROMIDE, CARBOLIC ACID, POTASSIUM BROMIDE,BROMINE, POTASSIUM ACETATE, CALCIUM SULPHITE (Suiphite nme),PROPYL AMINE, PROPYLAMINE CHLORIDE, SODIUM SULPHITE,SODIUM ACETATE, SCALE IRONS, ETC., ETC. MANUFACTURERS OF Elixir Peruvian Bark with Protoxide of Iron. AN OLD AND VALUABLE AGENT. Since 1856 we have supplied to Physicians this preparation, and it has Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/american-journal-of-pharmacy-suecesson-to-j-as-r-nichols-co-majvufjcturijvg-chemists-and-standard-pharmacsutlcal-preparations-boston-mass-20-liberty-street-n-y-we-solicit-correspondence-upon-the-following-specialties-ammonium-bromide-carbolic-acid-potassium-bromidebromine-potassium-acetate-calcium-sulphite-suiphite-nmepropyl-amine-propylamine-chloride-sodium-sulphitesodium-acetate-scale-irons-etc-etc-manufacturers-of-elixir-peruvian-bark-with-protoxide-of-iron-an-old-and-valuable-agent-since-1856-we-have-supplied-to-physicians-this-preparation-and-it-has-image338217261.htmlRM2AJ73FW–American journal of pharmacy . , Suecesson to J AS. R: NICHOLS & CO. MAJVUFJCTURIJVG CHEMISTS, AND Standard Pharmacsutlcal Preparations. BOSTON, MASS., & 20 LIBERTY STREET, N. Y. We solicit correspondence upon the following specialties: AMMONIUM BROMIDE, CARBOLIC ACID, POTASSIUM BROMIDE,BROMINE, POTASSIUM ACETATE, CALCIUM SULPHITE (Suiphite nme),PROPYL AMINE, PROPYLAMINE CHLORIDE, SODIUM SULPHITE,SODIUM ACETATE, SCALE IRONS, ETC., ETC. MANUFACTURERS OF Elixir Peruvian Bark with Protoxide of Iron. AN OLD AND VALUABLE AGENT. Since 1856 we have supplied to Physicians this preparation, and it has
 Anthony's photographic bulletin for .. . njunction withclouds lit from the left hand. In all cases where a foreign sky has been d into requisition, there must be inaccuracy.One or other of the above-described methods should be adoptedthat i^. either two negatives should be made, one immediately after ther, or an orthochromatic plate and color-screen should be cm plow I i is a method by which one plate may be made to give a close approximation to the sky present in the original subject, and that is by the sky by th<- aid of potassium bromide. Idle plate is eloper, and, as soon as the skj mak Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/anthonys-photographic-bulletin-for-njunction-withclouds-lit-from-the-left-hand-in-all-cases-where-a-foreign-sky-has-been-d-into-requisition-there-must-be-inaccuracyone-or-other-of-the-above-described-methods-should-be-adoptedthat-i-either-two-negatives-should-be-made-one-immediately-after-ther-or-an-orthochromatic-plate-and-color-screen-should-be-cm-plow-i-i-is-a-method-by-which-one-plate-may-be-made-to-give-a-close-approximation-to-the-sky-present-in-the-original-subject-and-that-is-by-the-sky-by-thlt-aid-of-potassium-bromide-idle-plate-is-eloper-and-as-soon-as-the-skj-mak-image339403032.html
Anthony's photographic bulletin for .. . njunction withclouds lit from the left hand. In all cases where a foreign sky has been d into requisition, there must be inaccuracy.One or other of the above-described methods should be adoptedthat i^. either two negatives should be made, one immediately after ther, or an orthochromatic plate and color-screen should be cm plow I i is a method by which one plate may be made to give a close approximation to the sky present in the original subject, and that is by the sky by th<- aid of potassium bromide. Idle plate is eloper, and, as soon as the skj mak Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/anthonys-photographic-bulletin-for-njunction-withclouds-lit-from-the-left-hand-in-all-cases-where-a-foreign-sky-has-been-d-into-requisition-there-must-be-inaccuracyone-or-other-of-the-above-described-methods-should-be-adoptedthat-i-either-two-negatives-should-be-made-one-immediately-after-ther-or-an-orthochromatic-plate-and-color-screen-should-be-cm-plow-i-i-is-a-method-by-which-one-plate-may-be-made-to-give-a-close-approximation-to-the-sky-present-in-the-original-subject-and-that-is-by-the-sky-by-thlt-aid-of-potassium-bromide-idle-plate-is-eloper-and-as-soon-as-the-skj-mak-image339403032.htmlRM2AM540T–Anthony's photographic bulletin for .. . njunction withclouds lit from the left hand. In all cases where a foreign sky has been d into requisition, there must be inaccuracy.One or other of the above-described methods should be adoptedthat i^. either two negatives should be made, one immediately after ther, or an orthochromatic plate and color-screen should be cm plow I i is a method by which one plate may be made to give a close approximation to the sky present in the original subject, and that is by the sky by th<- aid of potassium bromide. Idle plate is eloper, and, as soon as the skj mak
 . The American annual of photography. IN THE GARRET. HERMAN GABRIEL. 195. COLOR-TONING BROMIDES By A. T. LAKIN, M.D. OLOR-TONING of Bromides is rather fasci-nating, and is simply an application of the ordi-nary Sulphide Toning. My standard bleacher: Potassium Ferricyanide 300 grains Potassium Bromide ; 100 grains Water 20 ounces Variations of this formula are common, the only point thatneed concern us, is that if the proportion of bromide is overone-third the amount of the Ferricyanide, the Sepias tendtowards the Yellows. In the portrait, that I am using as an illustration (Figurei), the first Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-american-annual-of-photography-in-the-garret-herman-gabriel-195-color-toning-bromides-by-a-t-lakin-md-olor-toning-of-bromides-is-rather-fasci-nating-and-is-simply-an-application-of-the-ordi-nary-sulphide-toning-my-standard-bleacher-potassium-ferricyanide-300-grains-potassium-bromide-100-grains-water-20-ounces-variations-of-this-formula-are-common-the-only-point-thatneed-concern-us-is-that-if-the-proportion-of-bromide-is-overone-third-the-amount-of-the-ferricyanide-the-sepias-tendtowards-the-yellows-in-the-portrait-that-i-am-using-as-an-illustration-figurei-the-first-image337088707.html
. The American annual of photography. IN THE GARRET. HERMAN GABRIEL. 195. COLOR-TONING BROMIDES By A. T. LAKIN, M.D. OLOR-TONING of Bromides is rather fasci-nating, and is simply an application of the ordi-nary Sulphide Toning. My standard bleacher: Potassium Ferricyanide 300 grains Potassium Bromide ; 100 grains Water 20 ounces Variations of this formula are common, the only point thatneed concern us, is that if the proportion of bromide is overone-third the amount of the Ferricyanide, the Sepias tendtowards the Yellows. In the portrait, that I am using as an illustration (Figurei), the first Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-american-annual-of-photography-in-the-garret-herman-gabriel-195-color-toning-bromides-by-a-t-lakin-md-olor-toning-of-bromides-is-rather-fasci-nating-and-is-simply-an-application-of-the-ordi-nary-sulphide-toning-my-standard-bleacher-potassium-ferricyanide-300-grains-potassium-bromide-100-grains-water-20-ounces-variations-of-this-formula-are-common-the-only-point-thatneed-concern-us-is-that-if-the-proportion-of-bromide-is-overone-third-the-amount-of-the-ferricyanide-the-sepias-tendtowards-the-yellows-in-the-portrait-that-i-am-using-as-an-illustration-figurei-the-first-image337088707.htmlRM2AGBM2B–. The American annual of photography. IN THE GARRET. HERMAN GABRIEL. 195. COLOR-TONING BROMIDES By A. T. LAKIN, M.D. OLOR-TONING of Bromides is rather fasci-nating, and is simply an application of the ordi-nary Sulphide Toning. My standard bleacher: Potassium Ferricyanide 300 grains Potassium Bromide ; 100 grains Water 20 ounces Variations of this formula are common, the only point thatneed concern us, is that if the proportion of bromide is overone-third the amount of the Ferricyanide, the Sepias tendtowards the Yellows. In the portrait, that I am using as an illustration (Figurei), the first
 The Kodak Salesman . atent image is impressed, andthrough suitable chemical develop-ment brought out as a negative. Tomake a sensitive emulsion a silvernitrate solution is mixed with a so-lution of potassium bromide andgelatine dissolved in hot water, thusforming insoluble silver bromide inthe solution, which is the compoundthat is sensitive to light. The warmsolution of gelatine containing thesilver bromide is coated on thenitro-cellulose backing already de-scribed. The gelatine solution withthe silver compound in it is calledan emulsion because of the way inwhich the silver bromide remainssu Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-kodak-salesman-atent-image-is-impressed-andthrough-suitable-chemical-develop-ment-brought-out-as-a-negative-tomake-a-sensitive-emulsion-a-silvernitrate-solution-is-mixed-with-a-so-lution-of-potassium-bromide-andgelatine-dissolved-in-hot-water-thusforming-insoluble-silver-bromide-inthe-solution-which-is-the-compoundthat-is-sensitive-to-light-the-warmsolution-of-gelatine-containing-thesilver-bromide-is-coated-on-thenitro-cellulose-backing-already-de-scribed-the-gelatine-solution-withthe-silver-compound-in-it-is-calledan-emulsion-because-of-the-way-inwhich-the-silver-bromide-remainssu-image338179637.html
The Kodak Salesman . atent image is impressed, andthrough suitable chemical develop-ment brought out as a negative. Tomake a sensitive emulsion a silvernitrate solution is mixed with a so-lution of potassium bromide andgelatine dissolved in hot water, thusforming insoluble silver bromide inthe solution, which is the compoundthat is sensitive to light. The warmsolution of gelatine containing thesilver bromide is coated on thenitro-cellulose backing already de-scribed. The gelatine solution withthe silver compound in it is calledan emulsion because of the way inwhich the silver bromide remainssu Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-kodak-salesman-atent-image-is-impressed-andthrough-suitable-chemical-develop-ment-brought-out-as-a-negative-tomake-a-sensitive-emulsion-a-silvernitrate-solution-is-mixed-with-a-so-lution-of-potassium-bromide-andgelatine-dissolved-in-hot-water-thusforming-insoluble-silver-bromide-inthe-solution-which-is-the-compoundthat-is-sensitive-to-light-the-warmsolution-of-gelatine-containing-thesilver-bromide-is-coated-on-thenitro-cellulose-backing-already-de-scribed-the-gelatine-solution-withthe-silver-compound-in-it-is-calledan-emulsion-because-of-the-way-inwhich-the-silver-bromide-remainssu-image338179637.htmlRM2AJ5BG5–The Kodak Salesman . atent image is impressed, andthrough suitable chemical develop-ment brought out as a negative. Tomake a sensitive emulsion a silvernitrate solution is mixed with a so-lution of potassium bromide andgelatine dissolved in hot water, thusforming insoluble silver bromide inthe solution, which is the compoundthat is sensitive to light. The warmsolution of gelatine containing thesilver bromide is coated on thenitro-cellulose backing already de-scribed. The gelatine solution withthe silver compound in it is calledan emulsion because of the way inwhich the silver bromide remainssu
 The American annual of photography . at some length to the bromoil process, a processwhich in the hands of the enthusiast is hard to equal, andalmost impossible to excel. But to many who have tried itdiscouragement and failure have been the portion. To suchI say try again, and perhaps a few suggestions in the matterof working details may be of assistance.Try the following developer: Water 20 oz. Sulphite of soda (Anhydrous) ^ oz. Amidol 50 grs. Potassium Bromide 10 grs. Bi-Sulphite of soda 50 grs. Use the developer immediately after it has been compoundedand use fresh developer for each print. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-american-annual-of-photography-at-some-length-to-the-bromoil-process-a-processwhich-in-the-hands-of-the-enthusiast-is-hard-to-equal-andalmost-impossible-to-excel-but-to-many-who-have-tried-itdiscouragement-and-failure-have-been-the-portion-to-suchi-say-try-again-and-perhaps-a-few-suggestions-in-the-matterof-working-details-may-be-of-assistancetry-the-following-developer-water-20-oz-sulphite-of-soda-anhydrous-oz-amidol-50-grs-potassium-bromide-10-grs-bi-sulphite-of-soda-50-grs-use-the-developer-immediately-after-it-has-been-compoundedand-use-fresh-developer-for-each-print-image340007236.html
The American annual of photography . at some length to the bromoil process, a processwhich in the hands of the enthusiast is hard to equal, andalmost impossible to excel. But to many who have tried itdiscouragement and failure have been the portion. To suchI say try again, and perhaps a few suggestions in the matterof working details may be of assistance.Try the following developer: Water 20 oz. Sulphite of soda (Anhydrous) ^ oz. Amidol 50 grs. Potassium Bromide 10 grs. Bi-Sulphite of soda 50 grs. Use the developer immediately after it has been compoundedand use fresh developer for each print. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-american-annual-of-photography-at-some-length-to-the-bromoil-process-a-processwhich-in-the-hands-of-the-enthusiast-is-hard-to-equal-andalmost-impossible-to-excel-but-to-many-who-have-tried-itdiscouragement-and-failure-have-been-the-portion-to-suchi-say-try-again-and-perhaps-a-few-suggestions-in-the-matterof-working-details-may-be-of-assistancetry-the-following-developer-water-20-oz-sulphite-of-soda-anhydrous-oz-amidol-50-grs-potassium-bromide-10-grs-bi-sulphite-of-soda-50-grs-use-the-developer-immediately-after-it-has-been-compoundedand-use-fresh-developer-for-each-print-image340007236.htmlRM2AN4JKG–The American annual of photography . at some length to the bromoil process, a processwhich in the hands of the enthusiast is hard to equal, andalmost impossible to excel. But to many who have tried itdiscouragement and failure have been the portion. To suchI say try again, and perhaps a few suggestions in the matterof working details may be of assistance.Try the following developer: Water 20 oz. Sulphite of soda (Anhydrous) ^ oz. Amidol 50 grs. Potassium Bromide 10 grs. Bi-Sulphite of soda 50 grs. Use the developer immediately after it has been compoundedand use fresh developer for each print.
![American journal of pharmacy . Successors to J AS. R. NICHOLS & CO. MAJ^UFACTURIJVG CHEMISTS, • AND Standard Pharmacsutical Prsparations. BOSTON, MASS., «fe 20 I^IBERTY STREET,;]V. Y. We solicit correspondence upon the following specialties: AMMONIUM BROMIDE, CARBOLIC ACID, POTASSIUM BROMIDE,BROMINE, POTASSIUM ACETATE, CALCIUM SULPHITE (suiphite Lime),PROPYLAMINE, PROPYLAMINE CHLORIDE, SODIUM SULPHITE,SODIUM ACETATE, SCALE IRONS, ETC., ETC. MANUFACTURERS OF Elixir Peruvian Bark with Protoxide of Iron. AN OLD AND VALUABLE AGENT. Since 1856 we have supplied to Physicians this preparation, and it Stock Photo American journal of pharmacy . Successors to J AS. R. NICHOLS & CO. MAJ^UFACTURIJVG CHEMISTS, • AND Standard Pharmacsutical Prsparations. BOSTON, MASS., «fe 20 I^IBERTY STREET,;]V. Y. We solicit correspondence upon the following specialties: AMMONIUM BROMIDE, CARBOLIC ACID, POTASSIUM BROMIDE,BROMINE, POTASSIUM ACETATE, CALCIUM SULPHITE (suiphite Lime),PROPYLAMINE, PROPYLAMINE CHLORIDE, SODIUM SULPHITE,SODIUM ACETATE, SCALE IRONS, ETC., ETC. MANUFACTURERS OF Elixir Peruvian Bark with Protoxide of Iron. AN OLD AND VALUABLE AGENT. Since 1856 we have supplied to Physicians this preparation, and it Stock Photo](https://c8.alamy.com/comp/2AJ777D/american-journal-of-pharmacy-successors-to-j-as-r-nichols-co-majufacturijvg-chemists-and-standard-pharmacsutical-prsparations-boston-mass-fe-20-iiberty-street-v-y-we-solicit-correspondence-upon-the-following-specialties-ammonium-bromide-carbolic-acid-potassium-bromidebromine-potassium-acetate-calcium-sulphite-suiphite-limepropylamine-propylamine-chloride-sodium-sulphitesodium-acetate-scale-irons-etc-etc-manufacturers-of-elixir-peruvian-bark-with-protoxide-of-iron-an-old-and-valuable-agent-since-1856-we-have-supplied-to-physicians-this-preparation-and-it-2AJ777D.jpg) American journal of pharmacy . Successors to J AS. R. NICHOLS & CO. MAJ^UFACTURIJVG CHEMISTS, • AND Standard Pharmacsutical Prsparations. BOSTON, MASS., «fe 20 I^IBERTY STREET,;]V. Y. We solicit correspondence upon the following specialties: AMMONIUM BROMIDE, CARBOLIC ACID, POTASSIUM BROMIDE,BROMINE, POTASSIUM ACETATE, CALCIUM SULPHITE (suiphite Lime),PROPYLAMINE, PROPYLAMINE CHLORIDE, SODIUM SULPHITE,SODIUM ACETATE, SCALE IRONS, ETC., ETC. MANUFACTURERS OF Elixir Peruvian Bark with Protoxide of Iron. AN OLD AND VALUABLE AGENT. Since 1856 we have supplied to Physicians this preparation, and it Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/american-journal-of-pharmacy-successors-to-j-as-r-nichols-co-majufacturijvg-chemists-and-standard-pharmacsutical-prsparations-boston-mass-fe-20-iiberty-street-v-y-we-solicit-correspondence-upon-the-following-specialties-ammonium-bromide-carbolic-acid-potassium-bromidebromine-potassium-acetate-calcium-sulphite-suiphite-limepropylamine-propylamine-chloride-sodium-sulphitesodium-acetate-scale-irons-etc-etc-manufacturers-of-elixir-peruvian-bark-with-protoxide-of-iron-an-old-and-valuable-agent-since-1856-we-have-supplied-to-physicians-this-preparation-and-it-image338220161.html
American journal of pharmacy . Successors to J AS. R. NICHOLS & CO. MAJ^UFACTURIJVG CHEMISTS, • AND Standard Pharmacsutical Prsparations. BOSTON, MASS., «fe 20 I^IBERTY STREET,;]V. Y. We solicit correspondence upon the following specialties: AMMONIUM BROMIDE, CARBOLIC ACID, POTASSIUM BROMIDE,BROMINE, POTASSIUM ACETATE, CALCIUM SULPHITE (suiphite Lime),PROPYLAMINE, PROPYLAMINE CHLORIDE, SODIUM SULPHITE,SODIUM ACETATE, SCALE IRONS, ETC., ETC. MANUFACTURERS OF Elixir Peruvian Bark with Protoxide of Iron. AN OLD AND VALUABLE AGENT. Since 1856 we have supplied to Physicians this preparation, and it Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/american-journal-of-pharmacy-successors-to-j-as-r-nichols-co-majufacturijvg-chemists-and-standard-pharmacsutical-prsparations-boston-mass-fe-20-iiberty-street-v-y-we-solicit-correspondence-upon-the-following-specialties-ammonium-bromide-carbolic-acid-potassium-bromidebromine-potassium-acetate-calcium-sulphite-suiphite-limepropylamine-propylamine-chloride-sodium-sulphitesodium-acetate-scale-irons-etc-etc-manufacturers-of-elixir-peruvian-bark-with-protoxide-of-iron-an-old-and-valuable-agent-since-1856-we-have-supplied-to-physicians-this-preparation-and-it-image338220161.htmlRM2AJ777D–American journal of pharmacy . Successors to J AS. R. NICHOLS & CO. MAJ^UFACTURIJVG CHEMISTS, • AND Standard Pharmacsutical Prsparations. BOSTON, MASS., «fe 20 I^IBERTY STREET,;]V. Y. We solicit correspondence upon the following specialties: AMMONIUM BROMIDE, CARBOLIC ACID, POTASSIUM BROMIDE,BROMINE, POTASSIUM ACETATE, CALCIUM SULPHITE (suiphite Lime),PROPYLAMINE, PROPYLAMINE CHLORIDE, SODIUM SULPHITE,SODIUM ACETATE, SCALE IRONS, ETC., ETC. MANUFACTURERS OF Elixir Peruvian Bark with Protoxide of Iron. AN OLD AND VALUABLE AGENT. Since 1856 we have supplied to Physicians this preparation, and it
 American journal of pharmacy . BILT^TTVOH, CL^X^F Ac CO. Successors to J AS. R. NICHOLS & CO. MAJiUFACTURIJVG CHEMISTS, AND Standard FharmacGutical Preparations* BOSTON, MA8S., & 20 LIBERTY STREET, N. Y. We solicit correspondence upon the following specialties: AMMONIUM BROMIDE, CARBOLIC ACID, POTASSIUM BROMIDE,BROMINE, POTASSIUM ACETATE, CALCIUM SULPHITE (smphite Lime),PROPYLMINE, PROPYLAMINE CHLORIDE, SODIUM SULPHITE,SODIUM ACETATE, SCALE IRONS, ETC., ETC. MANUFACTURERS OF Elixir Peruvian Bark with Protoxide of Iron. AN OLD AND VALUABLE AGENT. Since 1856 we have supplied to Physicians this Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/american-journal-of-pharmacy-biltttvoh-clxf-ac-co-successors-to-j-as-r-nichols-co-majiufacturijvg-chemists-and-standard-fharmacgutical-preparations-boston-ma8s-20-liberty-street-n-y-we-solicit-correspondence-upon-the-following-specialties-ammonium-bromide-carbolic-acid-potassium-bromidebromine-potassium-acetate-calcium-sulphite-smphite-limepropylmine-propylamine-chloride-sodium-sulphitesodium-acetate-scale-irons-etc-etc-manufacturers-of-elixir-peruvian-bark-with-protoxide-of-iron-an-old-and-valuable-agent-since-1856-we-have-supplied-to-physicians-this-image338215387.html
American journal of pharmacy . BILT^TTVOH, CL^X^F Ac CO. Successors to J AS. R. NICHOLS & CO. MAJiUFACTURIJVG CHEMISTS, AND Standard FharmacGutical Preparations* BOSTON, MA8S., & 20 LIBERTY STREET, N. Y. We solicit correspondence upon the following specialties: AMMONIUM BROMIDE, CARBOLIC ACID, POTASSIUM BROMIDE,BROMINE, POTASSIUM ACETATE, CALCIUM SULPHITE (smphite Lime),PROPYLMINE, PROPYLAMINE CHLORIDE, SODIUM SULPHITE,SODIUM ACETATE, SCALE IRONS, ETC., ETC. MANUFACTURERS OF Elixir Peruvian Bark with Protoxide of Iron. AN OLD AND VALUABLE AGENT. Since 1856 we have supplied to Physicians this Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/american-journal-of-pharmacy-biltttvoh-clxf-ac-co-successors-to-j-as-r-nichols-co-majiufacturijvg-chemists-and-standard-fharmacgutical-preparations-boston-ma8s-20-liberty-street-n-y-we-solicit-correspondence-upon-the-following-specialties-ammonium-bromide-carbolic-acid-potassium-bromidebromine-potassium-acetate-calcium-sulphite-smphite-limepropylmine-propylamine-chloride-sodium-sulphitesodium-acetate-scale-irons-etc-etc-manufacturers-of-elixir-peruvian-bark-with-protoxide-of-iron-an-old-and-valuable-agent-since-1856-we-have-supplied-to-physicians-this-image338215387.htmlRM2AJ714Y–American journal of pharmacy . BILT^TTVOH, CL^X^F Ac CO. Successors to J AS. R. NICHOLS & CO. MAJiUFACTURIJVG CHEMISTS, AND Standard FharmacGutical Preparations* BOSTON, MA8S., & 20 LIBERTY STREET, N. Y. We solicit correspondence upon the following specialties: AMMONIUM BROMIDE, CARBOLIC ACID, POTASSIUM BROMIDE,BROMINE, POTASSIUM ACETATE, CALCIUM SULPHITE (smphite Lime),PROPYLMINE, PROPYLAMINE CHLORIDE, SODIUM SULPHITE,SODIUM ACETATE, SCALE IRONS, ETC., ETC. MANUFACTURERS OF Elixir Peruvian Bark with Protoxide of Iron. AN OLD AND VALUABLE AGENT. Since 1856 we have supplied to Physicians this
 The American annual of photography . .7 18 2 Sol. Sol. Potassium, Bicar-bonate Potassium, Bichro-mate Potassium, Bromide. Potassium, Carbon-ate Potassium, Chloro-platinite Potassium, Cyanide. Potassium, Ferricy-anide Potassium, Ferrocy-anide Potassium, Iodide... Potassium, Metabi-sulphite Potassium, Oxalate.. Potassium, Perman-ganate Potassium, Persul-phate Potassium, Sulpho-cyanide Pyrocatechin Rochelle Salt Silver, Nitrate Sodium, Acetate.... Sodium, Bicarbonate Sodium, Bisulphite.. Sodium, Bromide. . Sodium, Carbonate(dry) Sodium, Carbonate(cryst) Sodium, Chloride. . Sodium, Citrate.... Sod Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-american-annual-of-photography-7-18-2-sol-sol-potassium-bicar-bonate-potassium-bichro-mate-potassium-bromide-potassium-carbon-ate-potassium-chloro-platinite-potassium-cyanide-potassium-ferricy-anide-potassium-ferrocy-anide-potassium-iodide-potassium-metabi-sulphite-potassium-oxalate-potassium-perman-ganate-potassium-persul-phate-potassium-sulpho-cyanide-pyrocatechin-rochelle-salt-silver-nitrate-sodium-acetate-sodium-bicarbonate-sodium-bisulphite-sodium-bromide-sodium-carbonatedry-sodium-carbonatecryst-sodium-chloride-sodium-citrate-sod-image340054358.html
The American annual of photography . .7 18 2 Sol. Sol. Potassium, Bicar-bonate Potassium, Bichro-mate Potassium, Bromide. Potassium, Carbon-ate Potassium, Chloro-platinite Potassium, Cyanide. Potassium, Ferricy-anide Potassium, Ferrocy-anide Potassium, Iodide... Potassium, Metabi-sulphite Potassium, Oxalate.. Potassium, Perman-ganate Potassium, Persul-phate Potassium, Sulpho-cyanide Pyrocatechin Rochelle Salt Silver, Nitrate Sodium, Acetate.... Sodium, Bicarbonate Sodium, Bisulphite.. Sodium, Bromide. . Sodium, Carbonate(dry) Sodium, Carbonate(cryst) Sodium, Chloride. . Sodium, Citrate.... Sod Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-american-annual-of-photography-7-18-2-sol-sol-potassium-bicar-bonate-potassium-bichro-mate-potassium-bromide-potassium-carbon-ate-potassium-chloro-platinite-potassium-cyanide-potassium-ferricy-anide-potassium-ferrocy-anide-potassium-iodide-potassium-metabi-sulphite-potassium-oxalate-potassium-perman-ganate-potassium-persul-phate-potassium-sulpho-cyanide-pyrocatechin-rochelle-salt-silver-nitrate-sodium-acetate-sodium-bicarbonate-sodium-bisulphite-sodium-bromide-sodium-carbonatedry-sodium-carbonatecryst-sodium-chloride-sodium-citrate-sod-image340054358.htmlRM2AN6PPE–The American annual of photography . .7 18 2 Sol. Sol. Potassium, Bicar-bonate Potassium, Bichro-mate Potassium, Bromide. Potassium, Carbon-ate Potassium, Chloro-platinite Potassium, Cyanide. Potassium, Ferricy-anide Potassium, Ferrocy-anide Potassium, Iodide... Potassium, Metabi-sulphite Potassium, Oxalate.. Potassium, Perman-ganate Potassium, Persul-phate Potassium, Sulpho-cyanide Pyrocatechin Rochelle Salt Silver, Nitrate Sodium, Acetate.... Sodium, Bicarbonate Sodium, Bisulphite.. Sodium, Bromide. . Sodium, Carbonate(dry) Sodium, Carbonate(cryst) Sodium, Chloride. . Sodium, Citrate.... Sod
 The American annual of photography . Sol. Sol. Potassium, B i c a r -bonate Potassium, Bichro-mate Potassium, Bromide. Potassium, Carbon-ate Potassium, Chloro-platinite Potassium, Cyanide. Potassium, Ferricy-anide Potassium, Ferrocy-anide Potassium, Iodide... Potassium, Metabi-sulphite Potassium, Oxalate.. Potassium, Perman-ganate Potassium, Persul-phate Potassium, Sulpho-cyanide Pyrocatechin Rochelle Salt Silver, Nitrate Sodium, Acetate.... Sodium, Bicarbonate Sodium, Bisulphite.. Sodium, Bromide. . Sodium, Carbonate(dry) Sodium, Carbonate(cryst) Sodium, Chloride. . Sodium, Citrate.... Sodium Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-american-annual-of-photography-sol-sol-potassium-b-i-c-a-r-bonate-potassium-bichro-mate-potassium-bromide-potassium-carbon-ate-potassium-chloro-platinite-potassium-cyanide-potassium-ferricy-anide-potassium-ferrocy-anide-potassium-iodide-potassium-metabi-sulphite-potassium-oxalate-potassium-perman-ganate-potassium-persul-phate-potassium-sulpho-cyanide-pyrocatechin-rochelle-salt-silver-nitrate-sodium-acetate-sodium-bicarbonate-sodium-bisulphite-sodium-bromide-sodium-carbonatedry-sodium-carbonatecryst-sodium-chloride-sodium-citrate-sodium-image339090902.html
The American annual of photography . Sol. Sol. Potassium, B i c a r -bonate Potassium, Bichro-mate Potassium, Bromide. Potassium, Carbon-ate Potassium, Chloro-platinite Potassium, Cyanide. Potassium, Ferricy-anide Potassium, Ferrocy-anide Potassium, Iodide... Potassium, Metabi-sulphite Potassium, Oxalate.. Potassium, Perman-ganate Potassium, Persul-phate Potassium, Sulpho-cyanide Pyrocatechin Rochelle Salt Silver, Nitrate Sodium, Acetate.... Sodium, Bicarbonate Sodium, Bisulphite.. Sodium, Bromide. . Sodium, Carbonate(dry) Sodium, Carbonate(cryst) Sodium, Chloride. . Sodium, Citrate.... Sodium Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-american-annual-of-photography-sol-sol-potassium-b-i-c-a-r-bonate-potassium-bichro-mate-potassium-bromide-potassium-carbon-ate-potassium-chloro-platinite-potassium-cyanide-potassium-ferricy-anide-potassium-ferrocy-anide-potassium-iodide-potassium-metabi-sulphite-potassium-oxalate-potassium-perman-ganate-potassium-persul-phate-potassium-sulpho-cyanide-pyrocatechin-rochelle-salt-silver-nitrate-sodium-acetate-sodium-bicarbonate-sodium-bisulphite-sodium-bromide-sodium-carbonatedry-sodium-carbonatecryst-sodium-chloride-sodium-citrate-sodium-image339090902.htmlRM2AKJWWA–The American annual of photography . Sol. Sol. Potassium, B i c a r -bonate Potassium, Bichro-mate Potassium, Bromide. Potassium, Carbon-ate Potassium, Chloro-platinite Potassium, Cyanide. Potassium, Ferricy-anide Potassium, Ferrocy-anide Potassium, Iodide... Potassium, Metabi-sulphite Potassium, Oxalate.. Potassium, Perman-ganate Potassium, Persul-phate Potassium, Sulpho-cyanide Pyrocatechin Rochelle Salt Silver, Nitrate Sodium, Acetate.... Sodium, Bicarbonate Sodium, Bisulphite.. Sodium, Bromide. . Sodium, Carbonate(dry) Sodium, Carbonate(cryst) Sodium, Chloride. . Sodium, Citrate.... Sodium
 The American annual of photography . its ownfor artistic merit against collodion wet or dry. The emulsion work for gelatino-bromide plates is simplicityitself. Last winter when preparing the lecture referred toabove I found myself without lantern plates. The war was on,and the prices of plates were designed by a ring to establisha fortune unto the third or fourth generation. Under thesecircumstances I decided upon making a batch as I had thenecessary chemicals by me. Here is the formula, which willcoat one gross of lantern plates: A. Nelsons No. i Gelatine 40 grains Potassium bromide 100 grain Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-american-annual-of-photography-its-ownfor-artistic-merit-against-collodion-wet-or-dry-the-emulsion-work-for-gelatino-bromide-plates-is-simplicityitself-last-winter-when-preparing-the-lecture-referred-toabove-i-found-myself-without-lantern-plates-the-war-was-onand-the-prices-of-plates-were-designed-by-a-ring-to-establisha-fortune-unto-the-third-or-fourth-generation-under-thesecircumstances-i-decided-upon-making-a-batch-as-i-had-thenecessary-chemicals-by-me-here-is-the-formula-which-willcoat-one-gross-of-lantern-plates-a-nelsons-no-i-gelatine-40-grains-potassium-bromide-100-grain-image339029648.html
The American annual of photography . its ownfor artistic merit against collodion wet or dry. The emulsion work for gelatino-bromide plates is simplicityitself. Last winter when preparing the lecture referred toabove I found myself without lantern plates. The war was on,and the prices of plates were designed by a ring to establisha fortune unto the third or fourth generation. Under thesecircumstances I decided upon making a batch as I had thenecessary chemicals by me. Here is the formula, which willcoat one gross of lantern plates: A. Nelsons No. i Gelatine 40 grains Potassium bromide 100 grain Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-american-annual-of-photography-its-ownfor-artistic-merit-against-collodion-wet-or-dry-the-emulsion-work-for-gelatino-bromide-plates-is-simplicityitself-last-winter-when-preparing-the-lecture-referred-toabove-i-found-myself-without-lantern-plates-the-war-was-onand-the-prices-of-plates-were-designed-by-a-ring-to-establisha-fortune-unto-the-third-or-fourth-generation-under-thesecircumstances-i-decided-upon-making-a-batch-as-i-had-thenecessary-chemicals-by-me-here-is-the-formula-which-willcoat-one-gross-of-lantern-plates-a-nelsons-no-i-gelatine-40-grains-potassium-bromide-100-grain-image339029648.htmlRM2AKG3NM–The American annual of photography . its ownfor artistic merit against collodion wet or dry. The emulsion work for gelatino-bromide plates is simplicityitself. Last winter when preparing the lecture referred toabove I found myself without lantern plates. The war was on,and the prices of plates were designed by a ring to establisha fortune unto the third or fourth generation. Under thesecircumstances I decided upon making a batch as I had thenecessary chemicals by me. Here is the formula, which willcoat one gross of lantern plates: A. Nelsons No. i Gelatine 40 grains Potassium bromide 100 grain
 Chemical lecture experiments . atisfactoryfor the preparation of gaseous hydrobromic acid. Ten grams of potassium bromide are placed in a 300 cc.Erlenmeyer flask fitted with a dropping-funnel and a glasselbow (Fig. 54). A mixture is madeof 3 volumes of concentrated sul-phuric acid and 1 volume of waterby pouring the acid gradually intothe water and cooling the mixture.Sufficient of this mixture is addedto cover the potassium bromideand the contents of the flask arevery gently warmed. The gas issteadily evolved and may be col-lected in cylinders by displace-ment. The gas regulation is easily co Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/chemical-lecture-experiments-atisfactoryfor-the-preparation-of-gaseous-hydrobromic-acid-ten-grams-of-potassium-bromide-are-placed-in-a-300-ccerlenmeyer-flask-fitted-with-a-dropping-funnel-and-a-glasselbow-fig-54-a-mixture-is-madeof-3-volumes-of-concentrated-sul-phuric-acid-and-1-volume-of-waterby-pouring-the-acid-gradually-intothe-water-and-cooling-the-mixturesufficient-of-this-mixture-is-addedto-cover-the-potassium-bromideand-the-contents-of-the-flask-arevery-gently-warmed-the-gas-issteadily-evolved-and-may-be-col-lected-in-cylinders-by-displace-ment-the-gas-regulation-is-easily-co-image343376397.html
Chemical lecture experiments . atisfactoryfor the preparation of gaseous hydrobromic acid. Ten grams of potassium bromide are placed in a 300 cc.Erlenmeyer flask fitted with a dropping-funnel and a glasselbow (Fig. 54). A mixture is madeof 3 volumes of concentrated sul-phuric acid and 1 volume of waterby pouring the acid gradually intothe water and cooling the mixture.Sufficient of this mixture is addedto cover the potassium bromideand the contents of the flask arevery gently warmed. The gas issteadily evolved and may be col-lected in cylinders by displace-ment. The gas regulation is easily co Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/chemical-lecture-experiments-atisfactoryfor-the-preparation-of-gaseous-hydrobromic-acid-ten-grams-of-potassium-bromide-are-placed-in-a-300-ccerlenmeyer-flask-fitted-with-a-dropping-funnel-and-a-glasselbow-fig-54-a-mixture-is-madeof-3-volumes-of-concentrated-sul-phuric-acid-and-1-volume-of-waterby-pouring-the-acid-gradually-intothe-water-and-cooling-the-mixturesufficient-of-this-mixture-is-addedto-cover-the-potassium-bromideand-the-contents-of-the-flask-arevery-gently-warmed-the-gas-issteadily-evolved-and-may-be-col-lected-in-cylinders-by-displace-ment-the-gas-regulation-is-easily-co-image343376397.htmlRM2AXJ42N–Chemical lecture experiments . atisfactoryfor the preparation of gaseous hydrobromic acid. Ten grams of potassium bromide are placed in a 300 cc.Erlenmeyer flask fitted with a dropping-funnel and a glasselbow (Fig. 54). A mixture is madeof 3 volumes of concentrated sul-phuric acid and 1 volume of waterby pouring the acid gradually intothe water and cooling the mixture.Sufficient of this mixture is addedto cover the potassium bromideand the contents of the flask arevery gently warmed. The gas issteadily evolved and may be col-lected in cylinders by displace-ment. The gas regulation is easily co
 The American annual of photography . <a tm -A...d^l m-.^M;mmmm. veloper fog is caused by impurities or oxidation products ofthe developing agent formed during mixing. The importanceof careful mixing of developers for test purposes is thereforeapparent (See page i86 Mixing the Developer). Interesting facts about fog are that the fog layer is notdistributed evenly over the entire image, but is thinner in thehighlights because in those places where more image is de-veloped, more potassium bromide is formed as a reaction pro-duct of development which therefore restrains the fog adjacentto the d Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-american-annual-of-photography-lta-tm-adl-m-mmmmm-veloper-fog-is-caused-by-impurities-or-oxidation-products-ofthe-developing-agent-formed-during-mixing-the-importanceof-careful-mixing-of-developers-for-test-purposes-is-thereforeapparent-see-page-i86-mixing-the-developer-interesting-facts-about-fog-are-that-the-fog-layer-is-notdistributed-evenly-over-the-entire-image-but-is-thinner-in-thehighlights-because-in-those-places-where-more-image-is-de-veloped-more-potassium-bromide-is-formed-as-a-reaction-pro-duct-of-development-which-therefore-restrains-the-fog-adjacentto-the-d-image339977139.html
The American annual of photography . <a tm -A...d^l m-.^M;mmmm. veloper fog is caused by impurities or oxidation products ofthe developing agent formed during mixing. The importanceof careful mixing of developers for test purposes is thereforeapparent (See page i86 Mixing the Developer). Interesting facts about fog are that the fog layer is notdistributed evenly over the entire image, but is thinner in thehighlights because in those places where more image is de-veloped, more potassium bromide is formed as a reaction pro-duct of development which therefore restrains the fog adjacentto the d Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-american-annual-of-photography-lta-tm-adl-m-mmmmm-veloper-fog-is-caused-by-impurities-or-oxidation-products-ofthe-developing-agent-formed-during-mixing-the-importanceof-careful-mixing-of-developers-for-test-purposes-is-thereforeapparent-see-page-i86-mixing-the-developer-interesting-facts-about-fog-are-that-the-fog-layer-is-notdistributed-evenly-over-the-entire-image-but-is-thinner-in-thehighlights-because-in-those-places-where-more-image-is-de-veloped-more-potassium-bromide-is-formed-as-a-reaction-pro-duct-of-development-which-therefore-restrains-the-fog-adjacentto-the-d-image339977139.htmlRM2AN388K–The American annual of photography . <a tm -A...d^l m-.^M;mmmm. veloper fog is caused by impurities or oxidation products ofthe developing agent formed during mixing. The importanceof careful mixing of developers for test purposes is thereforeapparent (See page i86 Mixing the Developer). Interesting facts about fog are that the fog layer is notdistributed evenly over the entire image, but is thinner in thehighlights because in those places where more image is de-veloped, more potassium bromide is formed as a reaction pro-duct of development which therefore restrains the fog adjacentto the d
 The American annual of photography . Figure 2. The negative is washed until perfectly free from hypo, orif dry is soaked in running water for fifteen minutes. It isthen placed in the following solution:— Copper sulphate 3 grammes Potassium bromide 3 grammes Water 250 cc. which is allowed to act until the highest lights of the negativeare white when viewed from the back. It is then washed for fifteen minutes and developed in M-0,or a similar developer, until only the highest lights remainwhite as viewed from the back. The negative is then rinsed and placed in hypo 25%, whereit is allowed to rem Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-american-annual-of-photography-figure-2-the-negative-is-washed-until-perfectly-free-from-hypo-orif-dry-is-soaked-in-running-water-for-fifteen-minutes-it-isthen-placed-in-the-following-solution-copper-sulphate-3-grammes-potassium-bromide-3-grammes-water-250-cc-which-is-allowed-to-act-until-the-highest-lights-of-the-negativeare-white-when-viewed-from-the-back-it-is-then-washed-for-fifteen-minutes-and-developed-in-m-0or-a-similar-developer-until-only-the-highest-lights-remainwhite-as-viewed-from-the-back-the-negative-is-then-rinsed-and-placed-in-hypo-25-whereit-is-allowed-to-rem-image339154916.html
The American annual of photography . Figure 2. The negative is washed until perfectly free from hypo, orif dry is soaked in running water for fifteen minutes. It isthen placed in the following solution:— Copper sulphate 3 grammes Potassium bromide 3 grammes Water 250 cc. which is allowed to act until the highest lights of the negativeare white when viewed from the back. It is then washed for fifteen minutes and developed in M-0,or a similar developer, until only the highest lights remainwhite as viewed from the back. The negative is then rinsed and placed in hypo 25%, whereit is allowed to rem Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-american-annual-of-photography-figure-2-the-negative-is-washed-until-perfectly-free-from-hypo-orif-dry-is-soaked-in-running-water-for-fifteen-minutes-it-isthen-placed-in-the-following-solution-copper-sulphate-3-grammes-potassium-bromide-3-grammes-water-250-cc-which-is-allowed-to-act-until-the-highest-lights-of-the-negativeare-white-when-viewed-from-the-back-it-is-then-washed-for-fifteen-minutes-and-developed-in-m-0or-a-similar-developer-until-only-the-highest-lights-remainwhite-as-viewed-from-the-back-the-negative-is-then-rinsed-and-placed-in-hypo-25-whereit-is-allowed-to-rem-image339154916.htmlRM2AKNRFG–The American annual of photography . Figure 2. The negative is washed until perfectly free from hypo, orif dry is soaked in running water for fifteen minutes. It isthen placed in the following solution:— Copper sulphate 3 grammes Potassium bromide 3 grammes Water 250 cc. which is allowed to act until the highest lights of the negativeare white when viewed from the back. It is then washed for fifteen minutes and developed in M-0,or a similar developer, until only the highest lights remainwhite as viewed from the back. The negative is then rinsed and placed in hypo 25%, whereit is allowed to rem
 The American annual of photography . .75 .5 0.85 Potassium, B i c a r -bonate Potassium, Bichro-mate Potassium, Bromide. Potassium, Carbon-ate Potassium, Chloro-platinite Potassium, Cyanide. Potassium, Ferricy-anide Potassium, Ferrocy-anide Potassium, Iodide... Potassium, Metabi-sulphite Potassium, Oxalate.. Potassium, Perman-ganate Potassium, Persul-phate Potassium, Sulpho-cyanide Pyrocatechin Rochelle Salt Silver, Nitrate Sodium, Acetate.... Sodium, Bicarbonate Sodium, Bisulphite.. Sodium, Bromide. . Sodium, Carbonate(dry) Sodium, Carbonate(cryst) Sodium, Chloride. .. Sodium, Citrate.... Sod Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-american-annual-of-photography-75-5-085-potassium-b-i-c-a-r-bonate-potassium-bichro-mate-potassium-bromide-potassium-carbon-ate-potassium-chloro-platinite-potassium-cyanide-potassium-ferricy-anide-potassium-ferrocy-anide-potassium-iodide-potassium-metabi-sulphite-potassium-oxalate-potassium-perman-ganate-potassium-persul-phate-potassium-sulpho-cyanide-pyrocatechin-rochelle-salt-silver-nitrate-sodium-acetate-sodium-bicarbonate-sodium-bisulphite-sodium-bromide-sodium-carbonatedry-sodium-carbonatecryst-sodium-chloride-sodium-citrate-sod-image338931496.html
The American annual of photography . .75 .5 0.85 Potassium, B i c a r -bonate Potassium, Bichro-mate Potassium, Bromide. Potassium, Carbon-ate Potassium, Chloro-platinite Potassium, Cyanide. Potassium, Ferricy-anide Potassium, Ferrocy-anide Potassium, Iodide... Potassium, Metabi-sulphite Potassium, Oxalate.. Potassium, Perman-ganate Potassium, Persul-phate Potassium, Sulpho-cyanide Pyrocatechin Rochelle Salt Silver, Nitrate Sodium, Acetate.... Sodium, Bicarbonate Sodium, Bisulphite.. Sodium, Bromide. . Sodium, Carbonate(dry) Sodium, Carbonate(cryst) Sodium, Chloride. .. Sodium, Citrate.... Sod Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-american-annual-of-photography-75-5-085-potassium-b-i-c-a-r-bonate-potassium-bichro-mate-potassium-bromide-potassium-carbon-ate-potassium-chloro-platinite-potassium-cyanide-potassium-ferricy-anide-potassium-ferrocy-anide-potassium-iodide-potassium-metabi-sulphite-potassium-oxalate-potassium-perman-ganate-potassium-persul-phate-potassium-sulpho-cyanide-pyrocatechin-rochelle-salt-silver-nitrate-sodium-acetate-sodium-bicarbonate-sodium-bisulphite-sodium-bromide-sodium-carbonatedry-sodium-carbonatecryst-sodium-chloride-sodium-citrate-sod-image338931496.htmlRM2AKBJG8–The American annual of photography . .75 .5 0.85 Potassium, B i c a r -bonate Potassium, Bichro-mate Potassium, Bromide. Potassium, Carbon-ate Potassium, Chloro-platinite Potassium, Cyanide. Potassium, Ferricy-anide Potassium, Ferrocy-anide Potassium, Iodide... Potassium, Metabi-sulphite Potassium, Oxalate.. Potassium, Perman-ganate Potassium, Persul-phate Potassium, Sulpho-cyanide Pyrocatechin Rochelle Salt Silver, Nitrate Sodium, Acetate.... Sodium, Bicarbonate Sodium, Bisulphite.. Sodium, Bromide. . Sodium, Carbonate(dry) Sodium, Carbonate(cryst) Sodium, Chloride. .. Sodium, Citrate.... Sod
 The American annual of photography . ur, who issatisfied with anything as long as it is a print or printable, butfor the more advanced worker who knows what he wants toget in advance, and is willing to adapt his methods to meetthe desired results. We will start the process backwards, by making some printsfirst and our negatives later, as the results can be better ob-served in the prints. But first let us prepare our developer as follows: A Metol or Satropol i5 gi- Sodium Sulphite (dry). 80 Sodium Carbonate (dry). 60 Potassium Bromide i Water 5 oz. B Hydroquinone ^5 S^- Sodium Sulphite (dry). 8 Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-american-annual-of-photography-ur-who-issatisfied-with-anything-as-long-as-it-is-a-print-or-printable-butfor-the-more-advanced-worker-who-knows-what-he-wants-toget-in-advance-and-is-willing-to-adapt-his-methods-to-meetthe-desired-results-we-will-start-the-process-backwards-by-making-some-printsfirst-and-our-negatives-later-as-the-results-can-be-better-ob-served-in-the-prints-but-first-let-us-prepare-our-developer-as-follows-a-metol-or-satropol-i5-gi-sodium-sulphite-dry-80-sodium-carbonate-dry-60-potassium-bromide-i-water-5-oz-b-hydroquinone-5-s-sodium-sulphite-dry-8-image340220141.html
The American annual of photography . ur, who issatisfied with anything as long as it is a print or printable, butfor the more advanced worker who knows what he wants toget in advance, and is willing to adapt his methods to meetthe desired results. We will start the process backwards, by making some printsfirst and our negatives later, as the results can be better ob-served in the prints. But first let us prepare our developer as follows: A Metol or Satropol i5 gi- Sodium Sulphite (dry). 80 Sodium Carbonate (dry). 60 Potassium Bromide i Water 5 oz. B Hydroquinone ^5 S^- Sodium Sulphite (dry). 8 Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-american-annual-of-photography-ur-who-issatisfied-with-anything-as-long-as-it-is-a-print-or-printable-butfor-the-more-advanced-worker-who-knows-what-he-wants-toget-in-advance-and-is-willing-to-adapt-his-methods-to-meetthe-desired-results-we-will-start-the-process-backwards-by-making-some-printsfirst-and-our-negatives-later-as-the-results-can-be-better-ob-served-in-the-prints-but-first-let-us-prepare-our-developer-as-follows-a-metol-or-satropol-i5-gi-sodium-sulphite-dry-80-sodium-carbonate-dry-60-potassium-bromide-i-water-5-oz-b-hydroquinone-5-s-sodium-sulphite-dry-8-image340220141.htmlRM2ANEA79–The American annual of photography . ur, who issatisfied with anything as long as it is a print or printable, butfor the more advanced worker who knows what he wants toget in advance, and is willing to adapt his methods to meetthe desired results. We will start the process backwards, by making some printsfirst and our negatives later, as the results can be better ob-served in the prints. But first let us prepare our developer as follows: A Metol or Satropol i5 gi- Sodium Sulphite (dry). 80 Sodium Carbonate (dry). 60 Potassium Bromide i Water 5 oz. B Hydroquinone ^5 S^- Sodium Sulphite (dry). 8
 IA study of the conductivity of certain electrolytes in water, methyl and ethyl alcohols, and binary mixtures of these solventsIIThe relation between conductivity and viscosity .. . iodide, sodium iodide,i tloric acid, in mixtures The dissociation ... ! iodide and potassium bromide, Lcohol and to be gre - ? -respond i l >n . o f f or e<The phenomenon haa I •in fluidity resulting on mixing tie components of t othesisofDutoitandAstc titatively for cer i -ee solve its - . cohoi and ethyl alcohol.The ohlrausch (for oa- phere of tne solvent around the ions in solution )shown to hold for binar Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/ia-study-of-the-conductivity-of-certain-electrolytes-in-water-methyl-and-ethyl-alcohols-and-binary-mixtures-of-these-solventsiithe-relation-between-conductivity-and-viscosity-iodide-sodium-iodidei-tloric-acid-in-mixtures-the-dissociation-!-iodide-and-potassium-bromide-lcohol-and-to-be-gre-respond-i-l-gtn-o-f-f-or-eltthe-phenomenon-haa-i-in-fluidity-resulting-on-mixing-tie-components-of-t-othesisofdutoitandastc-titatively-for-cer-i-ee-solve-its-cohoi-and-ethyl-alcoholthe-ohlrausch-for-oa-phere-of-tne-solvent-around-the-ions-in-solution-shown-to-hold-for-binar-image339404223.html
IA study of the conductivity of certain electrolytes in water, methyl and ethyl alcohols, and binary mixtures of these solventsIIThe relation between conductivity and viscosity .. . iodide, sodium iodide,i tloric acid, in mixtures The dissociation ... ! iodide and potassium bromide, Lcohol and to be gre - ? -respond i l >n . o f f or e<The phenomenon haa I •in fluidity resulting on mixing tie components of t othesisofDutoitandAstc titatively for cer i -ee solve its - . cohoi and ethyl alcohol.The ohlrausch (for oa- phere of tne solvent around the ions in solution )shown to hold for binar Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/ia-study-of-the-conductivity-of-certain-electrolytes-in-water-methyl-and-ethyl-alcohols-and-binary-mixtures-of-these-solventsiithe-relation-between-conductivity-and-viscosity-iodide-sodium-iodidei-tloric-acid-in-mixtures-the-dissociation-!-iodide-and-potassium-bromide-lcohol-and-to-be-gre-respond-i-l-gtn-o-f-f-or-eltthe-phenomenon-haa-i-in-fluidity-resulting-on-mixing-tie-components-of-t-othesisofdutoitandastc-titatively-for-cer-i-ee-solve-its-cohoi-and-ethyl-alcoholthe-ohlrausch-for-oa-phere-of-tne-solvent-around-the-ions-in-solution-shown-to-hold-for-binar-image339404223.htmlRM2AM55FB–IA study of the conductivity of certain electrolytes in water, methyl and ethyl alcohols, and binary mixtures of these solventsIIThe relation between conductivity and viscosity .. . iodide, sodium iodide,i tloric acid, in mixtures The dissociation ... ! iodide and potassium bromide, Lcohol and to be gre - ? -respond i l >n . o f f or e<The phenomenon haa I •in fluidity resulting on mixing tie components of t othesisofDutoitandAstc titatively for cer i -ee solve its - . cohoi and ethyl alcohol.The ohlrausch (for oa- phere of tne solvent around the ions in solution )shown to hold for binar
 Outing . lmost any extentwithout producing fog. The One-Solution Developer. stock-solution. Metol So grains. Hydroquinone 40 Potassium bromide 40 Sodium sulphite 480 Potassium carbonate 240 Water to make 10 ounces. Put the salts into a ten-ounce bottle,nil it up with water and dissolve, apply-ing heat if necessary. To prepare adeveloping solution, take one part ofthe stock-solution and three, four, orfive parts of water. One to three willgenerally be found just right. In conclusion, I would repeat that thecamera should be part of the impedi-menta of every traveler, tourist andsportsman ; that Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/outing-lmost-any-extentwithout-producing-fog-the-one-solution-developer-stock-solution-metol-so-grains-hydroquinone-40-potassium-bromide-40-sodium-sulphite-480-potassium-carbonate-240-water-to-make-10-ounces-put-the-salts-into-a-ten-ounce-bottlenil-it-up-with-water-and-dissolve-apply-ing-heat-if-necessary-to-prepare-adeveloping-solution-take-one-part-ofthe-stock-solution-and-three-four-orfive-parts-of-water-one-to-three-willgenerally-be-found-just-right-in-conclusion-i-would-repeat-that-thecamera-should-be-part-of-the-impedi-menta-of-every-traveler-tourist-andsportsman-that-image339993192.html
Outing . lmost any extentwithout producing fog. The One-Solution Developer. stock-solution. Metol So grains. Hydroquinone 40 Potassium bromide 40 Sodium sulphite 480 Potassium carbonate 240 Water to make 10 ounces. Put the salts into a ten-ounce bottle,nil it up with water and dissolve, apply-ing heat if necessary. To prepare adeveloping solution, take one part ofthe stock-solution and three, four, orfive parts of water. One to three willgenerally be found just right. In conclusion, I would repeat that thecamera should be part of the impedi-menta of every traveler, tourist andsportsman ; that Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/outing-lmost-any-extentwithout-producing-fog-the-one-solution-developer-stock-solution-metol-so-grains-hydroquinone-40-potassium-bromide-40-sodium-sulphite-480-potassium-carbonate-240-water-to-make-10-ounces-put-the-salts-into-a-ten-ounce-bottlenil-it-up-with-water-and-dissolve-apply-ing-heat-if-necessary-to-prepare-adeveloping-solution-take-one-part-ofthe-stock-solution-and-three-four-orfive-parts-of-water-one-to-three-willgenerally-be-found-just-right-in-conclusion-i-would-repeat-that-thecamera-should-be-part-of-the-impedi-menta-of-every-traveler-tourist-andsportsman-that-image339993192.htmlRM2AN40P0–Outing . lmost any extentwithout producing fog. The One-Solution Developer. stock-solution. Metol So grains. Hydroquinone 40 Potassium bromide 40 Sodium sulphite 480 Potassium carbonate 240 Water to make 10 ounces. Put the salts into a ten-ounce bottle,nil it up with water and dissolve, apply-ing heat if necessary. To prepare adeveloping solution, take one part ofthe stock-solution and three, four, orfive parts of water. One to three willgenerally be found just right. In conclusion, I would repeat that thecamera should be part of the impedi-menta of every traveler, tourist andsportsman ; that
 The American annual of photography . be added to increase the bujk, the onlydifference behig that the time of development is somewhatlengthened. The following formula will be found most usefulas to keeping qualities and application: English. Metric. 80 ozs. Water 2500 c.c. I oz. 6 drams Sodium Carbonate 52.00 gms. 1 oz. 5 drams Sodium Sulphite 50.00 gms. 2 drams Hydrochinone 8.00 gms. 2 scruples Metol 2.60 gms. 5 grains Potassium bromide 0-325 gm. The sodas and bromide should be dissolved first, then thehydrochinone added, but the metol should be dissolved in aseparate quantity of water and th Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-american-annual-of-photography-be-added-to-increase-the-bujk-the-onlydifference-behig-that-the-time-of-development-is-somewhatlengthened-the-following-formula-will-be-found-most-usefulas-to-keeping-qualities-and-application-english-metric-80-ozs-water-2500-cc-i-oz-6-drams-sodium-carbonate-5200-gms-1-oz-5-drams-sodium-sulphite-5000-gms-2-drams-hydrochinone-800-gms-2-scruples-metol-260-gms-5-grains-potassium-bromide-0-325-gm-the-sodas-and-bromide-should-be-dissolved-first-then-thehydrochinone-added-but-the-metol-should-be-dissolved-in-aseparate-quantity-of-water-and-th-image338320784.html
The American annual of photography . be added to increase the bujk, the onlydifference behig that the time of development is somewhatlengthened. The following formula will be found most usefulas to keeping qualities and application: English. Metric. 80 ozs. Water 2500 c.c. I oz. 6 drams Sodium Carbonate 52.00 gms. 1 oz. 5 drams Sodium Sulphite 50.00 gms. 2 drams Hydrochinone 8.00 gms. 2 scruples Metol 2.60 gms. 5 grains Potassium bromide 0-325 gm. The sodas and bromide should be dissolved first, then thehydrochinone added, but the metol should be dissolved in aseparate quantity of water and th Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-american-annual-of-photography-be-added-to-increase-the-bujk-the-onlydifference-behig-that-the-time-of-development-is-somewhatlengthened-the-following-formula-will-be-found-most-usefulas-to-keeping-qualities-and-application-english-metric-80-ozs-water-2500-cc-i-oz-6-drams-sodium-carbonate-5200-gms-1-oz-5-drams-sodium-sulphite-5000-gms-2-drams-hydrochinone-800-gms-2-scruples-metol-260-gms-5-grains-potassium-bromide-0-325-gm-the-sodas-and-bromide-should-be-dissolved-first-then-thehydrochinone-added-but-the-metol-should-be-dissolved-in-aseparate-quantity-of-water-and-th-image338320784.htmlRM2AJBRH4–The American annual of photography . be added to increase the bujk, the onlydifference behig that the time of development is somewhatlengthened. The following formula will be found most usefulas to keeping qualities and application: English. Metric. 80 ozs. Water 2500 c.c. I oz. 6 drams Sodium Carbonate 52.00 gms. 1 oz. 5 drams Sodium Sulphite 50.00 gms. 2 drams Hydrochinone 8.00 gms. 2 scruples Metol 2.60 gms. 5 grains Potassium bromide 0-325 gm. The sodas and bromide should be dissolved first, then thehydrochinone added, but the metol should be dissolved in aseparate quantity of water and th
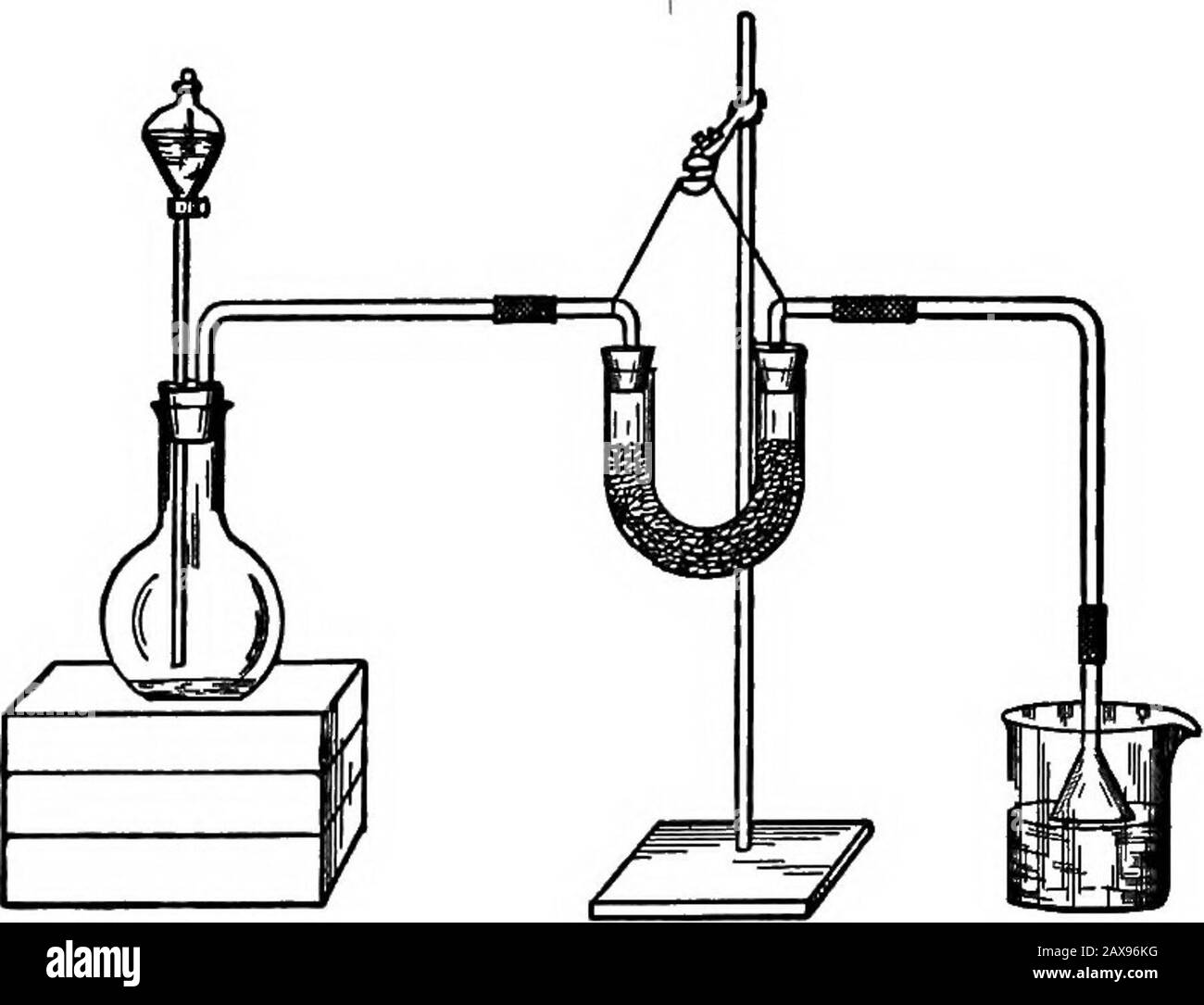 A college text-book of chemistry . ams manganese dioxide. Put the mixture into a 500 cc.. flask ; connect with a condenser (see Fig. 28). Mix 15 cc. con-centrated sulphuric acid and 90 cc. water. After cooling pourthe liquid on the mixture in the flask. Gently heat, when brominewill be given off in the form of vapor. A part of this will con-dense and collect in the receiver. Perform this experiment undera hood with a good draught. Htdrobromic Acid. ^ Experiment 82.—In a small porcelain evaporating-dish put afew crystals of potassium bromide. Pour on them a few drops ofconcentrated sulphuric ac Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/a-college-text-book-of-chemistry-ams-manganese-dioxide-put-the-mixture-into-a-500-cc-flask-connect-with-a-condenser-see-fig-28-mix-15-cc-con-centrated-sulphuric-acid-and-90-cc-water-after-cooling-pourthe-liquid-on-the-mixture-in-the-flask-gently-heat-when-brominewill-be-given-off-in-the-form-of-vapor-a-part-of-this-will-con-dense-and-collect-in-the-receiver-perform-this-experiment-undera-hood-with-a-good-draught-htdrobromic-acid-experiment-82in-a-small-porcelain-evaporating-dish-put-afew-crystals-of-potassium-bromide-pour-on-them-a-few-drops-ofconcentrated-sulphuric-ac-image343180868.html
A college text-book of chemistry . ams manganese dioxide. Put the mixture into a 500 cc.. flask ; connect with a condenser (see Fig. 28). Mix 15 cc. con-centrated sulphuric acid and 90 cc. water. After cooling pourthe liquid on the mixture in the flask. Gently heat, when brominewill be given off in the form of vapor. A part of this will con-dense and collect in the receiver. Perform this experiment undera hood with a good draught. Htdrobromic Acid. ^ Experiment 82.—In a small porcelain evaporating-dish put afew crystals of potassium bromide. Pour on them a few drops ofconcentrated sulphuric ac Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/a-college-text-book-of-chemistry-ams-manganese-dioxide-put-the-mixture-into-a-500-cc-flask-connect-with-a-condenser-see-fig-28-mix-15-cc-con-centrated-sulphuric-acid-and-90-cc-water-after-cooling-pourthe-liquid-on-the-mixture-in-the-flask-gently-heat-when-brominewill-be-given-off-in-the-form-of-vapor-a-part-of-this-will-con-dense-and-collect-in-the-receiver-perform-this-experiment-undera-hood-with-a-good-draught-htdrobromic-acid-experiment-82in-a-small-porcelain-evaporating-dish-put-afew-crystals-of-potassium-bromide-pour-on-them-a-few-drops-ofconcentrated-sulphuric-ac-image343180868.htmlRM2AX96KG–A college text-book of chemistry . ams manganese dioxide. Put the mixture into a 500 cc.. flask ; connect with a condenser (see Fig. 28). Mix 15 cc. con-centrated sulphuric acid and 90 cc. water. After cooling pourthe liquid on the mixture in the flask. Gently heat, when brominewill be given off in the form of vapor. A part of this will con-dense and collect in the receiver. Perform this experiment undera hood with a good draught. Htdrobromic Acid. ^ Experiment 82.—In a small porcelain evaporating-dish put afew crystals of potassium bromide. Pour on them a few drops ofconcentrated sulphuric ac
 . The book of photography; practical, theoretical and applied. excess, it may be boiled for twenty min-utes ; or with 160 grains in excess, forseven minutes. If these times are ex-ceeded, the emulsion comm-snces to showsigns of fogging, any trace of which would,of course, be exceedingly injurious. Theincrease of sensitiveness is said to be dueto the enlargement of the particles, whichmay be explained by the theory of crystal-lisation. Just as a crystal of alum willgrow in a saturated solution of the samesubstance, so the potassium bromide asa solvent of the haloids will cause growthof the part Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-book-of-photography-practical-theoretical-and-applied-excess-it-may-be-boiled-for-twenty-min-utes-or-with-160-grains-in-excess-forseven-minutes-if-these-times-are-ex-ceeded-the-emulsion-comm-snces-to-showsigns-of-fogging-any-trace-of-which-wouldof-course-be-exceedingly-injurious-theincrease-of-sensitiveness-is-said-to-be-dueto-the-enlargement-of-the-particles-whichmay-be-explained-by-the-theory-of-crystal-lisation-just-as-a-crystal-of-alum-willgrow-in-a-saturated-solution-of-the-samesubstance-so-the-potassium-bromide-asa-solvent-of-the-haloids-will-cause-growthof-the-part-image370333519.html
. The book of photography; practical, theoretical and applied. excess, it may be boiled for twenty min-utes ; or with 160 grains in excess, forseven minutes. If these times are ex-ceeded, the emulsion comm-snces to showsigns of fogging, any trace of which would,of course, be exceedingly injurious. Theincrease of sensitiveness is said to be dueto the enlargement of the particles, whichmay be explained by the theory of crystal-lisation. Just as a crystal of alum willgrow in a saturated solution of the samesubstance, so the potassium bromide asa solvent of the haloids will cause growthof the part Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-book-of-photography-practical-theoretical-and-applied-excess-it-may-be-boiled-for-twenty-min-utes-or-with-160-grains-in-excess-forseven-minutes-if-these-times-are-ex-ceeded-the-emulsion-comm-snces-to-showsigns-of-fogging-any-trace-of-which-wouldof-course-be-exceedingly-injurious-theincrease-of-sensitiveness-is-said-to-be-dueto-the-enlargement-of-the-particles-whichmay-be-explained-by-the-theory-of-crystal-lisation-just-as-a-crystal-of-alum-willgrow-in-a-saturated-solution-of-the-samesubstance-so-the-potassium-bromide-asa-solvent-of-the-haloids-will-cause-growthof-the-part-image370333519.htmlRM2CEE453–. The book of photography; practical, theoretical and applied. excess, it may be boiled for twenty min-utes ; or with 160 grains in excess, forseven minutes. If these times are ex-ceeded, the emulsion comm-snces to showsigns of fogging, any trace of which would,of course, be exceedingly injurious. Theincrease of sensitiveness is said to be dueto the enlargement of the particles, whichmay be explained by the theory of crystal-lisation. Just as a crystal of alum willgrow in a saturated solution of the samesubstance, so the potassium bromide asa solvent of the haloids will cause growthof the part
 . Save our birds and game . chloral, chloroform, chamomileoil, phsostigma, tobacco and potassium bromide, the latter being^soslow of action it is rarely available. The bladder must be frequentlyevacuated, to prevent re-absorbtion of the poison. If poisoned, use any of these remedies that can be the most easilyobtained and send for your physician at once. Vermin must be destroyed, if we hope to increase our small game,or other animals or birds, or our wild birds other than game birds. Respectfully yours, JOSEPH KALBFUS,Secretarv of Game Commission. 001 259 379 ft LIBRARY OF CONGRg 000ia5=13V^A Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/save-our-birds-and-game-chloral-chloroform-chamomileoil-phsostigma-tobacco-and-potassium-bromide-the-latter-beingsoslow-of-action-it-is-rarely-available-the-bladder-must-be-frequentlyevacuated-to-prevent-re-absorbtion-of-the-poison-if-poisoned-use-any-of-these-remedies-that-can-be-the-most-easilyobtained-and-send-for-your-physician-at-once-vermin-must-be-destroyed-if-we-hope-to-increase-our-small-gameor-other-animals-or-birds-or-our-wild-birds-other-than-game-birds-respectfully-yours-joseph-kalbfussecretarv-of-game-commission-001-259-379-ft-library-of-congrg-000ia5=13va-image374924249.html
. Save our birds and game . chloral, chloroform, chamomileoil, phsostigma, tobacco and potassium bromide, the latter being^soslow of action it is rarely available. The bladder must be frequentlyevacuated, to prevent re-absorbtion of the poison. If poisoned, use any of these remedies that can be the most easilyobtained and send for your physician at once. Vermin must be destroyed, if we hope to increase our small game,or other animals or birds, or our wild birds other than game birds. Respectfully yours, JOSEPH KALBFUS,Secretarv of Game Commission. 001 259 379 ft LIBRARY OF CONGRg 000ia5=13V^A Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/save-our-birds-and-game-chloral-chloroform-chamomileoil-phsostigma-tobacco-and-potassium-bromide-the-latter-beingsoslow-of-action-it-is-rarely-available-the-bladder-must-be-frequentlyevacuated-to-prevent-re-absorbtion-of-the-poison-if-poisoned-use-any-of-these-remedies-that-can-be-the-most-easilyobtained-and-send-for-your-physician-at-once-vermin-must-be-destroyed-if-we-hope-to-increase-our-small-gameor-other-animals-or-birds-or-our-wild-birds-other-than-game-birds-respectfully-yours-joseph-kalbfussecretarv-of-game-commission-001-259-379-ft-library-of-congrg-000ia5=13va-image374924249.htmlRM2CNY7KN–. Save our birds and game . chloral, chloroform, chamomileoil, phsostigma, tobacco and potassium bromide, the latter being^soslow of action it is rarely available. The bladder must be frequentlyevacuated, to prevent re-absorbtion of the poison. If poisoned, use any of these remedies that can be the most easilyobtained and send for your physician at once. Vermin must be destroyed, if we hope to increase our small game,or other animals or birds, or our wild birds other than game birds. Respectfully yours, JOSEPH KALBFUS,Secretarv of Game Commission. 001 259 379 ft LIBRARY OF CONGRg 000ia5=13V^A
 . American journal of pharmacy . , Potassium Bromide,Bromine, Potassium Acetate, Calcium Sulphite, (Sulphite Lime.) Propylamine. Propylamine Chloride, Sodium Sulphite,Sodium Acetate, Scale Irons, etc, etc. (COPT.) Award to BILLINGS, CLAPF <fc CO., BOSTON. The undersigned, having examined the products herein described, respectfully recommendsthe same to the United States Centennial Commission for Award, for the following reasons,namely: A very fine display of Chemicals, especially Carbolic Acidv Propylamine (Trimethylamine),Chloride of Propylamine, and also of Pharmaceutical Chemicals, such Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/american-journal-of-pharmacy-potassium-bromidebromine-potassium-acetate-calcium-sulphite-sulphite-lime-propylamine-propylamine-chloride-sodium-sulphitesodium-acetate-scale-irons-etc-etc-copt-award-to-billings-clapf-ltfc-co-boston-the-undersigned-having-examined-the-products-herein-described-respectfully-recommendsthe-same-to-the-united-states-centennial-commission-for-award-for-the-following-reasonsnamely-a-very-fine-display-of-chemicals-especially-carbolic-acidv-propylamine-trimethylaminechloride-of-propylamine-and-also-of-pharmaceutical-chemicals-such-image370157499.html
. American journal of pharmacy . , Potassium Bromide,Bromine, Potassium Acetate, Calcium Sulphite, (Sulphite Lime.) Propylamine. Propylamine Chloride, Sodium Sulphite,Sodium Acetate, Scale Irons, etc, etc. (COPT.) Award to BILLINGS, CLAPF <fc CO., BOSTON. The undersigned, having examined the products herein described, respectfully recommendsthe same to the United States Centennial Commission for Award, for the following reasons,namely: A very fine display of Chemicals, especially Carbolic Acidv Propylamine (Trimethylamine),Chloride of Propylamine, and also of Pharmaceutical Chemicals, such Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/american-journal-of-pharmacy-potassium-bromidebromine-potassium-acetate-calcium-sulphite-sulphite-lime-propylamine-propylamine-chloride-sodium-sulphitesodium-acetate-scale-irons-etc-etc-copt-award-to-billings-clapf-ltfc-co-boston-the-undersigned-having-examined-the-products-herein-described-respectfully-recommendsthe-same-to-the-united-states-centennial-commission-for-award-for-the-following-reasonsnamely-a-very-fine-display-of-chemicals-especially-carbolic-acidv-propylamine-trimethylaminechloride-of-propylamine-and-also-of-pharmaceutical-chemicals-such-image370157499.htmlRM2CE63JK–. American journal of pharmacy . , Potassium Bromide,Bromine, Potassium Acetate, Calcium Sulphite, (Sulphite Lime.) Propylamine. Propylamine Chloride, Sodium Sulphite,Sodium Acetate, Scale Irons, etc, etc. (COPT.) Award to BILLINGS, CLAPF <fc CO., BOSTON. The undersigned, having examined the products herein described, respectfully recommendsthe same to the United States Centennial Commission for Award, for the following reasons,namely: A very fine display of Chemicals, especially Carbolic Acidv Propylamine (Trimethylamine),Chloride of Propylamine, and also of Pharmaceutical Chemicals, such
 . Oral pathology and practice. A text-book for the use of students in dental colleges and a hand-book for dental practitioners. and thepulse grow rapid, a feverish condition supervene, and a state ensuethat causes the most intense anxiety, from the alarming symptomspresented. An acute glossitis will usually, however, end in complete reso-lution without such startling symptoms. It may be necessary,and it is usually advisable, to administer an active cathartic, and topromote diaphoresis by means of potassium bromide, or Doverspowders, with warm drinks. If there is a septic wound it shouldbe open Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/oral-pathology-and-practice-a-text-book-for-the-use-of-students-in-dental-colleges-and-a-hand-book-for-dental-practitioners-and-thepulse-grow-rapid-a-feverish-condition-supervene-and-a-state-ensuethat-causes-the-most-intense-anxiety-from-the-alarming-symptomspresented-an-acute-glossitis-will-usually-however-end-in-complete-reso-lution-without-such-startling-symptoms-it-may-be-necessaryand-it-is-usually-advisable-to-administer-an-active-cathartic-and-topromote-diaphoresis-by-means-of-potassium-bromide-or-doverspowders-with-warm-drinks-if-there-is-a-septic-wound-it-shouldbe-open-image370434013.html
. Oral pathology and practice. A text-book for the use of students in dental colleges and a hand-book for dental practitioners. and thepulse grow rapid, a feverish condition supervene, and a state ensuethat causes the most intense anxiety, from the alarming symptomspresented. An acute glossitis will usually, however, end in complete reso-lution without such startling symptoms. It may be necessary,and it is usually advisable, to administer an active cathartic, and topromote diaphoresis by means of potassium bromide, or Doverspowders, with warm drinks. If there is a septic wound it shouldbe open Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/oral-pathology-and-practice-a-text-book-for-the-use-of-students-in-dental-colleges-and-a-hand-book-for-dental-practitioners-and-thepulse-grow-rapid-a-feverish-condition-supervene-and-a-state-ensuethat-causes-the-most-intense-anxiety-from-the-alarming-symptomspresented-an-acute-glossitis-will-usually-however-end-in-complete-reso-lution-without-such-startling-symptoms-it-may-be-necessaryand-it-is-usually-advisable-to-administer-an-active-cathartic-and-topromote-diaphoresis-by-means-of-potassium-bromide-or-doverspowders-with-warm-drinks-if-there-is-a-septic-wound-it-shouldbe-open-image370434013.htmlRM2CEJMA5–. Oral pathology and practice. A text-book for the use of students in dental colleges and a hand-book for dental practitioners. and thepulse grow rapid, a feverish condition supervene, and a state ensuethat causes the most intense anxiety, from the alarming symptomspresented. An acute glossitis will usually, however, end in complete reso-lution without such startling symptoms. It may be necessary,and it is usually advisable, to administer an active cathartic, and topromote diaphoresis by means of potassium bromide, or Doverspowders, with warm drinks. If there is a septic wound it shouldbe open
 . Original photographs taken on the battlefields during the Civil War of the United States . collodion to whichhe added iodide of potassium and alittle potassium bromide, lie thenpoured the iodized collodion on aclean piece of sheet glass and allowedtwo or three minutes for the film toset. The coated plate was taken intoa dark room. which Brady carriedwith him, and immersed for about aminute in a bath of thirty grains ofsilver nitrate to every ounce of water.The plate was now sensitive to whitelight and must be placed immediatelyin the camera and exposed and devel-oped within five minutes to g Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/original-photographs-taken-on-the-battlefields-during-the-civil-war-of-the-united-states-collodion-to-whichhe-added-iodide-of-potassium-and-alittle-potassium-bromide-lie-thenpoured-the-iodized-collodion-on-aclean-piece-of-sheet-glass-and-allowedtwo-or-three-minutes-for-the-film-toset-the-coated-plate-was-taken-intoa-dark-room-which-brady-carriedwith-him-and-immersed-for-about-aminute-in-a-bath-of-thirty-grains-ofsilver-nitrate-to-every-ounce-of-waterthe-plate-was-now-sensitive-to-whitelight-and-must-be-placed-immediatelyin-the-camera-and-exposed-and-devel-oped-within-five-minutes-to-g-image371682288.html
. Original photographs taken on the battlefields during the Civil War of the United States . collodion to whichhe added iodide of potassium and alittle potassium bromide, lie thenpoured the iodized collodion on aclean piece of sheet glass and allowedtwo or three minutes for the film toset. The coated plate was taken intoa dark room. which Brady carriedwith him, and immersed for about aminute in a bath of thirty grains ofsilver nitrate to every ounce of water.The plate was now sensitive to whitelight and must be placed immediatelyin the camera and exposed and devel-oped within five minutes to g Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/original-photographs-taken-on-the-battlefields-during-the-civil-war-of-the-united-states-collodion-to-whichhe-added-iodide-of-potassium-and-alittle-potassium-bromide-lie-thenpoured-the-iodized-collodion-on-aclean-piece-of-sheet-glass-and-allowedtwo-or-three-minutes-for-the-film-toset-the-coated-plate-was-taken-intoa-dark-room-which-brady-carriedwith-him-and-immersed-for-about-aminute-in-a-bath-of-thirty-grains-ofsilver-nitrate-to-every-ounce-of-waterthe-plate-was-now-sensitive-to-whitelight-and-must-be-placed-immediatelyin-the-camera-and-exposed-and-devel-oped-within-five-minutes-to-g-image371682288.htmlRM2CGKGFC–. Original photographs taken on the battlefields during the Civil War of the United States . collodion to whichhe added iodide of potassium and alittle potassium bromide, lie thenpoured the iodized collodion on aclean piece of sheet glass and allowedtwo or three minutes for the film toset. The coated plate was taken intoa dark room. which Brady carriedwith him, and immersed for about aminute in a bath of thirty grains ofsilver nitrate to every ounce of water.The plate was now sensitive to whitelight and must be placed immediatelyin the camera and exposed and devel-oped within five minutes to g
 . Texas medical journal. d Bromides.—Drs. Eobert J. Preston, Brown-Sequard,Hazard, and other learned m.en of the profession, have stronglyadvocated a combination of bromide salts in preference to the useof potassium bromide alone. The salts of the lighter metals, assodium, ammonium and lithium, seem to have less of the un-toward action than the potassium salt. In Peacocks Bromides wehave a imion of these salts that has proven itself a most availableand trustworthy combination. In this regard we are pleased toquote Dr. Caldwell, who says: ^Tt is a mystery to me why bro-mide of potassium is so g Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/texas-medical-journal-d-bromidesdrs-eobert-j-preston-brown-sequardhazard-and-other-learned-men-of-the-profession-have-stronglyadvocated-a-combination-of-bromide-salts-in-preference-to-the-useof-potassium-bromide-alone-the-salts-of-the-lighter-metals-assodium-ammonium-and-lithium-seem-to-have-less-of-the-un-toward-action-than-the-potassium-salt-in-peacocks-bromides-wehave-a-imion-of-these-salts-that-has-proven-itself-a-most-availableand-trustworthy-combination-in-this-regard-we-are-pleased-toquote-dr-caldwell-who-says-tt-is-a-mystery-to-me-why-bro-mide-of-potassium-is-so-g-image370650593.html
. Texas medical journal. d Bromides.—Drs. Eobert J. Preston, Brown-Sequard,Hazard, and other learned m.en of the profession, have stronglyadvocated a combination of bromide salts in preference to the useof potassium bromide alone. The salts of the lighter metals, assodium, ammonium and lithium, seem to have less of the un-toward action than the potassium salt. In Peacocks Bromides wehave a imion of these salts that has proven itself a most availableand trustworthy combination. In this regard we are pleased toquote Dr. Caldwell, who says: ^Tt is a mystery to me why bro-mide of potassium is so g Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/texas-medical-journal-d-bromidesdrs-eobert-j-preston-brown-sequardhazard-and-other-learned-men-of-the-profession-have-stronglyadvocated-a-combination-of-bromide-salts-in-preference-to-the-useof-potassium-bromide-alone-the-salts-of-the-lighter-metals-assodium-ammonium-and-lithium-seem-to-have-less-of-the-un-toward-action-than-the-potassium-salt-in-peacocks-bromides-wehave-a-imion-of-these-salts-that-has-proven-itself-a-most-availableand-trustworthy-combination-in-this-regard-we-are-pleased-toquote-dr-caldwell-who-says-tt-is-a-mystery-to-me-why-bro-mide-of-potassium-is-so-g-image370650593.htmlRM2CF0GH5–. Texas medical journal. d Bromides.—Drs. Eobert J. Preston, Brown-Sequard,Hazard, and other learned m.en of the profession, have stronglyadvocated a combination of bromide salts in preference to the useof potassium bromide alone. The salts of the lighter metals, assodium, ammonium and lithium, seem to have less of the un-toward action than the potassium salt. In Peacocks Bromides wehave a imion of these salts that has proven itself a most availableand trustworthy combination. In this regard we are pleased toquote Dr. Caldwell, who says: ^Tt is a mystery to me why bro-mide of potassium is so g
 . Practical methods of organic chemistry . Fig. 84. Fig. 85. Hydrochloric acid gas may also be obtained by warming 10parts of sodium chloride with a cold mixture of 3 parts of waterand 18 parts of concentrated sulphuric acid. 3. HYDROBROMIC ACID (see Brombenzene) The hydrobromic acid obtained as a by-product in the bromina-tion reactions is purified by distilling it from a fractionating flask.Water first passes over until finally the temperature remains con-stant at 12 6°, when a 48 % acid goes over; this is collected. In order to prepare potassium bromide for use in the prepara-tion of ethyl Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/practical-methods-of-organic-chemistry-fig-84-fig-85-hydrochloric-acid-gas-may-also-be-obtained-by-warming-10parts-of-sodium-chloride-with-a-cold-mixture-of-3-parts-of-waterand-18-parts-of-concentrated-sulphuric-acid-3-hydrobromic-acid-see-brombenzene-the-hydrobromic-acid-obtained-as-a-by-product-in-the-bromina-tion-reactions-is-purified-by-distilling-it-from-a-fractionating-flaskwater-first-passes-over-until-finally-the-temperature-remains-con-stant-at-12-6-when-a-48-acid-goes-over-this-is-collected-in-order-to-prepare-potassium-bromide-for-use-in-the-prepara-tion-of-ethyl-image370116998.html
. Practical methods of organic chemistry . Fig. 84. Fig. 85. Hydrochloric acid gas may also be obtained by warming 10parts of sodium chloride with a cold mixture of 3 parts of waterand 18 parts of concentrated sulphuric acid. 3. HYDROBROMIC ACID (see Brombenzene) The hydrobromic acid obtained as a by-product in the bromina-tion reactions is purified by distilling it from a fractionating flask.Water first passes over until finally the temperature remains con-stant at 12 6°, when a 48 % acid goes over; this is collected. In order to prepare potassium bromide for use in the prepara-tion of ethyl Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/practical-methods-of-organic-chemistry-fig-84-fig-85-hydrochloric-acid-gas-may-also-be-obtained-by-warming-10parts-of-sodium-chloride-with-a-cold-mixture-of-3-parts-of-waterand-18-parts-of-concentrated-sulphuric-acid-3-hydrobromic-acid-see-brombenzene-the-hydrobromic-acid-obtained-as-a-by-product-in-the-bromina-tion-reactions-is-purified-by-distilling-it-from-a-fractionating-flaskwater-first-passes-over-until-finally-the-temperature-remains-con-stant-at-12-6-when-a-48-acid-goes-over-this-is-collected-in-order-to-prepare-potassium-bromide-for-use-in-the-prepara-tion-of-ethyl-image370116998.htmlRM2CE4806–. Practical methods of organic chemistry . Fig. 84. Fig. 85. Hydrochloric acid gas may also be obtained by warming 10parts of sodium chloride with a cold mixture of 3 parts of waterand 18 parts of concentrated sulphuric acid. 3. HYDROBROMIC ACID (see Brombenzene) The hydrobromic acid obtained as a by-product in the bromina-tion reactions is purified by distilling it from a fractionating flask.Water first passes over until finally the temperature remains con-stant at 12 6°, when a 48 % acid goes over; this is collected. In order to prepare potassium bromide for use in the prepara-tion of ethyl
 . The book of photography; practical, theoretical and applied. ne No. 1 soluble, 30 grs., water 1 oz. ;(6) silver nitrate IVS grs., water i oz. ; (c) potassium bromide 130 grs., water 1 oz. ; (d) potassium iodide 5 grs., water 1 oz. ;() hard gelatine 240 grs., water 2 oz. How Sensitiveness is Governed. The first question likely to be asked bythe novice is what governs the sensitive-ness of the emulsion, and this may bebroadly stated to be the ripening or boil-ing to which it is subjected. (There are,however, other methods of emulsion mak-ing which do not necessitate boiling;which will be dealt Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-book-of-photography-practical-theoretical-and-applied-ne-no-1-soluble-30-grs-water-1-oz-6-silver-nitrate-ivs-grs-water-i-oz-c-potassium-bromide-130-grs-water-1-oz-d-potassium-iodide-5-grs-water-1-oz-hard-gelatine-240-grs-water-2-oz-how-sensitiveness-is-governed-the-first-question-likely-to-be-asked-bythe-novice-is-what-governs-the-sensitive-ness-of-the-emulsion-and-this-may-bebroadly-stated-to-be-the-ripening-or-boil-ing-to-which-it-is-subjected-there-arehowever-other-methods-of-emulsion-mak-ing-which-do-not-necessitate-boilingwhich-will-be-dealt-image370333467.html
. The book of photography; practical, theoretical and applied. ne No. 1 soluble, 30 grs., water 1 oz. ;(6) silver nitrate IVS grs., water i oz. ; (c) potassium bromide 130 grs., water 1 oz. ; (d) potassium iodide 5 grs., water 1 oz. ;() hard gelatine 240 grs., water 2 oz. How Sensitiveness is Governed. The first question likely to be asked bythe novice is what governs the sensitive-ness of the emulsion, and this may bebroadly stated to be the ripening or boil-ing to which it is subjected. (There are,however, other methods of emulsion mak-ing which do not necessitate boiling;which will be dealt Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-book-of-photography-practical-theoretical-and-applied-ne-no-1-soluble-30-grs-water-1-oz-6-silver-nitrate-ivs-grs-water-i-oz-c-potassium-bromide-130-grs-water-1-oz-d-potassium-iodide-5-grs-water-1-oz-hard-gelatine-240-grs-water-2-oz-how-sensitiveness-is-governed-the-first-question-likely-to-be-asked-bythe-novice-is-what-governs-the-sensitive-ness-of-the-emulsion-and-this-may-bebroadly-stated-to-be-the-ripening-or-boil-ing-to-which-it-is-subjected-there-arehowever-other-methods-of-emulsion-mak-ing-which-do-not-necessitate-boilingwhich-will-be-dealt-image370333467.htmlRM2CEE437–. The book of photography; practical, theoretical and applied. ne No. 1 soluble, 30 grs., water 1 oz. ;(6) silver nitrate IVS grs., water i oz. ; (c) potassium bromide 130 grs., water 1 oz. ; (d) potassium iodide 5 grs., water 1 oz. ;() hard gelatine 240 grs., water 2 oz. How Sensitiveness is Governed. The first question likely to be asked bythe novice is what governs the sensitive-ness of the emulsion, and this may bebroadly stated to be the ripening or boil-ing to which it is subjected. (There are,however, other methods of emulsion mak-ing which do not necessitate boiling;which will be dealt
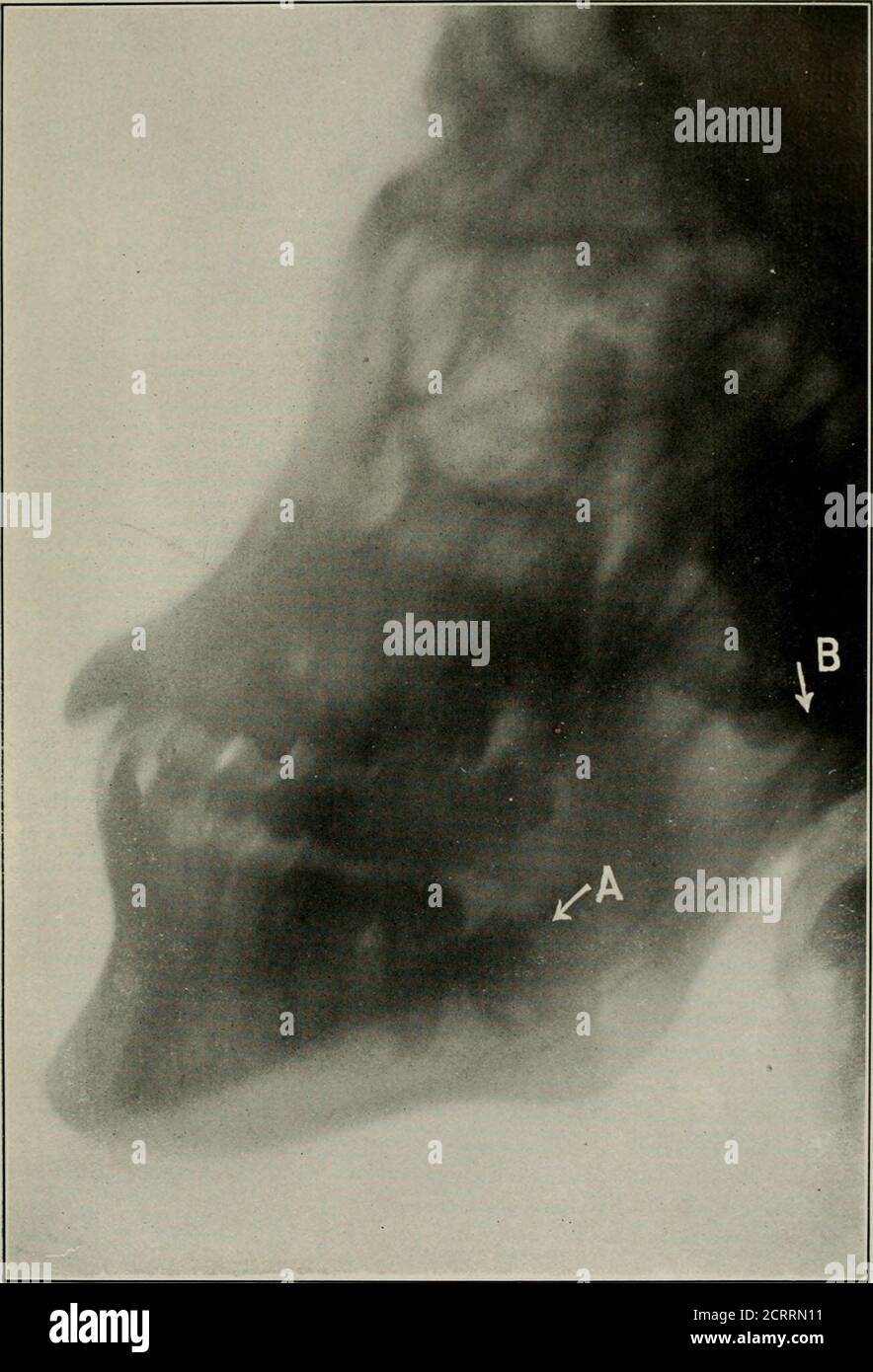 . Elementary and dental radiography . : About 19 inches. 5. Thickness of part: That of the cranium coverings and con-tents, about 8 inches. (Fig. 104.) 6. Density of parts: That of the cranium, coverings and contents. 7. Plate used: Cramer X-ray plate, 8x10 inches, with intensifyingscreen. 8. Time of exposure: Three (3) seconds. 9. Time in developer, and developer used: Seven (7) minutesin water, 32 oz.; soda sulphite, 12 dr.; hydrochinone, 2 dr.; edinol, 75gr.; potassium bromide, 90 gr.; potassium carbonate, 2 oz. Comment: Had an intensifying screen not been used, the time ofexposure would ha Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/elementary-and-dental-radiography-about-19-inches-5-thickness-of-part-that-of-the-cranium-coverings-and-con-tents-about-8-inches-fig-104-6-density-of-parts-that-of-the-cranium-coverings-and-contents-7-plate-used-cramer-x-ray-plate-8x10-inches-with-intensifyingscreen-8-time-of-exposure-three-3-seconds-9-time-in-developer-and-developer-used-seven-7-minutesin-water-32-oz-soda-sulphite-12-dr-hydrochinone-2-dr-edinol-75gr-potassium-bromide-90-gr-potassium-carbonate-2-oz-comment-had-an-intensifying-screen-not-been-used-the-time-ofexposure-would-ha-image376076205.html
. Elementary and dental radiography . : About 19 inches. 5. Thickness of part: That of the cranium coverings and con-tents, about 8 inches. (Fig. 104.) 6. Density of parts: That of the cranium, coverings and contents. 7. Plate used: Cramer X-ray plate, 8x10 inches, with intensifyingscreen. 8. Time of exposure: Three (3) seconds. 9. Time in developer, and developer used: Seven (7) minutesin water, 32 oz.; soda sulphite, 12 dr.; hydrochinone, 2 dr.; edinol, 75gr.; potassium bromide, 90 gr.; potassium carbonate, 2 oz. Comment: Had an intensifying screen not been used, the time ofexposure would ha Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/elementary-and-dental-radiography-about-19-inches-5-thickness-of-part-that-of-the-cranium-coverings-and-con-tents-about-8-inches-fig-104-6-density-of-parts-that-of-the-cranium-coverings-and-contents-7-plate-used-cramer-x-ray-plate-8x10-inches-with-intensifyingscreen-8-time-of-exposure-three-3-seconds-9-time-in-developer-and-developer-used-seven-7-minutesin-water-32-oz-soda-sulphite-12-dr-hydrochinone-2-dr-edinol-75gr-potassium-bromide-90-gr-potassium-carbonate-2-oz-comment-had-an-intensifying-screen-not-been-used-the-time-ofexposure-would-ha-image376076205.htmlRM2CRRN11–. Elementary and dental radiography . : About 19 inches. 5. Thickness of part: That of the cranium coverings and con-tents, about 8 inches. (Fig. 104.) 6. Density of parts: That of the cranium, coverings and contents. 7. Plate used: Cramer X-ray plate, 8x10 inches, with intensifyingscreen. 8. Time of exposure: Three (3) seconds. 9. Time in developer, and developer used: Seven (7) minutesin water, 32 oz.; soda sulphite, 12 dr.; hydrochinone, 2 dr.; edinol, 75gr.; potassium bromide, 90 gr.; potassium carbonate, 2 oz. Comment: Had an intensifying screen not been used, the time ofexposure would ha
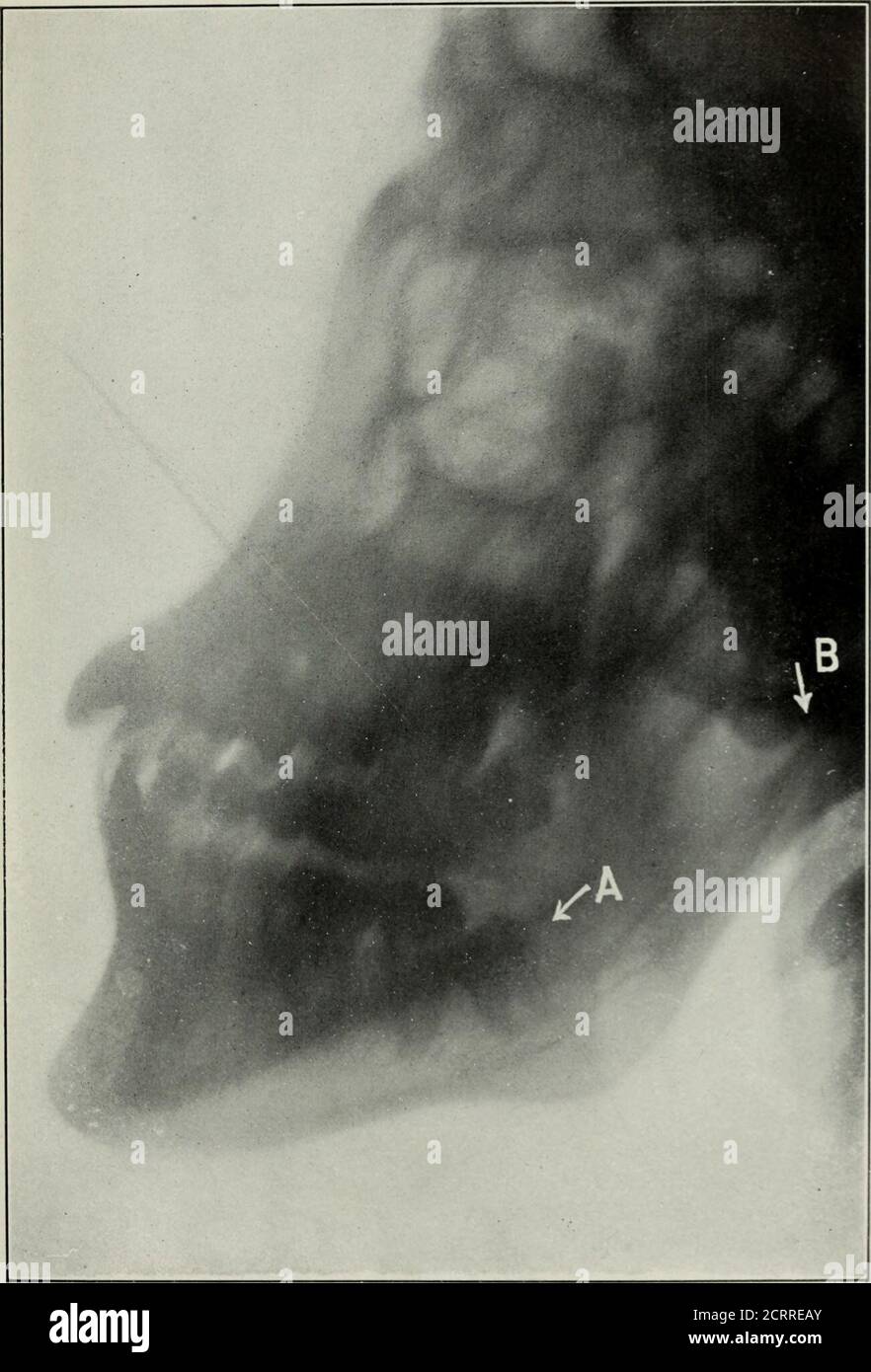 . Elementary and dental radiography . e: About ig inches. 5. Thickness of part: That of the cranium coverings and con-tents, about 8 inches. (Fig. 104.) 6. Density of parts: That of the cranium, coverings and contents. 7. Plate used: Cramer X-ray plate, 8x10 inches, with intensifyingscreen. 8. Time of exposure: Three (3) seconds. 9. Time in developer, and developer used: Seven (7) minutesin water, 32 oz.; soda sulphite, 12 dr.; hydrochinone, 2 dr.; edinol, 75gr.; potassium bromide, 90 gr.; potassium carbonate, 2 oz. Comment: Had an intensifying screen not been used, the time ofexposure would h Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/elementary-and-dental-radiography-e-about-ig-inches-5-thickness-of-part-that-of-the-cranium-coverings-and-con-tents-about-8-inches-fig-104-6-density-of-parts-that-of-the-cranium-coverings-and-contents-7-plate-used-cramer-x-ray-plate-8x10-inches-with-intensifyingscreen-8-time-of-exposure-three-3-seconds-9-time-in-developer-and-developer-used-seven-7-minutesin-water-32-oz-soda-sulphite-12-dr-hydrochinone-2-dr-edinol-75gr-potassium-bromide-90-gr-potassium-carbonate-2-oz-comment-had-an-intensifying-screen-not-been-used-the-time-ofexposure-would-h-image376070995.html
. Elementary and dental radiography . e: About ig inches. 5. Thickness of part: That of the cranium coverings and con-tents, about 8 inches. (Fig. 104.) 6. Density of parts: That of the cranium, coverings and contents. 7. Plate used: Cramer X-ray plate, 8x10 inches, with intensifyingscreen. 8. Time of exposure: Three (3) seconds. 9. Time in developer, and developer used: Seven (7) minutesin water, 32 oz.; soda sulphite, 12 dr.; hydrochinone, 2 dr.; edinol, 75gr.; potassium bromide, 90 gr.; potassium carbonate, 2 oz. Comment: Had an intensifying screen not been used, the time ofexposure would h Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/elementary-and-dental-radiography-e-about-ig-inches-5-thickness-of-part-that-of-the-cranium-coverings-and-con-tents-about-8-inches-fig-104-6-density-of-parts-that-of-the-cranium-coverings-and-contents-7-plate-used-cramer-x-ray-plate-8x10-inches-with-intensifyingscreen-8-time-of-exposure-three-3-seconds-9-time-in-developer-and-developer-used-seven-7-minutesin-water-32-oz-soda-sulphite-12-dr-hydrochinone-2-dr-edinol-75gr-potassium-bromide-90-gr-potassium-carbonate-2-oz-comment-had-an-intensifying-screen-not-been-used-the-time-ofexposure-would-h-image376070995.htmlRM2CRREAY–. Elementary and dental radiography . e: About ig inches. 5. Thickness of part: That of the cranium coverings and con-tents, about 8 inches. (Fig. 104.) 6. Density of parts: That of the cranium, coverings and contents. 7. Plate used: Cramer X-ray plate, 8x10 inches, with intensifyingscreen. 8. Time of exposure: Three (3) seconds. 9. Time in developer, and developer used: Seven (7) minutesin water, 32 oz.; soda sulphite, 12 dr.; hydrochinone, 2 dr.; edinol, 75gr.; potassium bromide, 90 gr.; potassium carbonate, 2 oz. Comment: Had an intensifying screen not been used, the time ofexposure would h
 . The silver sunbeam : a practical and theoretical text-book on sun drawing and photographic printing. Iodide of potassium.Bromide of potassium,Serum, 7 grains.5 «1 ounce. 478 ENLARGING BY THE SERUM OR WHEY PROCESS. This also should be spread upon the paper with a wadof canton flannel. For a developer I use the filtered solution of— Pyrogallic acid 48 grains. Acetic acid No. 8, 4 ounces. Water, 28 Fixing Bath. Hyposulphite of soda, 3 ounces. Carbonate of ammonia, .... ^ ounce. Water, ......... 16 ounces. Fig. 21.. From time to time a little carbonate of ammonia shouldbe added, as the bath is a Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-silver-sunbeam-a-practical-and-theoretical-text-book-on-sun-drawing-and-photographic-printing-iodide-of-potassiumbromide-of-potassiumserum-7-grains5-1-ounce-478-enlarging-by-the-serum-or-whey-process-this-also-should-be-spread-upon-the-paper-with-a-wadof-canton-flannel-for-a-developer-i-use-the-filtered-solution-of-pyrogallic-acid-48-grains-acetic-acid-no-8-4-ounces-water-28-fixing-bath-hyposulphite-of-soda-3-ounces-carbonate-of-ammonia-ounce-water-16-ounces-fig-21-from-time-to-time-a-little-carbonate-of-ammonia-shouldbe-added-as-the-bath-is-a-image370469770.html
. The silver sunbeam : a practical and theoretical text-book on sun drawing and photographic printing. Iodide of potassium.Bromide of potassium,Serum, 7 grains.5 «1 ounce. 478 ENLARGING BY THE SERUM OR WHEY PROCESS. This also should be spread upon the paper with a wadof canton flannel. For a developer I use the filtered solution of— Pyrogallic acid 48 grains. Acetic acid No. 8, 4 ounces. Water, 28 Fixing Bath. Hyposulphite of soda, 3 ounces. Carbonate of ammonia, .... ^ ounce. Water, ......... 16 ounces. Fig. 21.. From time to time a little carbonate of ammonia shouldbe added, as the bath is a Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-silver-sunbeam-a-practical-and-theoretical-text-book-on-sun-drawing-and-photographic-printing-iodide-of-potassiumbromide-of-potassiumserum-7-grains5-1-ounce-478-enlarging-by-the-serum-or-whey-process-this-also-should-be-spread-upon-the-paper-with-a-wadof-canton-flannel-for-a-developer-i-use-the-filtered-solution-of-pyrogallic-acid-48-grains-acetic-acid-no-8-4-ounces-water-28-fixing-bath-hyposulphite-of-soda-3-ounces-carbonate-of-ammonia-ounce-water-16-ounces-fig-21-from-time-to-time-a-little-carbonate-of-ammonia-shouldbe-added-as-the-bath-is-a-image370469770.htmlRM2CEM9Y6–. The silver sunbeam : a practical and theoretical text-book on sun drawing and photographic printing. Iodide of potassium.Bromide of potassium,Serum, 7 grains.5 «1 ounce. 478 ENLARGING BY THE SERUM OR WHEY PROCESS. This also should be spread upon the paper with a wadof canton flannel. For a developer I use the filtered solution of— Pyrogallic acid 48 grains. Acetic acid No. 8, 4 ounces. Water, 28 Fixing Bath. Hyposulphite of soda, 3 ounces. Carbonate of ammonia, .... ^ ounce. Water, ......... 16 ounces. Fig. 21.. From time to time a little carbonate of ammonia shouldbe added, as the bath is a
 . Elementary and dental radiography . n: Mercuric l)ichloride 200 grains (13.3 grammes) Potassium Bromide 120 grains (8.0 grammes) Water (SA ounces U^> c.c.) Keep the plate in this solution a short time, when it will be observed to be bleached uniformly white (the longer the negative is bleached the denser it will ultimately become). Remove from the bleaching solution. wash in running water for a few minutes, then blacken in the following solution : Sodium Sulphite 1 ounce (30 grammes) Water 4 ounces (120 grammes) Or Ammonia 20 minims (1 c.c.) Water 1 ounce * Electro-Therapeutics and Roentg Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/elementary-and-dental-radiography-n-mercuric-lichloride-200-grains-133-grammes-potassium-bromide-120-grains-80-grammes-water-sa-ounces-ugt-cc-keep-the-plate-in-this-solution-a-short-time-when-it-will-be-observed-to-be-bleached-uniformly-white-the-longer-the-negative-is-bleached-the-denser-it-will-ultimately-become-remove-from-the-bleaching-solution-wash-in-running-water-for-a-few-minutes-then-blacken-in-the-following-solution-sodium-sulphite-1-ounce-30-grammes-water-4-ounces-120-grammes-or-ammonia-20-minims-1-cc-water-1-ounce-electro-therapeutics-and-roentg-image376073484.html
. Elementary and dental radiography . n: Mercuric l)ichloride 200 grains (13.3 grammes) Potassium Bromide 120 grains (8.0 grammes) Water (SA ounces U^> c.c.) Keep the plate in this solution a short time, when it will be observed to be bleached uniformly white (the longer the negative is bleached the denser it will ultimately become). Remove from the bleaching solution. wash in running water for a few minutes, then blacken in the following solution : Sodium Sulphite 1 ounce (30 grammes) Water 4 ounces (120 grammes) Or Ammonia 20 minims (1 c.c.) Water 1 ounce * Electro-Therapeutics and Roentg Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/elementary-and-dental-radiography-n-mercuric-lichloride-200-grains-133-grammes-potassium-bromide-120-grains-80-grammes-water-sa-ounces-ugt-cc-keep-the-plate-in-this-solution-a-short-time-when-it-will-be-observed-to-be-bleached-uniformly-white-the-longer-the-negative-is-bleached-the-denser-it-will-ultimately-become-remove-from-the-bleaching-solution-wash-in-running-water-for-a-few-minutes-then-blacken-in-the-following-solution-sodium-sulphite-1-ounce-30-grammes-water-4-ounces-120-grammes-or-ammonia-20-minims-1-cc-water-1-ounce-electro-therapeutics-and-roentg-image376073484.htmlRM2CRRHFT–. Elementary and dental radiography . n: Mercuric l)ichloride 200 grains (13.3 grammes) Potassium Bromide 120 grains (8.0 grammes) Water (SA ounces U^> c.c.) Keep the plate in this solution a short time, when it will be observed to be bleached uniformly white (the longer the negative is bleached the denser it will ultimately become). Remove from the bleaching solution. wash in running water for a few minutes, then blacken in the following solution : Sodium Sulphite 1 ounce (30 grammes) Water 4 ounces (120 grammes) Or Ammonia 20 minims (1 c.c.) Water 1 ounce * Electro-Therapeutics and Roentg
 . Elementary and dental radiography . on: Mercuric Bichloride 200 grains (13.3 grammes) Potassium Bromide 120 grains (S.O grammes) Water §y2 ounces (195 c.c.) Keep the plate in this solution a short time, when it will be observed to be bleached uniformly white (the longer the negative is bleached the denser it will ultimately become). Remove from the bleaching solution. wash in running water for a few minutes, then blacken in the following solution : Sodium Sulphite 1 ounce (30 grammes) Water 4 ounces (120 grammes) Or Ammonia 20 minims (1 c.c.) Wrater 1 ounce * Electro-Therapeutics and Roentge Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/elementary-and-dental-radiography-on-mercuric-bichloride-200-grains-133-grammes-potassium-bromide-120-grains-so-grammes-water-y2-ounces-195-cc-keep-the-plate-in-this-solution-a-short-time-when-it-will-be-observed-to-be-bleached-uniformly-white-the-longer-the-negative-is-bleached-the-denser-it-will-ultimately-become-remove-from-the-bleaching-solution-wash-in-running-water-for-a-few-minutes-then-blacken-in-the-following-solution-sodium-sulphite-1-ounce-30-grammes-water-4-ounces-120-grammes-or-ammonia-20-minims-1-cc-wrater-1-ounce-electro-therapeutics-and-roentge-image376077789.html
. Elementary and dental radiography . on: Mercuric Bichloride 200 grains (13.3 grammes) Potassium Bromide 120 grains (S.O grammes) Water §y2 ounces (195 c.c.) Keep the plate in this solution a short time, when it will be observed to be bleached uniformly white (the longer the negative is bleached the denser it will ultimately become). Remove from the bleaching solution. wash in running water for a few minutes, then blacken in the following solution : Sodium Sulphite 1 ounce (30 grammes) Water 4 ounces (120 grammes) Or Ammonia 20 minims (1 c.c.) Wrater 1 ounce * Electro-Therapeutics and Roentge Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/elementary-and-dental-radiography-on-mercuric-bichloride-200-grains-133-grammes-potassium-bromide-120-grains-so-grammes-water-y2-ounces-195-cc-keep-the-plate-in-this-solution-a-short-time-when-it-will-be-observed-to-be-bleached-uniformly-white-the-longer-the-negative-is-bleached-the-denser-it-will-ultimately-become-remove-from-the-bleaching-solution-wash-in-running-water-for-a-few-minutes-then-blacken-in-the-following-solution-sodium-sulphite-1-ounce-30-grammes-water-4-ounces-120-grammes-or-ammonia-20-minims-1-cc-wrater-1-ounce-electro-therapeutics-and-roentge-image376077789.htmlRM2CRRR1H–. Elementary and dental radiography . on: Mercuric Bichloride 200 grains (13.3 grammes) Potassium Bromide 120 grains (S.O grammes) Water §y2 ounces (195 c.c.) Keep the plate in this solution a short time, when it will be observed to be bleached uniformly white (the longer the negative is bleached the denser it will ultimately become). Remove from the bleaching solution. wash in running water for a few minutes, then blacken in the following solution : Sodium Sulphite 1 ounce (30 grammes) Water 4 ounces (120 grammes) Or Ammonia 20 minims (1 c.c.) Wrater 1 ounce * Electro-Therapeutics and Roentge
 . Elementary and dental radiography / by Howard Riley Raper . n: Mercuric Bichloride 200 grains (13.:) grammes) Potassium Bromide 120 grains (8.0 grammes) Water 6^ ounces ( 195 c.c.) Keep the plate in this solution a short time, when it will be observed to be bleached uniformly white (the longer the negative is bleached the denser it will ultimately become). Remove from the bleaching solution. wash in running water for a few minutes, then blacken in the following solution : Sod;um Sulphite 1 ounce (30 grammes) Water 4 ounces (120 grammes) Or Ammonia 20 minims (1 c.c.) Water 1 ounce * Electro-T Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/elementary-and-dental-radiography-by-howard-riley-raper-n-mercuric-bichloride-200-grains-13-grammes-potassium-bromide-120-grains-80-grammes-water-6-ounces-195-cc-keep-the-plate-in-this-solution-a-short-time-when-it-will-be-observed-to-be-bleached-uniformly-white-the-longer-the-negative-is-bleached-the-denser-it-will-ultimately-become-remove-from-the-bleaching-solution-wash-in-running-water-for-a-few-minutes-then-blacken-in-the-following-solution-sodum-sulphite-1-ounce-30-grammes-water-4-ounces-120-grammes-or-ammonia-20-minims-1-cc-water-1-ounce-electro-t-image375979805.html
. Elementary and dental radiography / by Howard Riley Raper . n: Mercuric Bichloride 200 grains (13.:) grammes) Potassium Bromide 120 grains (8.0 grammes) Water 6^ ounces ( 195 c.c.) Keep the plate in this solution a short time, when it will be observed to be bleached uniformly white (the longer the negative is bleached the denser it will ultimately become). Remove from the bleaching solution. wash in running water for a few minutes, then blacken in the following solution : Sod;um Sulphite 1 ounce (30 grammes) Water 4 ounces (120 grammes) Or Ammonia 20 minims (1 c.c.) Water 1 ounce * Electro-T Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/elementary-and-dental-radiography-by-howard-riley-raper-n-mercuric-bichloride-200-grains-13-grammes-potassium-bromide-120-grains-80-grammes-water-6-ounces-195-cc-keep-the-plate-in-this-solution-a-short-time-when-it-will-be-observed-to-be-bleached-uniformly-white-the-longer-the-negative-is-bleached-the-denser-it-will-ultimately-become-remove-from-the-bleaching-solution-wash-in-running-water-for-a-few-minutes-then-blacken-in-the-following-solution-sodum-sulphite-1-ounce-30-grammes-water-4-ounces-120-grammes-or-ammonia-20-minims-1-cc-water-1-ounce-electro-t-image375979805.htmlRM2CRKA25–. Elementary and dental radiography / by Howard Riley Raper . n: Mercuric Bichloride 200 grains (13.:) grammes) Potassium Bromide 120 grains (8.0 grammes) Water 6^ ounces ( 195 c.c.) Keep the plate in this solution a short time, when it will be observed to be bleached uniformly white (the longer the negative is bleached the denser it will ultimately become). Remove from the bleaching solution. wash in running water for a few minutes, then blacken in the following solution : Sod;um Sulphite 1 ounce (30 grammes) Water 4 ounces (120 grammes) Or Ammonia 20 minims (1 c.c.) Water 1 ounce * Electro-T
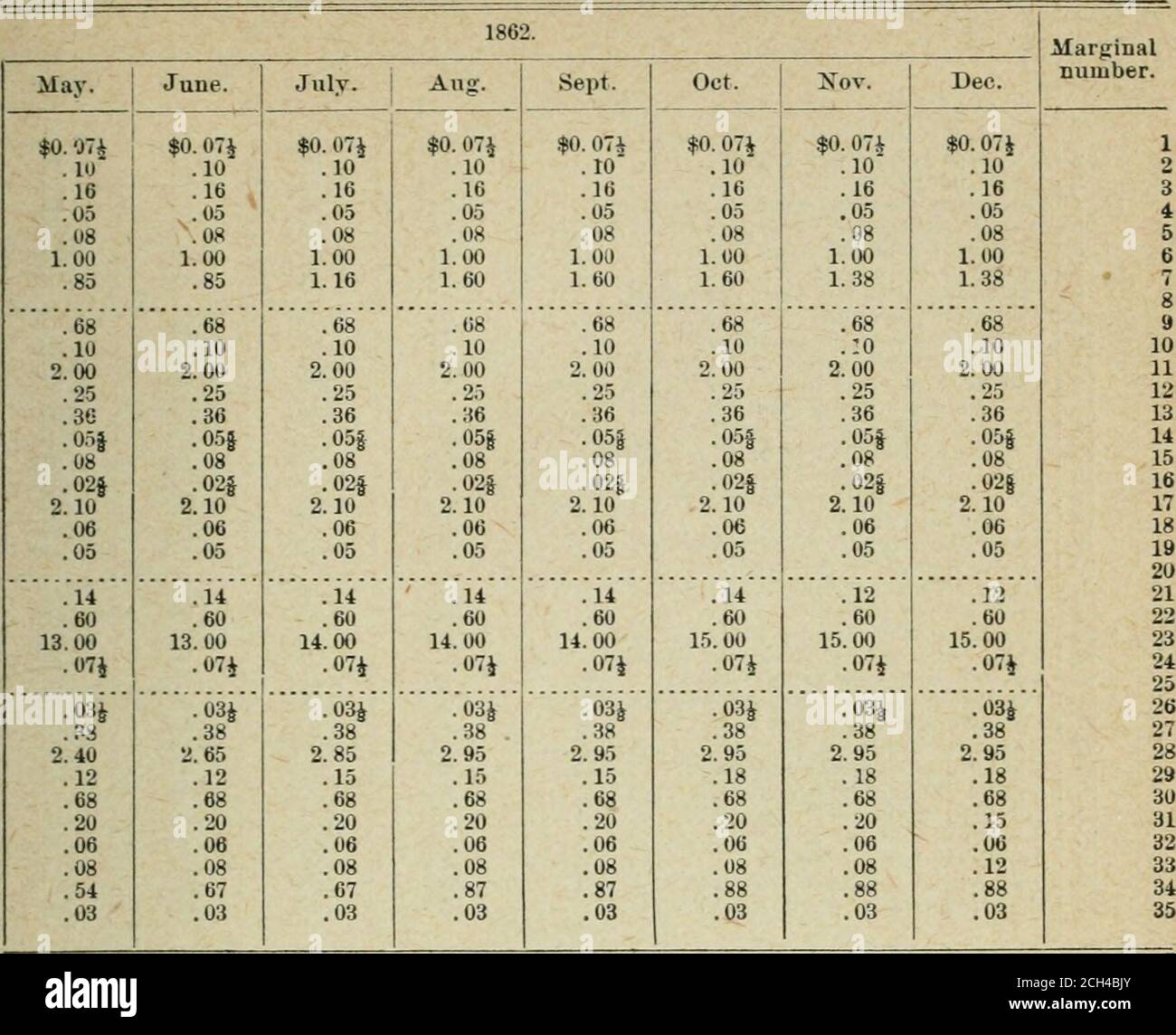 . Wholesale prices, wages, and transportation. Report by Mr. Aldrich, from the Committee on Finance, March 3, 1893 . r yard Nitric acid, per pound Olive oil, per pint Opium, powdered, per pound Pills, compoujid cathartic, per hundred Potassium, bromide of per ounce Potassium, chlorate of, j)owdered, per ounce Potassium, io^lide of per ounce Quinine, sulphate of per ounce Khubarb, powdered, per ounce Squills, syrup of, per pound Sulphur,washed, jier pound Sulphuric acid, per pound... Tartai- emetic, per ounce Turpentine, spirits of per quart Zinc, sulphate of, per ounce 1863. Jan. $0.07i.10.12. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/wholesale-prices-wages-and-transportation-report-by-mr-aldrich-from-the-committee-on-finance-march-3-1893-r-yard-nitric-acid-per-pound-olive-oil-per-pint-opium-powdered-per-pound-pills-compoujid-cathartic-per-hundred-potassium-bromide-of-per-ounce-potassium-chlorate-of-jowdered-per-ounce-potassium-iolide-of-per-ounce-quinine-sulphate-of-per-ounce-khubarb-powdered-per-ounce-squills-syrup-of-per-pound-sulphurwashed-jier-pound-sulphuric-acid-per-pound-tartai-emetic-per-ounce-turpentine-spirits-of-per-quart-zinc-sulphate-of-per-ounce-1863-jan-007i1012-image371963843.html
. Wholesale prices, wages, and transportation. Report by Mr. Aldrich, from the Committee on Finance, March 3, 1893 . r yard Nitric acid, per pound Olive oil, per pint Opium, powdered, per pound Pills, compoujid cathartic, per hundred Potassium, bromide of per ounce Potassium, chlorate of, j)owdered, per ounce Potassium, io^lide of per ounce Quinine, sulphate of per ounce Khubarb, powdered, per ounce Squills, syrup of, per pound Sulphur,washed, jier pound Sulphuric acid, per pound... Tartai- emetic, per ounce Turpentine, spirits of per quart Zinc, sulphate of, per ounce 1863. Jan. $0.07i.10.12. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/wholesale-prices-wages-and-transportation-report-by-mr-aldrich-from-the-committee-on-finance-march-3-1893-r-yard-nitric-acid-per-pound-olive-oil-per-pint-opium-powdered-per-pound-pills-compoujid-cathartic-per-hundred-potassium-bromide-of-per-ounce-potassium-chlorate-of-jowdered-per-ounce-potassium-iolide-of-per-ounce-quinine-sulphate-of-per-ounce-khubarb-powdered-per-ounce-squills-syrup-of-per-pound-sulphurwashed-jier-pound-sulphuric-acid-per-pound-tartai-emetic-per-ounce-turpentine-spirits-of-per-quart-zinc-sulphate-of-per-ounce-1863-jan-007i1012-image371963843.htmlRM2CH4BJY–. Wholesale prices, wages, and transportation. Report by Mr. Aldrich, from the Committee on Finance, March 3, 1893 . r yard Nitric acid, per pound Olive oil, per pint Opium, powdered, per pound Pills, compoujid cathartic, per hundred Potassium, bromide of per ounce Potassium, chlorate of, j)owdered, per ounce Potassium, io^lide of per ounce Quinine, sulphate of per ounce Khubarb, powdered, per ounce Squills, syrup of, per pound Sulphur,washed, jier pound Sulphuric acid, per pound... Tartai- emetic, per ounce Turpentine, spirits of per quart Zinc, sulphate of, per ounce 1863. Jan. $0.07i.10.12.
 . Pathogenic microörganisms; a practical manual for students, physicians, and health officers . 300 1 to 6000 Mercuric iodide 1 to 40,000 1 to9 Potassium bromide 1 to 10 1 to 143 Potassium iodide . 1 to 10 1 to 25 Potassium permanganate 1 to 300 1 to 1000 Pure formaldehyde 1 to 25,000 1 to 333 Quinine sulphate . 1 to 800 1 to 107 Silver nitrate .... 1 to 12,500 1 to 2000 Sodium borate 1 tol4 1 to 200 Sodium chloride . 1 to6 1 to 10,000 Zinc chloride .... 1 to 500 1 to 20,000 Zinc sulphate .... 1 to 20 1 These figures are approximately correct, and represent the percentage of disinfectionrequi Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/pathogenic-microorganisms-a-practical-manual-for-students-physicians-and-health-officers-300-1-to-6000-mercuric-iodide-1-to-40000-1-to9-potassium-bromide-1-to-10-1-to-143-potassium-iodide-1-to-10-1-to-25-potassium-permanganate-1-to-300-1-to-1000-pure-formaldehyde-1-to-25000-1-to-333-quinine-sulphate-1-to-800-1-to-107-silver-nitrate-1-to-12500-1-to-2000-sodium-borate-1-tol4-1-to-200-sodium-chloride-1-to6-1-to-10000-zinc-chloride-1-to-500-1-to-20000-zinc-sulphate-1-to-20-1-these-figures-are-approximately-correct-and-represent-the-percentage-of-disinfectionrequi-image370122898.html
. Pathogenic microörganisms; a practical manual for students, physicians, and health officers . 300 1 to 6000 Mercuric iodide 1 to 40,000 1 to9 Potassium bromide 1 to 10 1 to 143 Potassium iodide . 1 to 10 1 to 25 Potassium permanganate 1 to 300 1 to 1000 Pure formaldehyde 1 to 25,000 1 to 333 Quinine sulphate . 1 to 800 1 to 107 Silver nitrate .... 1 to 12,500 1 to 2000 Sodium borate 1 tol4 1 to 200 Sodium chloride . 1 to6 1 to 10,000 Zinc chloride .... 1 to 500 1 to 20,000 Zinc sulphate .... 1 to 20 1 These figures are approximately correct, and represent the percentage of disinfectionrequi Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/pathogenic-microorganisms-a-practical-manual-for-students-physicians-and-health-officers-300-1-to-6000-mercuric-iodide-1-to-40000-1-to9-potassium-bromide-1-to-10-1-to-143-potassium-iodide-1-to-10-1-to-25-potassium-permanganate-1-to-300-1-to-1000-pure-formaldehyde-1-to-25000-1-to-333-quinine-sulphate-1-to-800-1-to-107-silver-nitrate-1-to-12500-1-to-2000-sodium-borate-1-tol4-1-to-200-sodium-chloride-1-to6-1-to-10000-zinc-chloride-1-to-500-1-to-20000-zinc-sulphate-1-to-20-1-these-figures-are-approximately-correct-and-represent-the-percentage-of-disinfectionrequi-image370122898.htmlRM2CE4FEX–. Pathogenic microörganisms; a practical manual for students, physicians, and health officers . 300 1 to 6000 Mercuric iodide 1 to 40,000 1 to9 Potassium bromide 1 to 10 1 to 143 Potassium iodide . 1 to 10 1 to 25 Potassium permanganate 1 to 300 1 to 1000 Pure formaldehyde 1 to 25,000 1 to 333 Quinine sulphate . 1 to 800 1 to 107 Silver nitrate .... 1 to 12,500 1 to 2000 Sodium borate 1 tol4 1 to 200 Sodium chloride . 1 to6 1 to 10,000 Zinc chloride .... 1 to 500 1 to 20,000 Zinc sulphate .... 1 to 20 1 These figures are approximately correct, and represent the percentage of disinfectionrequi
 . Carnegie Institution of Washington publication. ELECTRICAL CONDUCTIVITIES, ETC. 21 Potassium Bromide (W. and C). Molecular Conductivity. HvO° 8 16 32 128 512 1024 2048 65.82 68.01 70.10 71.84 74.79 75.73 79.23 jM4.5° 93.21 98.45 101.7 104.6 109.0 111.3 115.6 M,25c 114.4 121.3 125.2 128.8 134.5 137.6 143.5 M»35° Mi.500 /xp65° 136.2 145.6 150.5 154.6 162.0 165.5 172.6 165.1 181.4 195.3 206.0 211.6 213.6 216.7 196.7 218.1 236.5 250.0 256.6 260.3 263.7 Percentage Dissociation. v a0° al4.5° a25° 16 32 128 512 1024 2048 83.1 85.8 88.5 90.8 94.4 95.6 100.0 80.6 85.2 88.0 90.5 94.3 96.3 100.0 79.7 8 Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/carnegie-institution-of-washington-publication-electrical-conductivities-etc-21-potassium-bromide-w-and-c-molecular-conductivity-hvo-8-16-32-128-512-1024-2048-6582-6801-7010-7184-7479-7573-7923-jm45-9321-9845-1017-1046-1090-1113-1156-m25c-1144-1213-1252-1288-1345-1376-1435-m35-mi500-xp65-1362-1456-1505-1546-1620-1655-1726-1651-1814-1953-2060-2116-2136-2167-1967-2181-2365-2500-2566-2603-2637-percentage-dissociation-v-a0-al45-a25-16-32-128-512-1024-2048-831-858-885-908-944-956-1000-806-852-880-905-943-963-1000-797-8-image233486181.html
. Carnegie Institution of Washington publication. ELECTRICAL CONDUCTIVITIES, ETC. 21 Potassium Bromide (W. and C). Molecular Conductivity. HvO° 8 16 32 128 512 1024 2048 65.82 68.01 70.10 71.84 74.79 75.73 79.23 jM4.5° 93.21 98.45 101.7 104.6 109.0 111.3 115.6 M,25c 114.4 121.3 125.2 128.8 134.5 137.6 143.5 M»35° Mi.500 /xp65° 136.2 145.6 150.5 154.6 162.0 165.5 172.6 165.1 181.4 195.3 206.0 211.6 213.6 216.7 196.7 218.1 236.5 250.0 256.6 260.3 263.7 Percentage Dissociation. v a0° al4.5° a25° 16 32 128 512 1024 2048 83.1 85.8 88.5 90.8 94.4 95.6 100.0 80.6 85.2 88.0 90.5 94.3 96.3 100.0 79.7 8 Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/carnegie-institution-of-washington-publication-electrical-conductivities-etc-21-potassium-bromide-w-and-c-molecular-conductivity-hvo-8-16-32-128-512-1024-2048-6582-6801-7010-7184-7479-7573-7923-jm45-9321-9845-1017-1046-1090-1113-1156-m25c-1144-1213-1252-1288-1345-1376-1435-m35-mi500-xp65-1362-1456-1505-1546-1620-1655-1726-1651-1814-1953-2060-2116-2136-2167-1967-2181-2365-2500-2566-2603-2637-percentage-dissociation-v-a0-al45-a25-16-32-128-512-1024-2048-831-858-885-908-944-956-1000-806-852-880-905-943-963-1000-797-8-image233486181.htmlRMRFT605–. Carnegie Institution of Washington publication. ELECTRICAL CONDUCTIVITIES, ETC. 21 Potassium Bromide (W. and C). Molecular Conductivity. HvO° 8 16 32 128 512 1024 2048 65.82 68.01 70.10 71.84 74.79 75.73 79.23 jM4.5° 93.21 98.45 101.7 104.6 109.0 111.3 115.6 M,25c 114.4 121.3 125.2 128.8 134.5 137.6 143.5 M»35° Mi.500 /xp65° 136.2 145.6 150.5 154.6 162.0 165.5 172.6 165.1 181.4 195.3 206.0 211.6 213.6 216.7 196.7 218.1 236.5 250.0 256.6 260.3 263.7 Percentage Dissociation. v a0° al4.5° a25° 16 32 128 512 1024 2048 83.1 85.8 88.5 90.8 94.4 95.6 100.0 80.6 85.2 88.0 90.5 94.3 96.3 100.0 79.7 8
 The fireside university of modern invention, discovery, industry and art for home circle study and entertainment . and Pennsylvania by the hundreds of thousandsof pounds. Its chief use is in medicine as Bromide of Potassium.The Bromides are taken at the drug-stores by people who feelnervous, and with Chloral, have done much to destroy health through unscientific use. Phy-sicians should always be consulted. Is Fluorine abundant ? It is widely diffused, but in smallquantities. It exists in sea-water,always in teeth and bones—more infossil bones than those of presentformation. It will corrode any Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-fireside-university-of-modern-invention-discovery-industry-and-art-for-home-circle-study-and-entertainment-and-pennsylvania-by-the-hundreds-of-thousandsof-pounds-its-chief-use-is-in-medicine-as-bromide-of-potassiumthe-bromides-are-taken-at-the-drug-stores-by-people-who-feelnervous-and-with-chloral-have-done-much-to-destroy-health-through-unscientific-use-phy-sicians-should-always-be-consulted-is-fluorine-abundant-it-is-widely-diffused-but-in-smallquantities-it-exists-in-sea-wateralways-in-teeth-and-bonesmore-infossil-bones-than-those-of-presentformation-it-will-corrode-any-image342720595.html
The fireside university of modern invention, discovery, industry and art for home circle study and entertainment . and Pennsylvania by the hundreds of thousandsof pounds. Its chief use is in medicine as Bromide of Potassium.The Bromides are taken at the drug-stores by people who feelnervous, and with Chloral, have done much to destroy health through unscientific use. Phy-sicians should always be consulted. Is Fluorine abundant ? It is widely diffused, but in smallquantities. It exists in sea-water,always in teeth and bones—more infossil bones than those of presentformation. It will corrode any Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-fireside-university-of-modern-invention-discovery-industry-and-art-for-home-circle-study-and-entertainment-and-pennsylvania-by-the-hundreds-of-thousandsof-pounds-its-chief-use-is-in-medicine-as-bromide-of-potassiumthe-bromides-are-taken-at-the-drug-stores-by-people-who-feelnervous-and-with-chloral-have-done-much-to-destroy-health-through-unscientific-use-phy-sicians-should-always-be-consulted-is-fluorine-abundant-it-is-widely-diffused-but-in-smallquantities-it-exists-in-sea-wateralways-in-teeth-and-bonesmore-infossil-bones-than-those-of-presentformation-it-will-corrode-any-image342720595.htmlRM2AWG7H7–The fireside university of modern invention, discovery, industry and art for home circle study and entertainment . and Pennsylvania by the hundreds of thousandsof pounds. Its chief use is in medicine as Bromide of Potassium.The Bromides are taken at the drug-stores by people who feelnervous, and with Chloral, have done much to destroy health through unscientific use. Phy-sicians should always be consulted. Is Fluorine abundant ? It is widely diffused, but in smallquantities. It exists in sea-water,always in teeth and bones—more infossil bones than those of presentformation. It will corrode any
 Gaillard's medical journal . f ammonia, or iodine have proven to be the best remedies in hypertrophiccatarrh ; and in atrophic and foetid catarrh, listerine, bromide of potassium,nitrate of silver, and the pinus canadensis and glycerine, with oil of euca-lyptus. The dose of any of these prescriptions is gtt. iij-x-xv, with to 3j of vase-line, and as the medicines approach the end of the spray-producer the fingershould be placed over the point in order to make the contents regurgitate, thusmixing them thoroughly and subdividing them into a molecular state. If the catarrhal inflammation has exte Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/gaillards-medical-journal-f-ammonia-or-iodine-have-proven-to-be-the-best-remedies-in-hypertrophiccatarrh-and-in-atrophic-and-foetid-catarrh-listerine-bromide-of-potassiumnitrate-of-silver-and-the-pinus-canadensis-and-glycerine-with-oil-of-euca-lyptus-the-dose-of-any-of-these-prescriptions-is-gtt-iij-x-xv-with-to-3j-of-vase-line-and-as-the-medicines-approach-the-end-of-the-spray-producer-the-fingershould-be-placed-over-the-point-in-order-to-make-the-contents-regurgitate-thusmixing-them-thoroughly-and-subdividing-them-into-a-molecular-state-if-the-catarrhal-inflammation-has-exte-image339995966.html
Gaillard's medical journal . f ammonia, or iodine have proven to be the best remedies in hypertrophiccatarrh ; and in atrophic and foetid catarrh, listerine, bromide of potassium,nitrate of silver, and the pinus canadensis and glycerine, with oil of euca-lyptus. The dose of any of these prescriptions is gtt. iij-x-xv, with to 3j of vase-line, and as the medicines approach the end of the spray-producer the fingershould be placed over the point in order to make the contents regurgitate, thusmixing them thoroughly and subdividing them into a molecular state. If the catarrhal inflammation has exte Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/gaillards-medical-journal-f-ammonia-or-iodine-have-proven-to-be-the-best-remedies-in-hypertrophiccatarrh-and-in-atrophic-and-foetid-catarrh-listerine-bromide-of-potassiumnitrate-of-silver-and-the-pinus-canadensis-and-glycerine-with-oil-of-euca-lyptus-the-dose-of-any-of-these-prescriptions-is-gtt-iij-x-xv-with-to-3j-of-vase-line-and-as-the-medicines-approach-the-end-of-the-spray-producer-the-fingershould-be-placed-over-the-point-in-order-to-make-the-contents-regurgitate-thusmixing-them-thoroughly-and-subdividing-them-into-a-molecular-state-if-the-catarrhal-inflammation-has-exte-image339995966.htmlRM2AN4492–Gaillard's medical journal . f ammonia, or iodine have proven to be the best remedies in hypertrophiccatarrh ; and in atrophic and foetid catarrh, listerine, bromide of potassium,nitrate of silver, and the pinus canadensis and glycerine, with oil of euca-lyptus. The dose of any of these prescriptions is gtt. iij-x-xv, with to 3j of vase-line, and as the medicines approach the end of the spray-producer the fingershould be placed over the point in order to make the contents regurgitate, thusmixing them thoroughly and subdividing them into a molecular state. If the catarrhal inflammation has exte
 . Diseases of the dog and their treatment. animals that are fat or have short necks; insert theneedle beneath the larynx or through the crico-thyroid ligament, the solu-tions to be used are 1 per cent, solution of morphine or codeine. Theseinjections should be made daily or every other day. Frequently in chronic catarrh we use narcotics to stop the severecough produced by irritation of the membrane—morphine, codeine,heroin, and in rare cases bromide of potassium or chloral hydrate.Expectorants are not of much use in the dog. As to other affections of the larynx, with the exception of tuberculo Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/diseases-of-the-dog-and-their-treatment-animals-that-are-fat-or-have-short-necks-insert-theneedle-beneath-the-larynx-or-through-the-crico-thyroid-ligament-the-solu-tions-to-be-used-are-1-per-cent-solution-of-morphine-or-codeine-theseinjections-should-be-made-daily-or-every-other-day-frequently-in-chronic-catarrh-we-use-narcotics-to-stop-the-severecough-produced-by-irritation-of-the-membranemorphine-codeineheroin-and-in-rare-cases-bromide-of-potassium-or-chloral-hydrateexpectorants-are-not-of-much-use-in-the-dog-as-to-other-affections-of-the-larynx-with-the-exception-of-tuberculo-image336690066.html
. Diseases of the dog and their treatment. animals that are fat or have short necks; insert theneedle beneath the larynx or through the crico-thyroid ligament, the solu-tions to be used are 1 per cent, solution of morphine or codeine. Theseinjections should be made daily or every other day. Frequently in chronic catarrh we use narcotics to stop the severecough produced by irritation of the membrane—morphine, codeine,heroin, and in rare cases bromide of potassium or chloral hydrate.Expectorants are not of much use in the dog. As to other affections of the larynx, with the exception of tuberculo Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/diseases-of-the-dog-and-their-treatment-animals-that-are-fat-or-have-short-necks-insert-theneedle-beneath-the-larynx-or-through-the-crico-thyroid-ligament-the-solu-tions-to-be-used-are-1-per-cent-solution-of-morphine-or-codeine-theseinjections-should-be-made-daily-or-every-other-day-frequently-in-chronic-catarrh-we-use-narcotics-to-stop-the-severecough-produced-by-irritation-of-the-membranemorphine-codeineheroin-and-in-rare-cases-bromide-of-potassium-or-chloral-hydrateexpectorants-are-not-of-much-use-in-the-dog-as-to-other-affections-of-the-larynx-with-the-exception-of-tuberculo-image336690066.htmlRM2AFNFH6–. Diseases of the dog and their treatment. animals that are fat or have short necks; insert theneedle beneath the larynx or through the crico-thyroid ligament, the solu-tions to be used are 1 per cent, solution of morphine or codeine. Theseinjections should be made daily or every other day. Frequently in chronic catarrh we use narcotics to stop the severecough produced by irritation of the membrane—morphine, codeine,heroin, and in rare cases bromide of potassium or chloral hydrate.Expectorants are not of much use in the dog. As to other affections of the larynx, with the exception of tuberculo
 Principles and practice of operative dentistry . d then,and if the bowels are overactive, a grain of Dovers powder at bedtime isoften of great service. If there is vomiting and flatulence, some carmina-tive will be necessary. In cases where there is much restlessness and dis-turbance of the nervous system, bromide and iodide of potassium with salvolatile will often abate sickness and relieve head-symptoms if present. Local Treatment.—In those cases in which the gums are considerablycongested and swollen, causing pain on pressure, and accompanied with gen-eral febrile symptoms, vomiting, diarrh Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/principles-and-practice-of-operative-dentistry-d-thenand-if-the-bowels-are-overactive-a-grain-of-dovers-powder-at-bedtime-isoften-of-great-service-if-there-is-vomiting-and-flatulence-some-carmina-tive-will-be-necessary-in-cases-where-there-is-much-restlessness-and-dis-turbance-of-the-nervous-system-bromide-and-iodide-of-potassium-with-salvolatile-will-often-abate-sickness-and-relieve-head-symptoms-if-present-local-treatmentin-those-cases-in-which-the-gums-are-considerablycongested-and-swollen-causing-pain-on-pressure-and-accompanied-with-gen-eral-febrile-symptoms-vomiting-diarrh-image340231134.html
Principles and practice of operative dentistry . d then,and if the bowels are overactive, a grain of Dovers powder at bedtime isoften of great service. If there is vomiting and flatulence, some carmina-tive will be necessary. In cases where there is much restlessness and dis-turbance of the nervous system, bromide and iodide of potassium with salvolatile will often abate sickness and relieve head-symptoms if present. Local Treatment.—In those cases in which the gums are considerablycongested and swollen, causing pain on pressure, and accompanied with gen-eral febrile symptoms, vomiting, diarrh Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/principles-and-practice-of-operative-dentistry-d-thenand-if-the-bowels-are-overactive-a-grain-of-dovers-powder-at-bedtime-isoften-of-great-service-if-there-is-vomiting-and-flatulence-some-carmina-tive-will-be-necessary-in-cases-where-there-is-much-restlessness-and-dis-turbance-of-the-nervous-system-bromide-and-iodide-of-potassium-with-salvolatile-will-often-abate-sickness-and-relieve-head-symptoms-if-present-local-treatmentin-those-cases-in-which-the-gums-are-considerablycongested-and-swollen-causing-pain-on-pressure-and-accompanied-with-gen-eral-febrile-symptoms-vomiting-diarrh-image340231134.htmlRM2ANET7X–Principles and practice of operative dentistry . d then,and if the bowels are overactive, a grain of Dovers powder at bedtime isoften of great service. If there is vomiting and flatulence, some carmina-tive will be necessary. In cases where there is much restlessness and dis-turbance of the nervous system, bromide and iodide of potassium with salvolatile will often abate sickness and relieve head-symptoms if present. Local Treatment.—In those cases in which the gums are considerablycongested and swollen, causing pain on pressure, and accompanied with gen-eral febrile symptoms, vomiting, diarrh
 A text-book of practical therapeutics . aceconcerning its effects may be taken as representing the whole classof bromides, except in the instances where slight differences exist,which will be pointed out under the various names of the respective 158 DRUGS salts. When taken internally in full doses it produces drowsiness,sleep, and a decrease in all the reflexes. (See Effects of ProlongedUse.) Because of its potash base it is more depressant than the othersalts, and should be used with care in nephritis. Nervous System.—The bromide of potassium acts as a distinctdepressant to the motor and to t Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/a-text-book-of-practical-therapeutics-aceconcerning-its-effects-may-be-taken-as-representing-the-whole-classof-bromides-except-in-the-instances-where-slight-differences-existwhich-will-be-pointed-out-under-the-various-names-of-the-respective-158-drugs-salts-when-taken-internally-in-full-doses-it-produces-drowsinesssleep-and-a-decrease-in-all-the-reflexes-see-effects-of-prolongeduse-because-of-its-potash-base-it-is-more-depressant-than-the-othersalts-and-should-be-used-with-care-in-nephritis-nervous-systemthe-bromide-of-potassium-acts-as-a-distinctdepressant-to-the-motor-and-to-t-image340213483.html
A text-book of practical therapeutics . aceconcerning its effects may be taken as representing the whole classof bromides, except in the instances where slight differences exist,which will be pointed out under the various names of the respective 158 DRUGS salts. When taken internally in full doses it produces drowsiness,sleep, and a decrease in all the reflexes. (See Effects of ProlongedUse.) Because of its potash base it is more depressant than the othersalts, and should be used with care in nephritis. Nervous System.—The bromide of potassium acts as a distinctdepressant to the motor and to t Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/a-text-book-of-practical-therapeutics-aceconcerning-its-effects-may-be-taken-as-representing-the-whole-classof-bromides-except-in-the-instances-where-slight-differences-existwhich-will-be-pointed-out-under-the-various-names-of-the-respective-158-drugs-salts-when-taken-internally-in-full-doses-it-produces-drowsinesssleep-and-a-decrease-in-all-the-reflexes-see-effects-of-prolongeduse-because-of-its-potash-base-it-is-more-depressant-than-the-othersalts-and-should-be-used-with-care-in-nephritis-nervous-systemthe-bromide-of-potassium-acts-as-a-distinctdepressant-to-the-motor-and-to-t-image340213483.htmlRM2ANE1NF–A text-book of practical therapeutics . aceconcerning its effects may be taken as representing the whole classof bromides, except in the instances where slight differences exist,which will be pointed out under the various names of the respective 158 DRUGS salts. When taken internally in full doses it produces drowsiness,sleep, and a decrease in all the reflexes. (See Effects of ProlongedUse.) Because of its potash base it is more depressant than the othersalts, and should be used with care in nephritis. Nervous System.—The bromide of potassium acts as a distinctdepressant to the motor and to t
 The American annual of photography . r bromide, suchas hypo, ammonia or an excess of sulphite or carbonate mustbe present, when under certain conditions the dissolved silveris reduced to metallic silver in a very fine state of division, par-ticularly in the shadow portions of the negative, where nobromide is liberated during development. Fine grained emulsions in which the grains of silver halideare very small and therefore more readily soluble are most sus-ceptible to this form of fog, especially if the development isforced, but the fog may be prevented by the addition of alittle potassium io Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-american-annual-of-photography-r-bromide-suchas-hypo-ammonia-or-an-excess-of-sulphite-or-carbonate-mustbe-present-when-under-certain-conditions-the-dissolved-silveris-reduced-to-metallic-silver-in-a-very-fine-state-of-division-par-ticularly-in-the-shadow-portions-of-the-negative-where-nobromide-is-liberated-during-development-fine-grained-emulsions-in-which-the-grains-of-silver-halideare-very-small-and-therefore-more-readily-soluble-are-most-sus-ceptible-to-this-form-of-fog-especially-if-the-development-isforced-but-the-fog-may-be-prevented-by-the-addition-of-alittle-potassium-io-image339159957.html
The American annual of photography . r bromide, suchas hypo, ammonia or an excess of sulphite or carbonate mustbe present, when under certain conditions the dissolved silveris reduced to metallic silver in a very fine state of division, par-ticularly in the shadow portions of the negative, where nobromide is liberated during development. Fine grained emulsions in which the grains of silver halideare very small and therefore more readily soluble are most sus-ceptible to this form of fog, especially if the development isforced, but the fog may be prevented by the addition of alittle potassium io Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-american-annual-of-photography-r-bromide-suchas-hypo-ammonia-or-an-excess-of-sulphite-or-carbonate-mustbe-present-when-under-certain-conditions-the-dissolved-silveris-reduced-to-metallic-silver-in-a-very-fine-state-of-division-par-ticularly-in-the-shadow-portions-of-the-negative-where-nobromide-is-liberated-during-development-fine-grained-emulsions-in-which-the-grains-of-silver-halideare-very-small-and-therefore-more-readily-soluble-are-most-sus-ceptible-to-this-form-of-fog-especially-if-the-development-isforced-but-the-fog-may-be-prevented-by-the-addition-of-alittle-potassium-io-image339159957.htmlRM2AKP1YH–The American annual of photography . r bromide, suchas hypo, ammonia or an excess of sulphite or carbonate mustbe present, when under certain conditions the dissolved silveris reduced to metallic silver in a very fine state of division, par-ticularly in the shadow portions of the negative, where nobromide is liberated during development. Fine grained emulsions in which the grains of silver halideare very small and therefore more readily soluble are most sus-ceptible to this form of fog, especially if the development isforced, but the fog may be prevented by the addition of alittle potassium io
 A supplement to Ures Dictionary of Arts, Manufactures, and Mines, : containing a clear exposition of their principles and practice. . me sheets of very superiorglazed post, and wash it on one side only with bromide of potassium—iO grains to 1 ounceof distilled water, over which, when dry, pass a solution of 100 grains of nitrate of silverin the same quantity of water. The paper must be dried as quickly as possible without es-posin it to too much heat; then again washed with the silver solution, and diied in thedark.° Such are the preparations of an ordinary kind, with which the photographer wi Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/a-supplement-to-ures-dictionary-of-arts-manufactures-and-mines-containing-a-clear-exposition-of-their-principles-and-practice-me-sheets-of-very-superiorglazed-post-and-wash-it-on-one-side-only-with-bromide-of-potassiumio-grains-to-1-ounceof-distilled-water-over-which-when-dry-pass-a-solution-of-100-grains-of-nitrate-of-silverin-the-same-quantity-of-water-the-paper-must-be-dried-as-quickly-as-possible-without-es-posin-it-to-too-much-heat-then-again-washed-with-the-silver-solution-and-diied-in-thedark-such-are-the-preparations-of-an-ordinary-kind-with-which-the-photographer-wi-image339456556.html
A supplement to Ures Dictionary of Arts, Manufactures, and Mines, : containing a clear exposition of their principles and practice. . me sheets of very superiorglazed post, and wash it on one side only with bromide of potassium—iO grains to 1 ounceof distilled water, over which, when dry, pass a solution of 100 grains of nitrate of silverin the same quantity of water. The paper must be dried as quickly as possible without es-posin it to too much heat; then again washed with the silver solution, and diied in thedark.° Such are the preparations of an ordinary kind, with which the photographer wi Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/a-supplement-to-ures-dictionary-of-arts-manufactures-and-mines-containing-a-clear-exposition-of-their-principles-and-practice-me-sheets-of-very-superiorglazed-post-and-wash-it-on-one-side-only-with-bromide-of-potassiumio-grains-to-1-ounceof-distilled-water-over-which-when-dry-pass-a-solution-of-100-grains-of-nitrate-of-silverin-the-same-quantity-of-water-the-paper-must-be-dried-as-quickly-as-possible-without-es-posin-it-to-too-much-heat-then-again-washed-with-the-silver-solution-and-diied-in-thedark-such-are-the-preparations-of-an-ordinary-kind-with-which-the-photographer-wi-image339456556.htmlRM2AM7G8C–A supplement to Ures Dictionary of Arts, Manufactures, and Mines, : containing a clear exposition of their principles and practice. . me sheets of very superiorglazed post, and wash it on one side only with bromide of potassium—iO grains to 1 ounceof distilled water, over which, when dry, pass a solution of 100 grains of nitrate of silverin the same quantity of water. The paper must be dried as quickly as possible without es-posin it to too much heat; then again washed with the silver solution, and diied in thedark.° Such are the preparations of an ordinary kind, with which the photographer wi
 Surgery; its theory and practice . ling the inflammation. It may be given by the mouth,or in the form of morphia as a subcutaneous injection. Quinine,salicylic acid, and antipyrin are sometimes employed when thetemperature is high, as is colchicum in gout, potash and salicylateof soda in rheumatism, perchloride of iron in erysipelas, andhyoscyamus, bromide of potassium, sulphonal, and chloral whenthere is want of sleep. Bleeding is not often employed in modern surgery, but it is attimes beneficial in very acute inflammations in young and pleth-oric subjects. Of lateFG- 2- bleeding has again be Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/surgery-its-theory-and-practice-ling-the-inflammation-it-may-be-given-by-the-mouthor-in-the-form-of-morphia-as-a-subcutaneous-injection-quininesalicylic-acid-and-antipyrin-are-sometimes-employed-when-thetemperature-is-high-as-is-colchicum-in-gout-potash-and-salicylateof-soda-in-rheumatism-perchloride-of-iron-in-erysipelas-andhyoscyamus-bromide-of-potassium-sulphonal-and-chloral-whenthere-is-want-of-sleep-bleeding-is-not-often-employed-in-modern-surgery-but-it-is-attimes-beneficial-in-very-acute-inflammations-in-young-and-pleth-oric-subjects-of-latefg-2-bleeding-has-again-be-image343141608.html
Surgery; its theory and practice . ling the inflammation. It may be given by the mouth,or in the form of morphia as a subcutaneous injection. Quinine,salicylic acid, and antipyrin are sometimes employed when thetemperature is high, as is colchicum in gout, potash and salicylateof soda in rheumatism, perchloride of iron in erysipelas, andhyoscyamus, bromide of potassium, sulphonal, and chloral whenthere is want of sleep. Bleeding is not often employed in modern surgery, but it is attimes beneficial in very acute inflammations in young and pleth-oric subjects. Of lateFG- 2- bleeding has again be Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/surgery-its-theory-and-practice-ling-the-inflammation-it-may-be-given-by-the-mouthor-in-the-form-of-morphia-as-a-subcutaneous-injection-quininesalicylic-acid-and-antipyrin-are-sometimes-employed-when-thetemperature-is-high-as-is-colchicum-in-gout-potash-and-salicylateof-soda-in-rheumatism-perchloride-of-iron-in-erysipelas-andhyoscyamus-bromide-of-potassium-sulphonal-and-chloral-whenthere-is-want-of-sleep-bleeding-is-not-often-employed-in-modern-surgery-but-it-is-attimes-beneficial-in-very-acute-inflammations-in-young-and-pleth-oric-subjects-of-latefg-2-bleeding-has-again-be-image343141608.htmlRM2AX7CHC–Surgery; its theory and practice . ling the inflammation. It may be given by the mouth,or in the form of morphia as a subcutaneous injection. Quinine,salicylic acid, and antipyrin are sometimes employed when thetemperature is high, as is colchicum in gout, potash and salicylateof soda in rheumatism, perchloride of iron in erysipelas, andhyoscyamus, bromide of potassium, sulphonal, and chloral whenthere is want of sleep. Bleeding is not often employed in modern surgery, but it is attimes beneficial in very acute inflammations in young and pleth-oric subjects. Of lateFG- 2- bleeding has again be
 A study of the conductivity of certain salts in water, methyl, ethyl, and propyl alcohols, and mixtures of these solvents . on- potassium iodide, strontiumiodide, ammonium bromide, cadmium bromide, ferric chloride,and lithium nitrate. Apparatus.In all these measurements the Kohlrausch method of meas--uring conductivitv v/as employed. The bridge wire used was ametre in length and of manganin. The resistance coils weremanufactured by Leeds and Co., Philadelphia, and were guar--anteed accurate to l/25 of 1 per cent. The cells used were all of the following form. The diff*erence betv/een them and Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/a-study-of-the-conductivity-of-certain-salts-in-water-methyl-ethyl-and-propyl-alcohols-and-mixtures-of-these-solvents-on-potassium-iodide-strontiumiodide-ammonium-bromide-cadmium-bromide-ferric-chlorideand-lithium-nitrate-apparatusin-all-these-measurements-the-kohlrausch-method-of-meas-uring-conductivitv-vas-employed-the-bridge-wire-used-was-ametre-in-length-and-of-manganin-the-resistance-coils-weremanufactured-by-leeds-and-co-philadelphia-and-were-guar-anteed-accurate-to-l25-of-1-per-cent-the-cells-used-were-all-of-the-following-form-the-difference-betveen-them-and-image340116329.html
A study of the conductivity of certain salts in water, methyl, ethyl, and propyl alcohols, and mixtures of these solvents . on- potassium iodide, strontiumiodide, ammonium bromide, cadmium bromide, ferric chloride,and lithium nitrate. Apparatus.In all these measurements the Kohlrausch method of meas--uring conductivitv v/as employed. The bridge wire used was ametre in length and of manganin. The resistance coils weremanufactured by Leeds and Co., Philadelphia, and were guar--anteed accurate to l/25 of 1 per cent. The cells used were all of the following form. The diff*erence betv/een them and Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/a-study-of-the-conductivity-of-certain-salts-in-water-methyl-ethyl-and-propyl-alcohols-and-mixtures-of-these-solvents-on-potassium-iodide-strontiumiodide-ammonium-bromide-cadmium-bromide-ferric-chlorideand-lithium-nitrate-apparatusin-all-these-measurements-the-kohlrausch-method-of-meas-uring-conductivitv-vas-employed-the-bridge-wire-used-was-ametre-in-length-and-of-manganin-the-resistance-coils-weremanufactured-by-leeds-and-co-philadelphia-and-were-guar-anteed-accurate-to-l25-of-1-per-cent-the-cells-used-were-all-of-the-following-form-the-difference-betveen-them-and-image340116329.htmlRM2AN9HRN–A study of the conductivity of certain salts in water, methyl, ethyl, and propyl alcohols, and mixtures of these solvents . on- potassium iodide, strontiumiodide, ammonium bromide, cadmium bromide, ferric chloride,and lithium nitrate. Apparatus.In all these measurements the Kohlrausch method of meas--uring conductivitv v/as employed. The bridge wire used was ametre in length and of manganin. The resistance coils weremanufactured by Leeds and Co., Philadelphia, and were guar--anteed accurate to l/25 of 1 per cent. The cells used were all of the following form. The diff*erence betv/een them and
 Brain surgery . ongest interval during the yearwas nine weeks. He had three fits in March, 1892. Hewas very dull mentally, and had been treated by verylarge doses of bromide of potassium, which diminished thefrequency of but did not arrest the fits. Operation by trephining was performed by Dr. McBur-ney on the 2d of April, 1892. The skull was opened atthe point of fracture over the arm centre on the right side(Fig. 14). The external table was found to be fracturedbut the internal table appeared to be uninjured, but a smallsplinter of bone was found indenting the dura. Thedura was very much thi Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/brain-surgery-ongest-interval-during-the-yearwas-nine-weeks-he-had-three-fits-in-march-1892-hewas-very-dull-mentally-and-had-been-treated-by-verylarge-doses-of-bromide-of-potassium-which-diminished-thefrequency-of-but-did-not-arrest-the-fits-operation-by-trephining-was-performed-by-dr-mcbur-ney-on-the-2d-of-april-1892-the-skull-was-opened-atthe-point-of-fracture-over-the-arm-centre-on-the-right-sidefig-14-the-external-table-was-found-to-be-fracturedbut-the-internal-table-appeared-to-be-uninjured-but-a-smallsplinter-of-bone-was-found-indenting-the-dura-thedura-was-very-much-thi-image340035820.html
Brain surgery . ongest interval during the yearwas nine weeks. He had three fits in March, 1892. Hewas very dull mentally, and had been treated by verylarge doses of bromide of potassium, which diminished thefrequency of but did not arrest the fits. Operation by trephining was performed by Dr. McBur-ney on the 2d of April, 1892. The skull was opened atthe point of fracture over the arm centre on the right side(Fig. 14). The external table was found to be fracturedbut the internal table appeared to be uninjured, but a smallsplinter of bone was found indenting the dura. Thedura was very much thi Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/brain-surgery-ongest-interval-during-the-yearwas-nine-weeks-he-had-three-fits-in-march-1892-hewas-very-dull-mentally-and-had-been-treated-by-verylarge-doses-of-bromide-of-potassium-which-diminished-thefrequency-of-but-did-not-arrest-the-fits-operation-by-trephining-was-performed-by-dr-mcbur-ney-on-the-2d-of-april-1892-the-skull-was-opened-atthe-point-of-fracture-over-the-arm-centre-on-the-right-sidefig-14-the-external-table-was-found-to-be-fracturedbut-the-internal-table-appeared-to-be-uninjured-but-a-smallsplinter-of-bone-was-found-indenting-the-dura-thedura-was-very-much-thi-image340035820.htmlRM2AN5Y4C–Brain surgery . ongest interval during the yearwas nine weeks. He had three fits in March, 1892. Hewas very dull mentally, and had been treated by verylarge doses of bromide of potassium, which diminished thefrequency of but did not arrest the fits. Operation by trephining was performed by Dr. McBur-ney on the 2d of April, 1892. The skull was opened atthe point of fracture over the arm centre on the right side(Fig. 14). The external table was found to be fracturedbut the internal table appeared to be uninjured, but a smallsplinter of bone was found indenting the dura. Thedura was very much thi
![American journal of pharmacy . Am. J. Ph.] 5 [May, 1884. ESTABLISHED 1822. ROSENGARTEN & SONS,MANUFACTURING CHEMISTS,PHILADELPHIA. Silver Medal Awarded by Franklin Institute, Nov., 1874,FOR PURITY OF CHEMICAL PREPARATIONS. Sulphate of Quinine. Muriate of Quinine. titrate of Iron and Quinine,Sulphate of Cinchonine, Sulphate of Cinchonidine,Sulphate of Quinidine, Bromide of Ammonium,Bromide of Potassium,Nitrate of Silver, Sulphate of Morphine. Acetate of Morphir«,Muriate of Morphine.Nitrate of Ammonia,Hypophosphites, Subnitrate of Bismuth,Spirits of Nitre,C. P, Acids,Tannin, AND A GENERAL ASSORT Stock Photo American journal of pharmacy . Am. J. Ph.] 5 [May, 1884. ESTABLISHED 1822. ROSENGARTEN & SONS,MANUFACTURING CHEMISTS,PHILADELPHIA. Silver Medal Awarded by Franklin Institute, Nov., 1874,FOR PURITY OF CHEMICAL PREPARATIONS. Sulphate of Quinine. Muriate of Quinine. titrate of Iron and Quinine,Sulphate of Cinchonine, Sulphate of Cinchonidine,Sulphate of Quinidine, Bromide of Ammonium,Bromide of Potassium,Nitrate of Silver, Sulphate of Morphine. Acetate of Morphir«,Muriate of Morphine.Nitrate of Ammonia,Hypophosphites, Subnitrate of Bismuth,Spirits of Nitre,C. P, Acids,Tannin, AND A GENERAL ASSORT Stock Photo](https://c8.alamy.com/comp/2AN1N69/american-journal-of-pharmacy-am-j-ph-5-may-1884-established-1822-rosengarten-sonsmanufacturing-chemistsphiladelphia-silver-medal-awarded-by-franklin-institute-nov-1874for-purity-of-chemical-preparations-sulphate-of-quinine-muriate-of-quinine-titrate-of-iron-and-quininesulphate-of-cinchonine-sulphate-of-cinchonidinesulphate-of-quinidine-bromide-of-ammoniumbromide-of-potassiumnitrate-of-silver-sulphate-of-morphine-acetate-of-morphirmuriate-of-morphinenitrate-of-ammoniahypophosphites-subnitrate-of-bismuthspirits-of-nitrec-p-acidstannin-and-a-general-assort-2AN1N69.jpg) American journal of pharmacy . Am. J. Ph.] 5 [May, 1884. ESTABLISHED 1822. ROSENGARTEN & SONS,MANUFACTURING CHEMISTS,PHILADELPHIA. Silver Medal Awarded by Franklin Institute, Nov., 1874,FOR PURITY OF CHEMICAL PREPARATIONS. Sulphate of Quinine. Muriate of Quinine. titrate of Iron and Quinine,Sulphate of Cinchonine, Sulphate of Cinchonidine,Sulphate of Quinidine, Bromide of Ammonium,Bromide of Potassium,Nitrate of Silver, Sulphate of Morphine. Acetate of Morphir«,Muriate of Morphine.Nitrate of Ammonia,Hypophosphites, Subnitrate of Bismuth,Spirits of Nitre,C. P, Acids,Tannin, AND A GENERAL ASSORT Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/american-journal-of-pharmacy-am-j-ph-5-may-1884-established-1822-rosengarten-sonsmanufacturing-chemistsphiladelphia-silver-medal-awarded-by-franklin-institute-nov-1874for-purity-of-chemical-preparations-sulphate-of-quinine-muriate-of-quinine-titrate-of-iron-and-quininesulphate-of-cinchonine-sulphate-of-cinchonidinesulphate-of-quinidine-bromide-of-ammoniumbromide-of-potassiumnitrate-of-silver-sulphate-of-morphine-acetate-of-morphirmuriate-of-morphinenitrate-of-ammoniahypophosphites-subnitrate-of-bismuthspirits-of-nitrec-p-acidstannin-and-a-general-assort-image339943361.html
American journal of pharmacy . Am. J. Ph.] 5 [May, 1884. ESTABLISHED 1822. ROSENGARTEN & SONS,MANUFACTURING CHEMISTS,PHILADELPHIA. Silver Medal Awarded by Franklin Institute, Nov., 1874,FOR PURITY OF CHEMICAL PREPARATIONS. Sulphate of Quinine. Muriate of Quinine. titrate of Iron and Quinine,Sulphate of Cinchonine, Sulphate of Cinchonidine,Sulphate of Quinidine, Bromide of Ammonium,Bromide of Potassium,Nitrate of Silver, Sulphate of Morphine. Acetate of Morphir«,Muriate of Morphine.Nitrate of Ammonia,Hypophosphites, Subnitrate of Bismuth,Spirits of Nitre,C. P, Acids,Tannin, AND A GENERAL ASSORT Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/american-journal-of-pharmacy-am-j-ph-5-may-1884-established-1822-rosengarten-sonsmanufacturing-chemistsphiladelphia-silver-medal-awarded-by-franklin-institute-nov-1874for-purity-of-chemical-preparations-sulphate-of-quinine-muriate-of-quinine-titrate-of-iron-and-quininesulphate-of-cinchonine-sulphate-of-cinchonidinesulphate-of-quinidine-bromide-of-ammoniumbromide-of-potassiumnitrate-of-silver-sulphate-of-morphine-acetate-of-morphirmuriate-of-morphinenitrate-of-ammoniahypophosphites-subnitrate-of-bismuthspirits-of-nitrec-p-acidstannin-and-a-general-assort-image339943361.htmlRM2AN1N69–American journal of pharmacy . Am. J. Ph.] 5 [May, 1884. ESTABLISHED 1822. ROSENGARTEN & SONS,MANUFACTURING CHEMISTS,PHILADELPHIA. Silver Medal Awarded by Franklin Institute, Nov., 1874,FOR PURITY OF CHEMICAL PREPARATIONS. Sulphate of Quinine. Muriate of Quinine. titrate of Iron and Quinine,Sulphate of Cinchonine, Sulphate of Cinchonidine,Sulphate of Quinidine, Bromide of Ammonium,Bromide of Potassium,Nitrate of Silver, Sulphate of Morphine. Acetate of Morphir«,Muriate of Morphine.Nitrate of Ammonia,Hypophosphites, Subnitrate of Bismuth,Spirits of Nitre,C. P, Acids,Tannin, AND A GENERAL ASSORT
 Pediatrics (N.Y.; Lond.) . es the diphtheria intoxication. Diphtheriaantitoxins neutralize the effects of that diphtheria intoxication.Serum is a good cultivation medium for the diphtheria bacillus. Now, we are evidently no more entitled to infer a priori fromthe one series of facts that the presence of antitoxins in the serumwould render it a bad culture medium for the diphtheria bacillusthan we are entitled to infer a priori from the other series of factSrthat the presence of opium or bromide of potassium in a sugar solu-tion would render that sugar solution a bad culture medium for theyeast Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/pediatrics-ny-lond-es-the-diphtheria-intoxication-diphtheriaantitoxins-neutralize-the-effects-of-that-diphtheria-intoxicationserum-is-a-good-cultivation-medium-for-the-diphtheria-bacillus-now-we-are-evidently-no-more-entitled-to-infer-a-priori-fromthe-one-series-of-facts-that-the-presence-of-antitoxins-in-the-serumwould-render-it-a-bad-culture-medium-for-the-diphtheria-bacillusthan-we-are-entitled-to-infer-a-priori-from-the-other-series-of-factsrthat-the-presence-of-opium-or-bromide-of-potassium-in-a-sugar-solu-tion-would-render-that-sugar-solution-a-bad-culture-medium-for-theyeast-image338513197.html
Pediatrics (N.Y.; Lond.) . es the diphtheria intoxication. Diphtheriaantitoxins neutralize the effects of that diphtheria intoxication.Serum is a good cultivation medium for the diphtheria bacillus. Now, we are evidently no more entitled to infer a priori fromthe one series of facts that the presence of antitoxins in the serumwould render it a bad culture medium for the diphtheria bacillusthan we are entitled to infer a priori from the other series of factSrthat the presence of opium or bromide of potassium in a sugar solu-tion would render that sugar solution a bad culture medium for theyeast Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/pediatrics-ny-lond-es-the-diphtheria-intoxication-diphtheriaantitoxins-neutralize-the-effects-of-that-diphtheria-intoxicationserum-is-a-good-cultivation-medium-for-the-diphtheria-bacillus-now-we-are-evidently-no-more-entitled-to-infer-a-priori-fromthe-one-series-of-facts-that-the-presence-of-antitoxins-in-the-serumwould-render-it-a-bad-culture-medium-for-the-diphtheria-bacillusthan-we-are-entitled-to-infer-a-priori-from-the-other-series-of-factsrthat-the-presence-of-opium-or-bromide-of-potassium-in-a-sugar-solu-tion-would-render-that-sugar-solution-a-bad-culture-medium-for-theyeast-image338513197.htmlRM2AJMH11–Pediatrics (N.Y.; Lond.) . es the diphtheria intoxication. Diphtheriaantitoxins neutralize the effects of that diphtheria intoxication.Serum is a good cultivation medium for the diphtheria bacillus. Now, we are evidently no more entitled to infer a priori fromthe one series of facts that the presence of antitoxins in the serumwould render it a bad culture medium for the diphtheria bacillusthan we are entitled to infer a priori from the other series of factSrthat the presence of opium or bromide of potassium in a sugar solu-tion would render that sugar solution a bad culture medium for theyeast
 A treatise on the diseases of the nervous system . the instrument withall its parts in their places). It was by this instrument that I was enabled to demonstrate, in themost conclusive manner, that during sleep the amount of blood circu-lating in the cerebral vessels is much less than during wakefulness, to 1 Also Journal of Psycholoyical Medicine, January, 1869, p. 47. 20 DISEASES OF THE NERVOUS SYSTEM. show the effect of bromide of potassium in lessening the amount ofblood in the brain, of the sulphate of quinine in increasing it, etc,,1 Fio. 1.. iESTHESIOMETEE. The aesthesiometer is an inst Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/a-treatise-on-the-diseases-of-the-nervous-system-the-instrument-withall-its-parts-in-their-places-it-was-by-this-instrument-that-i-was-enabled-to-demonstrate-in-themost-conclusive-manner-that-during-sleep-the-amount-of-blood-circu-lating-in-the-cerebral-vessels-is-much-less-than-during-wakefulness-to-1-also-journal-of-psycholoyical-medicine-january-1869-p-47-20-diseases-of-the-nervous-system-show-the-effect-of-bromide-of-potassium-in-lessening-the-amount-ofblood-in-the-brain-of-the-sulphate-of-quinine-in-increasing-it-etc1-fio-1-iesthesiometee-the-aesthesiometer-is-an-inst-image343076031.html
A treatise on the diseases of the nervous system . the instrument withall its parts in their places). It was by this instrument that I was enabled to demonstrate, in themost conclusive manner, that during sleep the amount of blood circu-lating in the cerebral vessels is much less than during wakefulness, to 1 Also Journal of Psycholoyical Medicine, January, 1869, p. 47. 20 DISEASES OF THE NERVOUS SYSTEM. show the effect of bromide of potassium in lessening the amount ofblood in the brain, of the sulphate of quinine in increasing it, etc,,1 Fio. 1.. iESTHESIOMETEE. The aesthesiometer is an inst Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/a-treatise-on-the-diseases-of-the-nervous-system-the-instrument-withall-its-parts-in-their-places-it-was-by-this-instrument-that-i-was-enabled-to-demonstrate-in-themost-conclusive-manner-that-during-sleep-the-amount-of-blood-circu-lating-in-the-cerebral-vessels-is-much-less-than-during-wakefulness-to-1-also-journal-of-psycholoyical-medicine-january-1869-p-47-20-diseases-of-the-nervous-system-show-the-effect-of-bromide-of-potassium-in-lessening-the-amount-ofblood-in-the-brain-of-the-sulphate-of-quinine-in-increasing-it-etc1-fio-1-iesthesiometee-the-aesthesiometer-is-an-inst-image343076031.htmlRM2AX4CYB–A treatise on the diseases of the nervous system . the instrument withall its parts in their places). It was by this instrument that I was enabled to demonstrate, in themost conclusive manner, that during sleep the amount of blood circu-lating in the cerebral vessels is much less than during wakefulness, to 1 Also Journal of Psycholoyical Medicine, January, 1869, p. 47. 20 DISEASES OF THE NERVOUS SYSTEM. show the effect of bromide of potassium in lessening the amount ofblood in the brain, of the sulphate of quinine in increasing it, etc,,1 Fio. 1.. iESTHESIOMETEE. The aesthesiometer is an inst
 Elements of chemistry : including the applications of the science in the arts . grains of iodine and 30 grains of iodide of potassiumbeiiif usually dissolved together in 1 ounce of water. The bromide of potassium i*capable also of dissolving bromine, but the solution of chloride of potassium has noaffinity for chlorine. Ferrocyanide of potassium. Yellow prussiate of potassa; K2FeCy3 + 3HQ>;eq. 184 + 27 or 2300 + 337.5. — This important salt is formed when carbonate ofpotassa is fused at a red heat in an iron pot, with animal mat-ter, such as dried blood, hoofs, clippings of hides, &c, and i Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/elements-of-chemistry-including-the-applications-of-the-science-in-the-arts-grains-of-iodine-and-30-grains-of-iodide-of-potassiumbeiiif-usually-dissolved-together-in-1-ounce-of-water-the-bromide-of-potassium-icapable-also-of-dissolving-bromine-but-the-solution-of-chloride-of-potassium-has-noaffinity-for-chlorine-ferrocyanide-of-potassium-yellow-prussiate-of-potassa-k2fecy3-3hqgteq-184-27-or-2300-3375-this-important-salt-is-formed-when-carbonate-ofpotassa-is-fused-at-a-red-heat-in-an-iron-pot-with-animal-mat-ter-such-as-dried-blood-hoofs-clippings-of-hides-c-and-i-image339431109.html
Elements of chemistry : including the applications of the science in the arts . grains of iodine and 30 grains of iodide of potassiumbeiiif usually dissolved together in 1 ounce of water. The bromide of potassium i*capable also of dissolving bromine, but the solution of chloride of potassium has noaffinity for chlorine. Ferrocyanide of potassium. Yellow prussiate of potassa; K2FeCy3 + 3HQ>;eq. 184 + 27 or 2300 + 337.5. — This important salt is formed when carbonate ofpotassa is fused at a red heat in an iron pot, with animal mat-ter, such as dried blood, hoofs, clippings of hides, &c, and i Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/elements-of-chemistry-including-the-applications-of-the-science-in-the-arts-grains-of-iodine-and-30-grains-of-iodide-of-potassiumbeiiif-usually-dissolved-together-in-1-ounce-of-water-the-bromide-of-potassium-icapable-also-of-dissolving-bromine-but-the-solution-of-chloride-of-potassium-has-noaffinity-for-chlorine-ferrocyanide-of-potassium-yellow-prussiate-of-potassa-k2fecy3-3hqgteq-184-27-or-2300-3375-this-important-salt-is-formed-when-carbonate-ofpotassa-is-fused-at-a-red-heat-in-an-iron-pot-with-animal-mat-ter-such-as-dried-blood-hoofs-clippings-of-hides-c-and-i-image339431109.htmlRM2AM6BRH–Elements of chemistry : including the applications of the science in the arts . grains of iodine and 30 grains of iodide of potassiumbeiiif usually dissolved together in 1 ounce of water. The bromide of potassium i*capable also of dissolving bromine, but the solution of chloride of potassium has noaffinity for chlorine. Ferrocyanide of potassium. Yellow prussiate of potassa; K2FeCy3 + 3HQ>;eq. 184 + 27 or 2300 + 337.5. — This important salt is formed when carbonate ofpotassa is fused at a red heat in an iron pot, with animal mat-ter, such as dried blood, hoofs, clippings of hides, &c, and i
 The Medical and surgical reporter . halfa glass of water. It is claimed by some prominent specialists in nervousdiseases that the Sodium Salt is more acceptable to thestomach than the Bromide Potassium. An almost cer-tain relief is given by the administration of this Effer-vescing Salt. It is also used with advantage in Indi-GBSTioN, Depression following alcoholic and otherexcesses, as well as Nervous Headache. It affordsspeedy relief for Mental and Physical Exhaustion. PREPARED ONLY BY WM. R. WARNER & CO. WM. R. WARNER & CO. Mellins Food A SOLUBLE DRY EXTRACT of Barley Malt andWheat, for addi Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-medical-and-surgical-reporter-halfa-glass-of-water-it-is-claimed-by-some-prominent-specialists-in-nervousdiseases-that-the-sodium-salt-is-more-acceptable-to-thestomach-than-the-bromide-potassium-an-almost-cer-tain-relief-is-given-by-the-administration-of-this-effer-vescing-salt-it-is-also-used-with-advantage-in-indi-gbstion-depression-following-alcoholic-and-otherexcesses-as-well-as-nervous-headache-it-affordsspeedy-relief-for-mental-and-physical-exhaustion-prepared-only-by-wm-r-warner-co-wm-r-warner-co-mellins-food-a-soluble-dry-extract-of-barley-malt-andwheat-for-addi-image338366126.html
The Medical and surgical reporter . halfa glass of water. It is claimed by some prominent specialists in nervousdiseases that the Sodium Salt is more acceptable to thestomach than the Bromide Potassium. An almost cer-tain relief is given by the administration of this Effer-vescing Salt. It is also used with advantage in Indi-GBSTioN, Depression following alcoholic and otherexcesses, as well as Nervous Headache. It affordsspeedy relief for Mental and Physical Exhaustion. PREPARED ONLY BY WM. R. WARNER & CO. WM. R. WARNER & CO. Mellins Food A SOLUBLE DRY EXTRACT of Barley Malt andWheat, for addi Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-medical-and-surgical-reporter-halfa-glass-of-water-it-is-claimed-by-some-prominent-specialists-in-nervousdiseases-that-the-sodium-salt-is-more-acceptable-to-thestomach-than-the-bromide-potassium-an-almost-cer-tain-relief-is-given-by-the-administration-of-this-effer-vescing-salt-it-is-also-used-with-advantage-in-indi-gbstion-depression-following-alcoholic-and-otherexcesses-as-well-as-nervous-headache-it-affordsspeedy-relief-for-mental-and-physical-exhaustion-prepared-only-by-wm-r-warner-co-wm-r-warner-co-mellins-food-a-soluble-dry-extract-of-barley-malt-andwheat-for-addi-image338366126.htmlRM2AJDWCE–The Medical and surgical reporter . halfa glass of water. It is claimed by some prominent specialists in nervousdiseases that the Sodium Salt is more acceptable to thestomach than the Bromide Potassium. An almost cer-tain relief is given by the administration of this Effer-vescing Salt. It is also used with advantage in Indi-GBSTioN, Depression following alcoholic and otherexcesses, as well as Nervous Headache. It affordsspeedy relief for Mental and Physical Exhaustion. PREPARED ONLY BY WM. R. WARNER & CO. WM. R. WARNER & CO. Mellins Food A SOLUBLE DRY EXTRACT of Barley Malt andWheat, for addi
 The Medical and surgical reporter . halfa glass of water. It is claimed by some prominent specialists in nervousdiseases that the Sodium Salt is more acceptable to thestomach than the Bromide Potassium. An almost cer-tain relief is given by the administration of this Eflfer-vescing Salt. It is also used with advantage in Indi-gestion, Depression following alcoholic and otherexcesses, as well as Nervous Headache. It affordsspeedy relief for Mental and Physical Exhaitstion. prepared only by WM. R. WARNER & CO. WM. R. WARNER & CO MelUns Food A SOLUBLE DRY EXTRACT of Barley Malt andWheat, for addi Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-medical-and-surgical-reporter-halfa-glass-of-water-it-is-claimed-by-some-prominent-specialists-in-nervousdiseases-that-the-sodium-salt-is-more-acceptable-to-thestomach-than-the-bromide-potassium-an-almost-cer-tain-relief-is-given-by-the-administration-of-this-eflfer-vescing-salt-it-is-also-used-with-advantage-in-indi-gestion-depression-following-alcoholic-and-otherexcesses-as-well-as-nervous-headache-it-affordsspeedy-relief-for-mental-and-physical-exhaitstion-prepared-only-by-wm-r-warner-co-wm-r-warner-co-meluns-food-a-soluble-dry-extract-of-barley-malt-andwheat-for-addi-image338362148.html
The Medical and surgical reporter . halfa glass of water. It is claimed by some prominent specialists in nervousdiseases that the Sodium Salt is more acceptable to thestomach than the Bromide Potassium. An almost cer-tain relief is given by the administration of this Eflfer-vescing Salt. It is also used with advantage in Indi-gestion, Depression following alcoholic and otherexcesses, as well as Nervous Headache. It affordsspeedy relief for Mental and Physical Exhaitstion. prepared only by WM. R. WARNER & CO. WM. R. WARNER & CO MelUns Food A SOLUBLE DRY EXTRACT of Barley Malt andWheat, for addi Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-medical-and-surgical-reporter-halfa-glass-of-water-it-is-claimed-by-some-prominent-specialists-in-nervousdiseases-that-the-sodium-salt-is-more-acceptable-to-thestomach-than-the-bromide-potassium-an-almost-cer-tain-relief-is-given-by-the-administration-of-this-eflfer-vescing-salt-it-is-also-used-with-advantage-in-indi-gestion-depression-following-alcoholic-and-otherexcesses-as-well-as-nervous-headache-it-affordsspeedy-relief-for-mental-and-physical-exhaitstion-prepared-only-by-wm-r-warner-co-wm-r-warner-co-meluns-food-a-soluble-dry-extract-of-barley-malt-andwheat-for-addi-image338362148.htmlRM2AJDMAC–The Medical and surgical reporter . halfa glass of water. It is claimed by some prominent specialists in nervousdiseases that the Sodium Salt is more acceptable to thestomach than the Bromide Potassium. An almost cer-tain relief is given by the administration of this Eflfer-vescing Salt. It is also used with advantage in Indi-gestion, Depression following alcoholic and otherexcesses, as well as Nervous Headache. It affordsspeedy relief for Mental and Physical Exhaitstion. prepared only by WM. R. WARNER & CO. WM. R. WARNER & CO MelUns Food A SOLUBLE DRY EXTRACT of Barley Malt andWheat, for addi
 The Medical and surgical reporter . in nervousdiseases that the Sodium Salt is more acceptable to thestomach than the Bromide Potassium. An almost cer-tain relief is given by the administration of this Effer-vescing Salt. It is also used with advantage in Indi-gestion, Depression following alcoholic and otherexcesses, as well as Nervous Headache. It affordsspeedy relief for Mental and Physical Exhaustion. prepared only by WM. R. WARNER & CO. WM. R. WARNER & CO, Mellins Food IFor IxxffeMi-ts «,xica. icis, K SOLUBLE DRY EXTRACT of Barley Malt andWheat, for addition to Fresh Cows Milk. Prepared u Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-medical-and-surgical-reporter-in-nervousdiseases-that-the-sodium-salt-is-more-acceptable-to-thestomach-than-the-bromide-potassium-an-almost-cer-tain-relief-is-given-by-the-administration-of-this-effer-vescing-salt-it-is-also-used-with-advantage-in-indi-gestion-depression-following-alcoholic-and-otherexcesses-as-well-as-nervous-headache-it-affordsspeedy-relief-for-mental-and-physical-exhaustion-prepared-only-by-wm-r-warner-co-wm-r-warner-co-mellins-food-ifor-ixxffemi-ts-xica-icis-k-soluble-dry-extract-of-barley-malt-andwheat-for-addition-to-fresh-cows-milk-prepared-u-image338194696.html
The Medical and surgical reporter . in nervousdiseases that the Sodium Salt is more acceptable to thestomach than the Bromide Potassium. An almost cer-tain relief is given by the administration of this Effer-vescing Salt. It is also used with advantage in Indi-gestion, Depression following alcoholic and otherexcesses, as well as Nervous Headache. It affordsspeedy relief for Mental and Physical Exhaustion. prepared only by WM. R. WARNER & CO. WM. R. WARNER & CO, Mellins Food IFor IxxffeMi-ts «,xica. icis, K SOLUBLE DRY EXTRACT of Barley Malt andWheat, for addition to Fresh Cows Milk. Prepared u Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-medical-and-surgical-reporter-in-nervousdiseases-that-the-sodium-salt-is-more-acceptable-to-thestomach-than-the-bromide-potassium-an-almost-cer-tain-relief-is-given-by-the-administration-of-this-effer-vescing-salt-it-is-also-used-with-advantage-in-indi-gestion-depression-following-alcoholic-and-otherexcesses-as-well-as-nervous-headache-it-affordsspeedy-relief-for-mental-and-physical-exhaustion-prepared-only-by-wm-r-warner-co-wm-r-warner-co-mellins-food-ifor-ixxffemi-ts-xica-icis-k-soluble-dry-extract-of-barley-malt-andwheat-for-addition-to-fresh-cows-milk-prepared-u-image338194696.htmlRM2AJ62P0–The Medical and surgical reporter . in nervousdiseases that the Sodium Salt is more acceptable to thestomach than the Bromide Potassium. An almost cer-tain relief is given by the administration of this Effer-vescing Salt. It is also used with advantage in Indi-gestion, Depression following alcoholic and otherexcesses, as well as Nervous Headache. It affordsspeedy relief for Mental and Physical Exhaustion. prepared only by WM. R. WARNER & CO. WM. R. WARNER & CO, Mellins Food IFor IxxffeMi-ts «,xica. icis, K SOLUBLE DRY EXTRACT of Barley Malt andWheat, for addition to Fresh Cows Milk. Prepared u
 The American annual of photography . mulsions and should be compared on repre-sentative emulsions in such classes as follows: r NC film. Negative emulsions { Commercial film ( Process film. ^ . - . ( Positive motion picture film. Positive emulsions i ^ , t . 1 ( beed Lantern plates. Paper emulsions ^ , . ^ . I Bromide and Artura Iris. Mixing the Developer When compounding test developers from the solid develop-ing agent, it is important to weigh out the various ingredientsvery carefully in order to insure an accurate comparison. In-stead of weighing out small quantities of potassium bromidei Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-american-annual-of-photography-mulsions-and-should-be-compared-on-repre-sentative-emulsions-in-such-classes-as-follows-r-nc-film-negative-emulsions-commercial-film-process-film-positive-motion-picture-film-positive-emulsions-i-t-1-beed-lantern-plates-paper-emulsions-i-bromide-and-artura-iris-mixing-the-developer-when-compounding-test-developers-from-the-solid-develop-ing-agent-it-is-important-to-weigh-out-the-various-ingredientsvery-carefully-in-order-to-insure-an-accurate-comparison-in-stead-of-weighing-out-small-quantities-of-potassium-bromidei-image339981133.html
The American annual of photography . mulsions and should be compared on repre-sentative emulsions in such classes as follows: r NC film. Negative emulsions { Commercial film ( Process film. ^ . - . ( Positive motion picture film. Positive emulsions i ^ , t . 1 ( beed Lantern plates. Paper emulsions ^ , . ^ . I Bromide and Artura Iris. Mixing the Developer When compounding test developers from the solid develop-ing agent, it is important to weigh out the various ingredientsvery carefully in order to insure an accurate comparison. In-stead of weighing out small quantities of potassium bromidei Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-american-annual-of-photography-mulsions-and-should-be-compared-on-repre-sentative-emulsions-in-such-classes-as-follows-r-nc-film-negative-emulsions-commercial-film-process-film-positive-motion-picture-film-positive-emulsions-i-t-1-beed-lantern-plates-paper-emulsions-i-bromide-and-artura-iris-mixing-the-developer-when-compounding-test-developers-from-the-solid-develop-ing-agent-it-is-important-to-weigh-out-the-various-ingredientsvery-carefully-in-order-to-insure-an-accurate-comparison-in-stead-of-weighing-out-small-quantities-of-potassium-bromidei-image339981133.htmlRM2AN3DB9–The American annual of photography . mulsions and should be compared on repre-sentative emulsions in such classes as follows: r NC film. Negative emulsions { Commercial film ( Process film. ^ . - . ( Positive motion picture film. Positive emulsions i ^ , t . 1 ( beed Lantern plates. Paper emulsions ^ , . ^ . I Bromide and Artura Iris. Mixing the Developer When compounding test developers from the solid develop-ing agent, it is important to weigh out the various ingredientsvery carefully in order to insure an accurate comparison. In-stead of weighing out small quantities of potassium bromidei
![. Rod and gun. o dry. Berkley ... Port Hope, Out. — It ig possible to giX greentones on bromide of silver paj>or by treating with lulers lead-intensilier and cobalt solution, which will result in the pro-duction of a very bright green t<uie. Sniooth pa])er is mostsuitable. As the resulting tone is very bright it is essentialthat the |)rint have plenty of contrast and clear whites. TheolH-ratiou ought to be started and liiiished with a good washing.The prints to be toiud are lirst bleached in the followingsolulion : Nitrate of lead 4 grams. Ueil prussiate of potassium ... 4 Water 300 .l Stock Photo . Rod and gun. o dry. Berkley ... Port Hope, Out. — It ig possible to giX greentones on bromide of silver paj>or by treating with lulers lead-intensilier and cobalt solution, which will result in the pro-duction of a very bright green t<uie. Sniooth pa])er is mostsuitable. As the resulting tone is very bright it is essentialthat the |)rint have plenty of contrast and clear whites. TheolH-ratiou ought to be started and liiiished with a good washing.The prints to be toiud are lirst bleached in the followingsolulion : Nitrate of lead 4 grams. Ueil prussiate of potassium ... 4 Water 300 .l Stock Photo](https://c8.alamy.com/comp/2AG9A2X/rod-and-gun-o-dry-berkley-port-hope-out-it-ig-possible-to-gix-greentones-on-bromide-of-silver-pajgtor-by-treating-with-lulers-lead-intensilier-and-cobalt-solution-which-will-result-in-the-pro-duction-of-a-very-bright-green-tltuie-sniooth-pa-er-is-mostsuitable-as-the-resulting-tone-is-very-bright-it-is-essentialthat-the-rint-have-plenty-of-contrast-and-clear-whites-theolh-ratiou-ought-to-be-started-and-liiiished-with-a-good-washingthe-prints-to-be-toiud-are-lirst-bleached-in-the-followingsolulion-nitrate-of-lead-4-grams-ueil-prussiate-of-potassium-4-water-300-l-2AG9A2X.jpg) . Rod and gun. o dry. Berkley ... Port Hope, Out. — It ig possible to giX greentones on bromide of silver paj>or by treating with lulers lead-intensilier and cobalt solution, which will result in the pro-duction of a very bright green t<uie. Sniooth pa])er is mostsuitable. As the resulting tone is very bright it is essentialthat the |)rint have plenty of contrast and clear whites. TheolH-ratiou ought to be started and liiiished with a good washing.The prints to be toiud are lirst bleached in the followingsolulion : Nitrate of lead 4 grams. Ueil prussiate of potassium ... 4 Water 300 .l Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/rod-and-gun-o-dry-berkley-port-hope-out-it-ig-possible-to-gix-greentones-on-bromide-of-silver-pajgtor-by-treating-with-lulers-lead-intensilier-and-cobalt-solution-which-will-result-in-the-pro-duction-of-a-very-bright-green-tltuie-sniooth-pa-er-is-mostsuitable-as-the-resulting-tone-is-very-bright-it-is-essentialthat-the-rint-have-plenty-of-contrast-and-clear-whites-theolh-ratiou-ought-to-be-started-and-liiiished-with-a-good-washingthe-prints-to-be-toiud-are-lirst-bleached-in-the-followingsolulion-nitrate-of-lead-4-grams-ueil-prussiate-of-potassium-4-water-300-l-image337036978.html
. Rod and gun. o dry. Berkley ... Port Hope, Out. — It ig possible to giX greentones on bromide of silver paj>or by treating with lulers lead-intensilier and cobalt solution, which will result in the pro-duction of a very bright green t<uie. Sniooth pa])er is mostsuitable. As the resulting tone is very bright it is essentialthat the |)rint have plenty of contrast and clear whites. TheolH-ratiou ought to be started and liiiished with a good washing.The prints to be toiud are lirst bleached in the followingsolulion : Nitrate of lead 4 grams. Ueil prussiate of potassium ... 4 Water 300 .l Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/rod-and-gun-o-dry-berkley-port-hope-out-it-ig-possible-to-gix-greentones-on-bromide-of-silver-pajgtor-by-treating-with-lulers-lead-intensilier-and-cobalt-solution-which-will-result-in-the-pro-duction-of-a-very-bright-green-tltuie-sniooth-pa-er-is-mostsuitable-as-the-resulting-tone-is-very-bright-it-is-essentialthat-the-rint-have-plenty-of-contrast-and-clear-whites-theolh-ratiou-ought-to-be-started-and-liiiished-with-a-good-washingthe-prints-to-be-toiud-are-lirst-bleached-in-the-followingsolulion-nitrate-of-lead-4-grams-ueil-prussiate-of-potassium-4-water-300-l-image337036978.htmlRM2AG9A2X–. Rod and gun. o dry. Berkley ... Port Hope, Out. — It ig possible to giX greentones on bromide of silver paj>or by treating with lulers lead-intensilier and cobalt solution, which will result in the pro-duction of a very bright green t<uie. Sniooth pa])er is mostsuitable. As the resulting tone is very bright it is essentialthat the |)rint have plenty of contrast and clear whites. TheolH-ratiou ought to be started and liiiished with a good washing.The prints to be toiud are lirst bleached in the followingsolulion : Nitrate of lead 4 grams. Ueil prussiate of potassium ... 4 Water 300 .l
 The Richmond medical journal . Vol. V. JANUARY, 1868. No. J. ORIGINAL COMMUNICATIONS. Qui. Docet Discit. Art. I.—Bromide of Potassium. By A. M. Fauntleroy,M. D., Staunton, Va. In the old chemical nomenclature, this salt was called thehydrobromate of potassa. The first description which we haveof it is by Balard, in 1826. Pourche first applied it remediallyin scrofula and goitre, 1828. Being a haloid salt, its effectswere thought to be analogous, though feebler, to the iodide ofpotassium, in syphilis and glandular enlargements. In thecomparative trial of their virtues, the bromide yielded unsat Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-richmond-medical-journal-vol-v-january-1868-no-j-original-communications-qui-docet-discit-art-ibromide-of-potassium-by-a-m-fauntleroym-d-staunton-va-in-the-old-chemical-nomenclature-this-salt-was-called-thehydrobromate-of-potassa-the-first-description-which-we-haveof-it-is-by-balard-in-1826-pourche-first-applied-it-remediallyin-scrofula-and-goitre-1828-being-a-haloid-salt-its-effectswere-thought-to-be-analogous-though-feebler-to-the-iodide-ofpotassium-in-syphilis-and-glandular-enlargements-in-thecomparative-trial-of-their-virtues-the-bromide-yielded-unsat-image338363163.html
The Richmond medical journal . Vol. V. JANUARY, 1868. No. J. ORIGINAL COMMUNICATIONS. Qui. Docet Discit. Art. I.—Bromide of Potassium. By A. M. Fauntleroy,M. D., Staunton, Va. In the old chemical nomenclature, this salt was called thehydrobromate of potassa. The first description which we haveof it is by Balard, in 1826. Pourche first applied it remediallyin scrofula and goitre, 1828. Being a haloid salt, its effectswere thought to be analogous, though feebler, to the iodide ofpotassium, in syphilis and glandular enlargements. In thecomparative trial of their virtues, the bromide yielded unsat Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-richmond-medical-journal-vol-v-january-1868-no-j-original-communications-qui-docet-discit-art-ibromide-of-potassium-by-a-m-fauntleroym-d-staunton-va-in-the-old-chemical-nomenclature-this-salt-was-called-thehydrobromate-of-potassa-the-first-description-which-we-haveof-it-is-by-balard-in-1826-pourche-first-applied-it-remediallyin-scrofula-and-goitre-1828-being-a-haloid-salt-its-effectswere-thought-to-be-analogous-though-feebler-to-the-iodide-ofpotassium-in-syphilis-and-glandular-enlargements-in-thecomparative-trial-of-their-virtues-the-bromide-yielded-unsat-image338363163.htmlRM2AJDNJK–The Richmond medical journal . Vol. V. JANUARY, 1868. No. J. ORIGINAL COMMUNICATIONS. Qui. Docet Discit. Art. I.—Bromide of Potassium. By A. M. Fauntleroy,M. D., Staunton, Va. In the old chemical nomenclature, this salt was called thehydrobromate of potassa. The first description which we haveof it is by Balard, in 1826. Pourche first applied it remediallyin scrofula and goitre, 1828. Being a haloid salt, its effectswere thought to be analogous, though feebler, to the iodide ofpotassium, in syphilis and glandular enlargements. In thecomparative trial of their virtues, the bromide yielded unsat
 Portrait . when added inlarge quantities. One drop of a saturated solution to each ounce ofdeveloper is usually sufficient to prevent abrasionmarks, and one additional drop of bromide of potassiumto each ounce should also be added, as it tends to pre-serve the color and vigor of the print. When iodide of potassium is used in the hypo-alum toning bath it should never be added except insolution. A hypo-alum bath, to which crystals of iodide havebeen added, may cause yellow stains on the prints. 11 *P o r t r a it A Useful Electric Heater With the almost universal use of the hot hypo-alum method Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/portrait-when-added-inlarge-quantities-one-drop-of-a-saturated-solution-to-each-ounce-ofdeveloper-is-usually-sufficient-to-prevent-abrasionmarks-and-one-additional-drop-of-bromide-of-potassiumto-each-ounce-should-also-be-added-as-it-tends-to-pre-serve-the-color-and-vigor-of-the-print-when-iodide-of-potassium-is-used-in-the-hypo-alum-toning-bath-it-should-never-be-added-except-insolution-a-hypo-alum-bath-to-which-crystals-of-iodide-havebeen-added-may-cause-yellow-stains-on-the-prints-11-p-o-r-t-r-a-it-a-useful-electric-heater-with-the-almost-universal-use-of-the-hot-hypo-alum-method-image342813706.html
Portrait . when added inlarge quantities. One drop of a saturated solution to each ounce ofdeveloper is usually sufficient to prevent abrasionmarks, and one additional drop of bromide of potassiumto each ounce should also be added, as it tends to pre-serve the color and vigor of the print. When iodide of potassium is used in the hypo-alum toning bath it should never be added except insolution. A hypo-alum bath, to which crystals of iodide havebeen added, may cause yellow stains on the prints. 11 *P o r t r a it A Useful Electric Heater With the almost universal use of the hot hypo-alum method Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/portrait-when-added-inlarge-quantities-one-drop-of-a-saturated-solution-to-each-ounce-ofdeveloper-is-usually-sufficient-to-prevent-abrasionmarks-and-one-additional-drop-of-bromide-of-potassiumto-each-ounce-should-also-be-added-as-it-tends-to-pre-serve-the-color-and-vigor-of-the-print-when-iodide-of-potassium-is-used-in-the-hypo-alum-toning-bath-it-should-never-be-added-except-insolution-a-hypo-alum-bath-to-which-crystals-of-iodide-havebeen-added-may-cause-yellow-stains-on-the-prints-11-p-o-r-t-r-a-it-a-useful-electric-heater-with-the-almost-universal-use-of-the-hot-hypo-alum-method-image342813706.htmlRM2AWMEAJ–Portrait . when added inlarge quantities. One drop of a saturated solution to each ounce ofdeveloper is usually sufficient to prevent abrasionmarks, and one additional drop of bromide of potassiumto each ounce should also be added, as it tends to pre-serve the color and vigor of the print. When iodide of potassium is used in the hypo-alum toning bath it should never be added except insolution. A hypo-alum bath, to which crystals of iodide havebeen added, may cause yellow stains on the prints. 11 *P o r t r a it A Useful Electric Heater With the almost universal use of the hot hypo-alum method
 The American annual of photography . olution and ten or twentyminims added to the ounce of developer. An excellent method of obtaining warm colours is by way ofa good black image subsequently toned by potassium f erricyan-ide and sulphide of soda, as when toning bromide prints. Veilor stain of any kind must be carefully avoided if brilliance ofimage on the screen is desired. On the other hand, a veryvigorous image is very far from pleasant when projected.Strive to obtain a slide that will give a soft well gradatedimage on the screen with just the extreme high lights havingsome sparkle. It is n Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-american-annual-of-photography-olution-and-ten-or-twentyminims-added-to-the-ounce-of-developer-an-excellent-method-of-obtaining-warm-colours-is-by-way-ofa-good-black-image-subsequently-toned-by-potassium-f-erricyan-ide-and-sulphide-of-soda-as-when-toning-bromide-prints-veilor-stain-of-any-kind-must-be-carefully-avoided-if-brilliance-ofimage-on-the-screen-is-desired-on-the-other-hand-a-veryvigorous-image-is-very-far-from-pleasant-when-projectedstrive-to-obtain-a-slide-that-will-give-a-soft-well-gradatedimage-on-the-screen-with-just-the-extreme-high-lights-havingsome-sparkle-it-is-n-image339029083.html
The American annual of photography . olution and ten or twentyminims added to the ounce of developer. An excellent method of obtaining warm colours is by way ofa good black image subsequently toned by potassium f erricyan-ide and sulphide of soda, as when toning bromide prints. Veilor stain of any kind must be carefully avoided if brilliance ofimage on the screen is desired. On the other hand, a veryvigorous image is very far from pleasant when projected.Strive to obtain a slide that will give a soft well gradatedimage on the screen with just the extreme high lights havingsome sparkle. It is n Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-american-annual-of-photography-olution-and-ten-or-twentyminims-added-to-the-ounce-of-developer-an-excellent-method-of-obtaining-warm-colours-is-by-way-ofa-good-black-image-subsequently-toned-by-potassium-f-erricyan-ide-and-sulphide-of-soda-as-when-toning-bromide-prints-veilor-stain-of-any-kind-must-be-carefully-avoided-if-brilliance-ofimage-on-the-screen-is-desired-on-the-other-hand-a-veryvigorous-image-is-very-far-from-pleasant-when-projectedstrive-to-obtain-a-slide-that-will-give-a-soft-well-gradatedimage-on-the-screen-with-just-the-extreme-high-lights-havingsome-sparkle-it-is-n-image339029083.htmlRM2AKG31F–The American annual of photography . olution and ten or twentyminims added to the ounce of developer. An excellent method of obtaining warm colours is by way ofa good black image subsequently toned by potassium f erricyan-ide and sulphide of soda, as when toning bromide prints. Veilor stain of any kind must be carefully avoided if brilliance ofimage on the screen is desired. On the other hand, a veryvigorous image is very far from pleasant when projected.Strive to obtain a slide that will give a soft well gradatedimage on the screen with just the extreme high lights havingsome sparkle. It is n
 The American annual of photography . ss of ruby cloth, andinside is a 60 watt tungsten lamp. This gives me ample light,and I place my developing tray directly beneath the light. DEVELOPER: Amidol (Johnsons British) made up asfollows: Stock solution: Water 32 oz. Sulphite Soda (dry) 2 oz. Citric Acid 25 gr. For use dilute with equal amount of water to which addwhen ready to use 25 grains of Amidol to each 32 oz. of work-ing solution. Bromide of Potassium (saturated solution) 3 drops to eachoz. of working developer. The sulphite stock solution keeps, but the amidol should notbe added until ready Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-american-annual-of-photography-ss-of-ruby-cloth-andinside-is-a-60-watt-tungsten-lamp-this-gives-me-ample-lightand-i-place-my-developing-tray-directly-beneath-the-light-developer-amidol-johnsons-british-made-up-asfollows-stock-solution-water-32-oz-sulphite-soda-dry-2-oz-citric-acid-25-gr-for-use-dilute-with-equal-amount-of-water-to-which-addwhen-ready-to-use-25-grains-of-amidol-to-each-32-oz-of-work-ing-solution-bromide-of-potassium-saturated-solution-3-drops-to-eachoz-of-working-developer-the-sulphite-stock-solution-keeps-but-the-amidol-should-notbe-added-until-ready-image339131096.html
The American annual of photography . ss of ruby cloth, andinside is a 60 watt tungsten lamp. This gives me ample light,and I place my developing tray directly beneath the light. DEVELOPER: Amidol (Johnsons British) made up asfollows: Stock solution: Water 32 oz. Sulphite Soda (dry) 2 oz. Citric Acid 25 gr. For use dilute with equal amount of water to which addwhen ready to use 25 grains of Amidol to each 32 oz. of work-ing solution. Bromide of Potassium (saturated solution) 3 drops to eachoz. of working developer. The sulphite stock solution keeps, but the amidol should notbe added until ready Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-american-annual-of-photography-ss-of-ruby-cloth-andinside-is-a-60-watt-tungsten-lamp-this-gives-me-ample-lightand-i-place-my-developing-tray-directly-beneath-the-light-developer-amidol-johnsons-british-made-up-asfollows-stock-solution-water-32-oz-sulphite-soda-dry-2-oz-citric-acid-25-gr-for-use-dilute-with-equal-amount-of-water-to-which-addwhen-ready-to-use-25-grains-of-amidol-to-each-32-oz-of-work-ing-solution-bromide-of-potassium-saturated-solution-3-drops-to-eachoz-of-working-developer-the-sulphite-stock-solution-keeps-but-the-amidol-should-notbe-added-until-ready-image339131096.htmlRM2AKMN4T–The American annual of photography . ss of ruby cloth, andinside is a 60 watt tungsten lamp. This gives me ample light,and I place my developing tray directly beneath the light. DEVELOPER: Amidol (Johnsons British) made up asfollows: Stock solution: Water 32 oz. Sulphite Soda (dry) 2 oz. Citric Acid 25 gr. For use dilute with equal amount of water to which addwhen ready to use 25 grains of Amidol to each 32 oz. of work-ing solution. Bromide of Potassium (saturated solution) 3 drops to eachoz. of working developer. The sulphite stock solution keeps, but the amidol should notbe added until ready
 American journal of pharmacy . ectly Soluble SUGAR-COATED PILLS & GRANULES.Chemical and Pharmaceutical Apparatus,Chemical Reagents. PHAEMACEUTICAL PREPARATIONS,SELECTED DRUGS,ESSENTIAL OILS, EXTRACTS. &c., kc, &c. BII^LIIMG^N, CL^I^F» &c CO. Succeisort to JAS. B. NICHOLS * CO. MAJVUFACTURrJVG CHEMISTS, AND Standard Pharmacsutical Prsparations. BOSTON, MASS., & 20 LIBERTY STREET, N. Y. We solicit correspondence npon the following specialties: AMMONIUM BROMIDE, CARBOLIC ACID, P0TASSIU3I BROMIDE,BROMIXE, POTASSIUM ACETATE, CALCIUM SULPHITE (suiphite L!n.«).PROPYLAMINE, PROPYLAMINE CHLORIDE, SODIU Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/american-journal-of-pharmacy-ectly-soluble-sugar-coated-pills-granuleschemical-and-pharmaceutical-apparatuschemical-reagents-phaemaceutical-preparationsselected-drugsessential-oils-extracts-c-kc-c-biiliimgn-clif-c-co-succeisort-to-jas-b-nichols-co-majvufacturrjvg-chemists-and-standard-pharmacsutical-prsparations-boston-mass-20-liberty-street-n-y-we-solicit-correspondence-npon-the-following-specialties-ammonium-bromide-carbolic-acid-p0tassiu3i-bromidebromixe-potassium-acetate-calcium-sulphite-suiphite-l!npropylamine-propylamine-chloride-sodiu-image338226011.html
American journal of pharmacy . ectly Soluble SUGAR-COATED PILLS & GRANULES.Chemical and Pharmaceutical Apparatus,Chemical Reagents. PHAEMACEUTICAL PREPARATIONS,SELECTED DRUGS,ESSENTIAL OILS, EXTRACTS. &c., kc, &c. BII^LIIMG^N, CL^I^F» &c CO. Succeisort to JAS. B. NICHOLS * CO. MAJVUFACTURrJVG CHEMISTS, AND Standard Pharmacsutical Prsparations. BOSTON, MASS., & 20 LIBERTY STREET, N. Y. We solicit correspondence npon the following specialties: AMMONIUM BROMIDE, CARBOLIC ACID, P0TASSIU3I BROMIDE,BROMIXE, POTASSIUM ACETATE, CALCIUM SULPHITE (suiphite L!n.«).PROPYLAMINE, PROPYLAMINE CHLORIDE, SODIU Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/american-journal-of-pharmacy-ectly-soluble-sugar-coated-pills-granuleschemical-and-pharmaceutical-apparatuschemical-reagents-phaemaceutical-preparationsselected-drugsessential-oils-extracts-c-kc-c-biiliimgn-clif-c-co-succeisort-to-jas-b-nichols-co-majvufacturrjvg-chemists-and-standard-pharmacsutical-prsparations-boston-mass-20-liberty-street-n-y-we-solicit-correspondence-npon-the-following-specialties-ammonium-bromide-carbolic-acid-p0tassiu3i-bromidebromixe-potassium-acetate-calcium-sulphite-suiphite-l!npropylamine-propylamine-chloride-sodiu-image338226011.htmlRM2AJ7EMB–American journal of pharmacy . ectly Soluble SUGAR-COATED PILLS & GRANULES.Chemical and Pharmaceutical Apparatus,Chemical Reagents. PHAEMACEUTICAL PREPARATIONS,SELECTED DRUGS,ESSENTIAL OILS, EXTRACTS. &c., kc, &c. BII^LIIMG^N, CL^I^F» &c CO. Succeisort to JAS. B. NICHOLS * CO. MAJVUFACTURrJVG CHEMISTS, AND Standard Pharmacsutical Prsparations. BOSTON, MASS., & 20 LIBERTY STREET, N. Y. We solicit correspondence npon the following specialties: AMMONIUM BROMIDE, CARBOLIC ACID, P0TASSIU3I BROMIDE,BROMIXE, POTASSIUM ACETATE, CALCIUM SULPHITE (suiphite L!n.«).PROPYLAMINE, PROPYLAMINE CHLORIDE, SODIU
 The American annual of photography . ter rinsing) anddeveloped in daylight. The developer should be Metol-hydro-quinone and sodium or potassium hydrate and containing con-siderable bromide. Another curiosity that I have found is thefollowing method of developing plates in daylight, with de-velopment and fixing simultaneous. Make a powder as follows— Magnesium picrate 8i gms Sodium Sulphite (Dry) 544 gms Hypo C. P 250 gms Dianol 125 gms 4 grams of this mixture in 100 cc. of water is used and whenthe plate is once placed in this, it may be brought out into thelight and development and fixing are Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-american-annual-of-photography-ter-rinsing-anddeveloped-in-daylight-the-developer-should-be-metol-hydro-quinone-and-sodium-or-potassium-hydrate-and-containing-con-siderable-bromide-another-curiosity-that-i-have-found-is-thefollowing-method-of-developing-plates-in-daylight-with-de-velopment-and-fixing-simultaneous-make-a-powder-as-follows-magnesium-picrate-8i-gms-sodium-sulphite-dry-544-gms-hypo-c-p-250-gms-dianol-125-gms-4-grams-of-this-mixture-in-100-cc-of-water-is-used-and-whenthe-plate-is-once-placed-in-this-it-may-be-brought-out-into-thelight-and-development-and-fixing-are-image340251455.html
The American annual of photography . ter rinsing) anddeveloped in daylight. The developer should be Metol-hydro-quinone and sodium or potassium hydrate and containing con-siderable bromide. Another curiosity that I have found is thefollowing method of developing plates in daylight, with de-velopment and fixing simultaneous. Make a powder as follows— Magnesium picrate 8i gms Sodium Sulphite (Dry) 544 gms Hypo C. P 250 gms Dianol 125 gms 4 grams of this mixture in 100 cc. of water is used and whenthe plate is once placed in this, it may be brought out into thelight and development and fixing are Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-american-annual-of-photography-ter-rinsing-anddeveloped-in-daylight-the-developer-should-be-metol-hydro-quinone-and-sodium-or-potassium-hydrate-and-containing-con-siderable-bromide-another-curiosity-that-i-have-found-is-thefollowing-method-of-developing-plates-in-daylight-with-de-velopment-and-fixing-simultaneous-make-a-powder-as-follows-magnesium-picrate-8i-gms-sodium-sulphite-dry-544-gms-hypo-c-p-250-gms-dianol-125-gms-4-grams-of-this-mixture-in-100-cc-of-water-is-used-and-whenthe-plate-is-once-placed-in-this-it-may-be-brought-out-into-thelight-and-development-and-fixing-are-image340251455.htmlRM2ANFP5K–The American annual of photography . ter rinsing) anddeveloped in daylight. The developer should be Metol-hydro-quinone and sodium or potassium hydrate and containing con-siderable bromide. Another curiosity that I have found is thefollowing method of developing plates in daylight, with de-velopment and fixing simultaneous. Make a powder as follows— Magnesium picrate 8i gms Sodium Sulphite (Dry) 544 gms Hypo C. P 250 gms Dianol 125 gms 4 grams of this mixture in 100 cc. of water is used and whenthe plate is once placed in this, it may be brought out into thelight and development and fixing are
 The American annual of photography . ON A MOUNTAIN ROAD. EDGAR A. COHEN. ^Z7. A WORD FOR DIAMIDOPHENOL By EDWIN LOKER HE above stumper in the way of a name is bet-ter known by the trade as Amidol, Dianol, etc.,and in caustic solution as Rodinal. Thischemical is to my mind one of the most satis-factory all-round developers, especially in acidsolution, that I have ever come across, both in working andcheapness, especially for the amateur, and I can confidentlyrecommend the following: Water 20 oz Sodium Sulphite ij/^ oz. Sodium Bisulphite 10 dr. Bromide Potassium JO gi- Bottle the above in 3 or 6 Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-american-annual-of-photography-on-a-mountain-road-edgar-a-cohen-z7-a-word-for-diamidophenol-by-edwin-loker-he-above-stumper-in-the-way-of-a-name-is-bet-ter-known-by-the-trade-as-amidol-dianol-etcand-in-caustic-solution-as-rodinal-thischemical-is-to-my-mind-one-of-the-most-satis-factory-all-round-developers-especially-in-acidsolution-that-i-have-ever-come-across-both-in-working-andcheapness-especially-for-the-amateur-and-i-can-confidentlyrecommend-the-following-water-20-oz-sodium-sulphite-ij-oz-sodium-bisulphite-10-dr-bromide-potassium-jo-gi-bottle-the-above-in-3-or-6-image339103414.html
The American annual of photography . ON A MOUNTAIN ROAD. EDGAR A. COHEN. ^Z7. A WORD FOR DIAMIDOPHENOL By EDWIN LOKER HE above stumper in the way of a name is bet-ter known by the trade as Amidol, Dianol, etc.,and in caustic solution as Rodinal. Thischemical is to my mind one of the most satis-factory all-round developers, especially in acidsolution, that I have ever come across, both in working andcheapness, especially for the amateur, and I can confidentlyrecommend the following: Water 20 oz Sodium Sulphite ij/^ oz. Sodium Bisulphite 10 dr. Bromide Potassium JO gi- Bottle the above in 3 or 6 Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-american-annual-of-photography-on-a-mountain-road-edgar-a-cohen-z7-a-word-for-diamidophenol-by-edwin-loker-he-above-stumper-in-the-way-of-a-name-is-bet-ter-known-by-the-trade-as-amidol-dianol-etcand-in-caustic-solution-as-rodinal-thischemical-is-to-my-mind-one-of-the-most-satis-factory-all-round-developers-especially-in-acidsolution-that-i-have-ever-come-across-both-in-working-andcheapness-especially-for-the-amateur-and-i-can-confidentlyrecommend-the-following-water-20-oz-sodium-sulphite-ij-oz-sodium-bisulphite-10-dr-bromide-potassium-jo-gi-bottle-the-above-in-3-or-6-image339103414.htmlRM2AKKDT6–The American annual of photography . ON A MOUNTAIN ROAD. EDGAR A. COHEN. ^Z7. A WORD FOR DIAMIDOPHENOL By EDWIN LOKER HE above stumper in the way of a name is bet-ter known by the trade as Amidol, Dianol, etc.,and in caustic solution as Rodinal. Thischemical is to my mind one of the most satis-factory all-round developers, especially in acidsolution, that I have ever come across, both in working andcheapness, especially for the amateur, and I can confidentlyrecommend the following: Water 20 oz Sodium Sulphite ij/^ oz. Sodium Bisulphite 10 dr. Bromide Potassium JO gi- Bottle the above in 3 or 6
 A manual of photographic chemistry, theoretical and practical . emulsion plate is com-posed by— » j Pyrogallic acid, ....... 3 grains. I Water, L ounce. p i Bromide of potassium 10 grains. I Water, 1 ounce. p ) Carbonate of ammonia 30 grains. Water, 1 ounce. 318 PRACTICE OF PHOTOGRAPHY. To every half-ounce of A add two drops of B and threedrops of C. The negative is fixed with either cyanide of potassium orhyposulphite of soda. Dry Bath Plates.—Dry collodion plates may be preparedby the bath by sensitizing as usual and then thoroughly wash-ing off the nitrate of silver. A preservative is the Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/a-manual-of-photographic-chemistry-theoretical-and-practical-emulsion-plate-is-com-posed-by-j-pyrogallic-acid-3-grains-i-water-l-ounce-p-i-bromide-of-potassium-10-grains-i-water-1-ounce-p-carbonate-of-ammonia-30-grains-water-1-ounce-318-practice-of-photography-to-every-half-ounce-of-a-add-two-drops-of-b-and-threedrops-of-c-the-negative-is-fixed-with-either-cyanide-of-potassium-orhyposulphite-of-soda-dry-bath-platesdry-collodion-plates-may-be-preparedby-the-bath-by-sensitizing-as-usual-and-then-thoroughly-wash-ing-off-the-nitrate-of-silver-a-preservative-is-the-image340242747.html
A manual of photographic chemistry, theoretical and practical . emulsion plate is com-posed by— » j Pyrogallic acid, ....... 3 grains. I Water, L ounce. p i Bromide of potassium 10 grains. I Water, 1 ounce. p ) Carbonate of ammonia 30 grains. Water, 1 ounce. 318 PRACTICE OF PHOTOGRAPHY. To every half-ounce of A add two drops of B and threedrops of C. The negative is fixed with either cyanide of potassium orhyposulphite of soda. Dry Bath Plates.—Dry collodion plates may be preparedby the bath by sensitizing as usual and then thoroughly wash-ing off the nitrate of silver. A preservative is the Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/a-manual-of-photographic-chemistry-theoretical-and-practical-emulsion-plate-is-com-posed-by-j-pyrogallic-acid-3-grains-i-water-l-ounce-p-i-bromide-of-potassium-10-grains-i-water-1-ounce-p-carbonate-of-ammonia-30-grains-water-1-ounce-318-practice-of-photography-to-every-half-ounce-of-a-add-two-drops-of-b-and-threedrops-of-c-the-negative-is-fixed-with-either-cyanide-of-potassium-orhyposulphite-of-soda-dry-bath-platesdry-collodion-plates-may-be-preparedby-the-bath-by-sensitizing-as-usual-and-then-thoroughly-wash-ing-off-the-nitrate-of-silver-a-preservative-is-the-image340242747.htmlRM2ANFB2K–A manual of photographic chemistry, theoretical and practical . emulsion plate is com-posed by— » j Pyrogallic acid, ....... 3 grains. I Water, L ounce. p i Bromide of potassium 10 grains. I Water, 1 ounce. p ) Carbonate of ammonia 30 grains. Water, 1 ounce. 318 PRACTICE OF PHOTOGRAPHY. To every half-ounce of A add two drops of B and threedrops of C. The negative is fixed with either cyanide of potassium orhyposulphite of soda. Dry Bath Plates.—Dry collodion plates may be preparedby the bath by sensitizing as usual and then thoroughly wash-ing off the nitrate of silver. A preservative is the
Search Results for Potassium bromide Stock Photos and Images (118)
Page 1 of 2