Quick filters:
Page 1 of 2
Absorption spectrometer Stock Photos and Images

RF2CD7NA3–Kirchhoff optical spectrometer or spectroscope, instrument to measure properties of light over a specific portion of the electromagnetic spectrum in spectroscopic analysis to identify materials

RM2E3XXGB–Nicolet iS5 FTIR Spectrometer provides information and for product assurance testing and material. It is designed for scientists, production managers

RMA9G2G2–Laboratory chemical analysis. Absorption Spectrometer used by Indian scientist in the production of polymers

RF2RNN481–Fourier Transform Infrared (FTIR) Spectroscopy: Place the sample in the cuvette and measure its absorption of infrared light using a Fourier transform

RMDP5MBE–Sep 19, 2006; San Diego, CA, USA; Chemist MANUEL ROMERO, checked for iron in water samples with an atomic absorption spectrometer at the City of San Diego Water Quality Laboratory Environmental Monitoring and Technical Services Division on Monday, September 19, 2006. Mandatory Credit: Photo by John Gibbins/SDU-T/ZUMA Press. (©) Copyright 2006 by SDU-T

RFKD5DDK–Identification of white powder raw material by FTIR method, advance technology without sample preparation high sensitivity and accuracy method, concept of pharmaceutical industrial laboratory
![. Spectroradiometric investigation of the transmission of various substances . ork several years ago; this Bulletin, 7, p.24s, 1911; 10, p. 38(Fig. s), 1912. The mirror spectrometer and fluorite prism is described in this Bulletin,10, p. 1, 1913; and the thermopile is described in this Bulletin, 11, p. 132, 1914- * In this Bulletin, 9, p. n6,1912, data are given on the transmission of green fluorites. some of which hadmarked absorption bands. Coblentz,Emerson, Long] Transmission of Various Substances 655 2. GELATIN LIGHT FILTERS In photographing the infra-red spectrum using plates sensitizedwi Stock Photo . Spectroradiometric investigation of the transmission of various substances . ork several years ago; this Bulletin, 7, p.24s, 1911; 10, p. 38(Fig. s), 1912. The mirror spectrometer and fluorite prism is described in this Bulletin,10, p. 1, 1913; and the thermopile is described in this Bulletin, 11, p. 132, 1914- * In this Bulletin, 9, p. n6,1912, data are given on the transmission of green fluorites. some of which hadmarked absorption bands. Coblentz,Emerson, Long] Transmission of Various Substances 655 2. GELATIN LIGHT FILTERS In photographing the infra-red spectrum using plates sensitizedwi Stock Photo](https://c8.alamy.com/comp/2CRR80R/spectroradiometric-investigation-of-the-transmission-of-various-substances-ork-several-years-ago-this-bulletin-7-p24s-1911-10-p-38fig-s-1912-the-mirror-spectrometer-and-fluorite-prism-is-described-in-this-bulletin10-p-1-1913-and-the-thermopile-is-described-in-this-bulletin-11-p-132-1914-in-this-bulletin-9-p-n61912-data-are-given-on-the-transmission-of-green-fluorites-some-of-which-hadmarked-absorption-bands-coblentzemerson-long-transmission-of-various-substances-655-2-gelatin-light-filters-in-photographing-the-infra-red-spectrum-using-plates-sensitizedwi-2CRR80R.jpg)
RM2CRR80R–. Spectroradiometric investigation of the transmission of various substances . ork several years ago; this Bulletin, 7, p.24s, 1911; 10, p. 38(Fig. s), 1912. The mirror spectrometer and fluorite prism is described in this Bulletin,10, p. 1, 1913; and the thermopile is described in this Bulletin, 11, p. 132, 1914- * In this Bulletin, 9, p. n6,1912, data are given on the transmission of green fluorites. some of which hadmarked absorption bands. Coblentz,Emerson, Long] Transmission of Various Substances 655 2. GELATIN LIGHT FILTERS In photographing the infra-red spectrum using plates sensitizedwi

RF2CD7NA7–The spectroscope developed in 19th century by Bunsen and Kirchhoff provides a high quality optical system and an easy-to-read scale, allowing to measure discrete atomic spectral lines

RF2CD7NBT–Spectroscope of the Observatoy of the University of Kiev allowing to study solar processes and solar activity

RM2E3XXG5–Nicolet iS5 FTIR Spectrometer provides information and for product assurance testing and material. It is designed for scientists, production managers

RF2CD7NBF–Huggins spectroscope developed by Sir William Huggins with the technical innovation of by widening the slit and measuring the radial velocity of a star and to study the evolution of the universe

RF2CD7NBJ–Universal spectroscope complete apparatus with universal stand for general use developed by Hermann Wilhelm Vogel German photochemist

RFKD5DD8–Identification of white powder raw material by FTIR method, advance technology without sample preparation high sensitivity and accuracy method, concept of pharmaceutical industrial laboratory

RMRFWBW0–. Carnegie Institution of Washington publication. ^^ Fig. I. -Inner room containing radiometer R, spectrometer and prism P, exhaust pump G, lamps and scale L, and telescope T. C is the holder for the absorption cell. S is the shutter, H the Nernst lamp-heater.. Fig. 1 A.—Large spectrometer with Nernst heater, /*, to the right, and radiometer, r, to the left. The gas-cell holde and glass cells are shown at 9; Ceissler pump in the rear. Photograph taken through doorway of inner room.. Please note that these images are extracted from scanned page images that may have been digitally enhanced for

RM2E3XXG7–Nicolet iS5 FTIR Spectrometer provides information and for product assurance testing and material. It is designed for scientists, production managers

RFKD5DEN–Identification of white powder raw material by FTIR method, advance technology without sample preparation high sensitivity and accuracy method, concept of pharmaceutical industrial laboratory
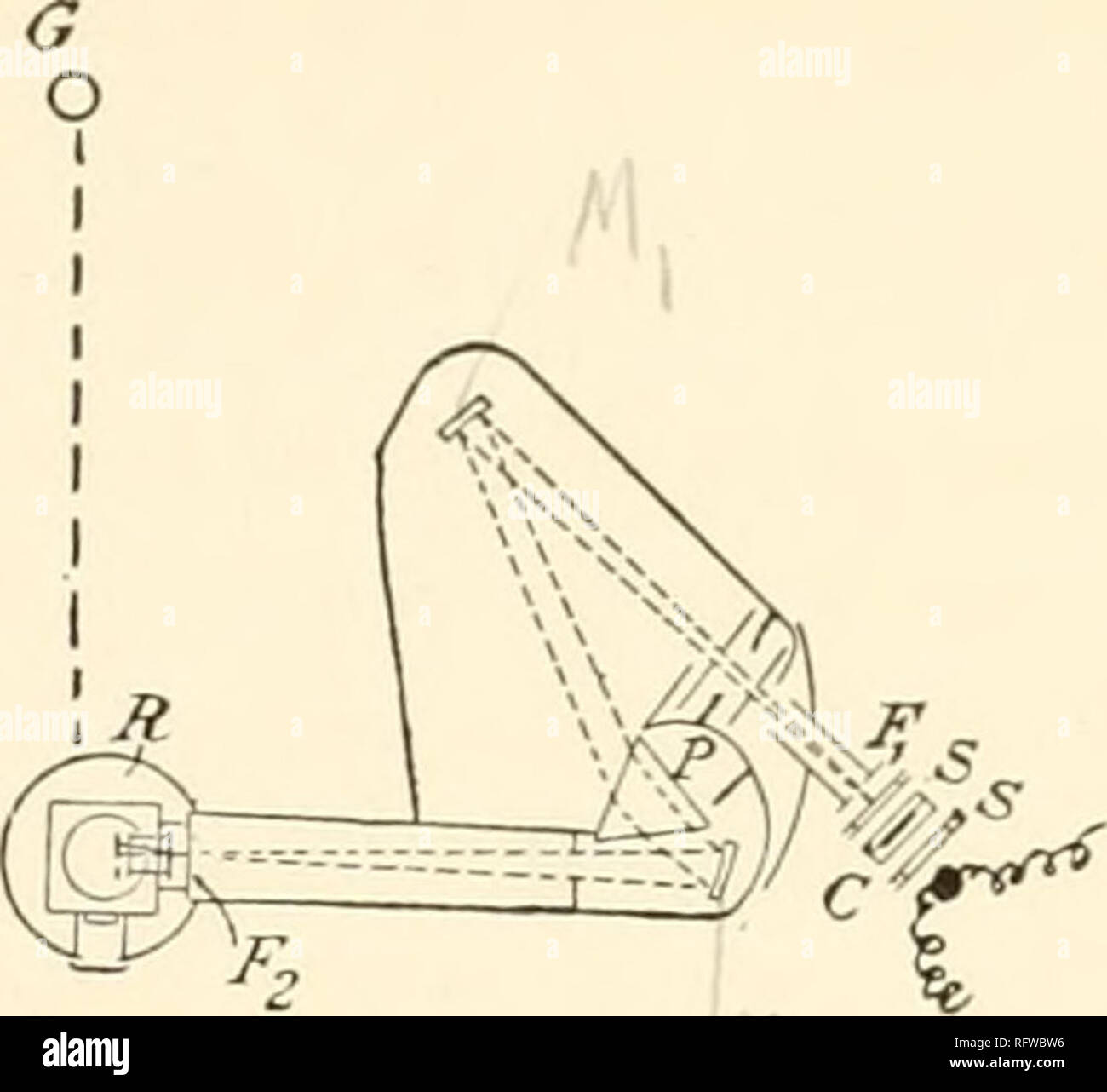
RMRFWBW6–. Carnegie Institution of Washington publication. . ^^ Fig. I. -Inner room containing radiometer R, spectrometer and prism P, exhaust pump G, lamps and scale L, and telescope T. C is the holder for the absorption cell. S is the shutter, H the Nernst lamp-heater.. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations may not perfectly resemble the original work.. Carnegie Institution of Washington. Washington, Carnegie Institution of Washington

RFKD5DDE–Identification of white powder raw material by FTIR method, advance technology without sample preparation high sensitivity and accuracy method, concept of pharmaceutical industrial laboratory
RMRJNCJY–. Bacterial photosynthesis. Bacteria -- Physiology; Photosynthesis. ACTION SPECTRUM I r^ —ABSORPTION I /T. ^ % 7000 8000 9000 10,000 WAVELENGTH (A) Fig. 4. Action spectrum of chromatophores from R. riihvum, taken at constant quanta/sec-cm2 incident on the sample. The absorption spectrum is shown in dotted lines for comparison. quanta/sec-cm2), and is shown in Fig. 4. Distortions still mask pos- sible contributions by the secondary peak. Further reduction of light intensity and/or concentration reduces the magnitude of the signal to nearly that of the noise observed in our spectrometer. Thus we
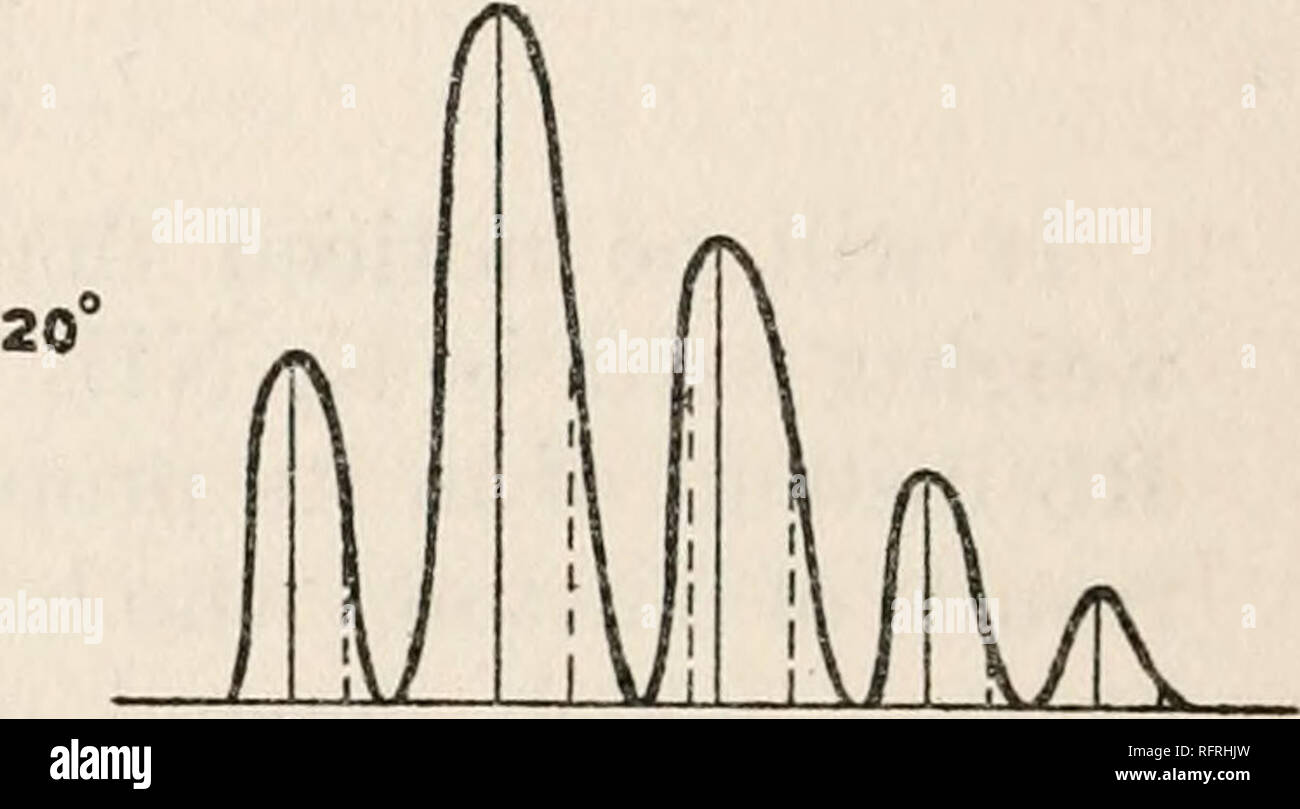
RMRFRHJW–. Carnegie Institution of Washington publication. A X. B C D E A -185 iA FIG. 56. B?C Cf D D EE A A FIG. 57. The crystal was mounted so as to cover a transverse slot in the copper rod. It could thus be illuminated either from the front, as shown above, or from behind by light transmitted through the slot. The latter arrangement was employed especially in the study of the absorption spectrum. When the substance, excited to fluorescence in the manner already described, was gradually cooled to the temperature of liquid air and the spectrum was observed through the Hilger spectrometer, the fol- lo
Search Results for Absorption spectrometer Stock Photos and Images (128)
Page 1 of 2