Quick filters:
Gravity cell battery Stock Photos and Images
 A well used car battery acid hydrometer which is used for checking wet cell battery electrolyte levels. Stock Photohttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/a-well-used-car-battery-acid-hydrometer-which-is-used-for-checking-wet-cell-battery-electrolyte-levels-image330895482.html
A well used car battery acid hydrometer which is used for checking wet cell battery electrolyte levels. Stock Photohttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/a-well-used-car-battery-acid-hydrometer-which-is-used-for-checking-wet-cell-battery-electrolyte-levels-image330895482.htmlRF2A69GFP–A well used car battery acid hydrometer which is used for checking wet cell battery electrolyte levels.
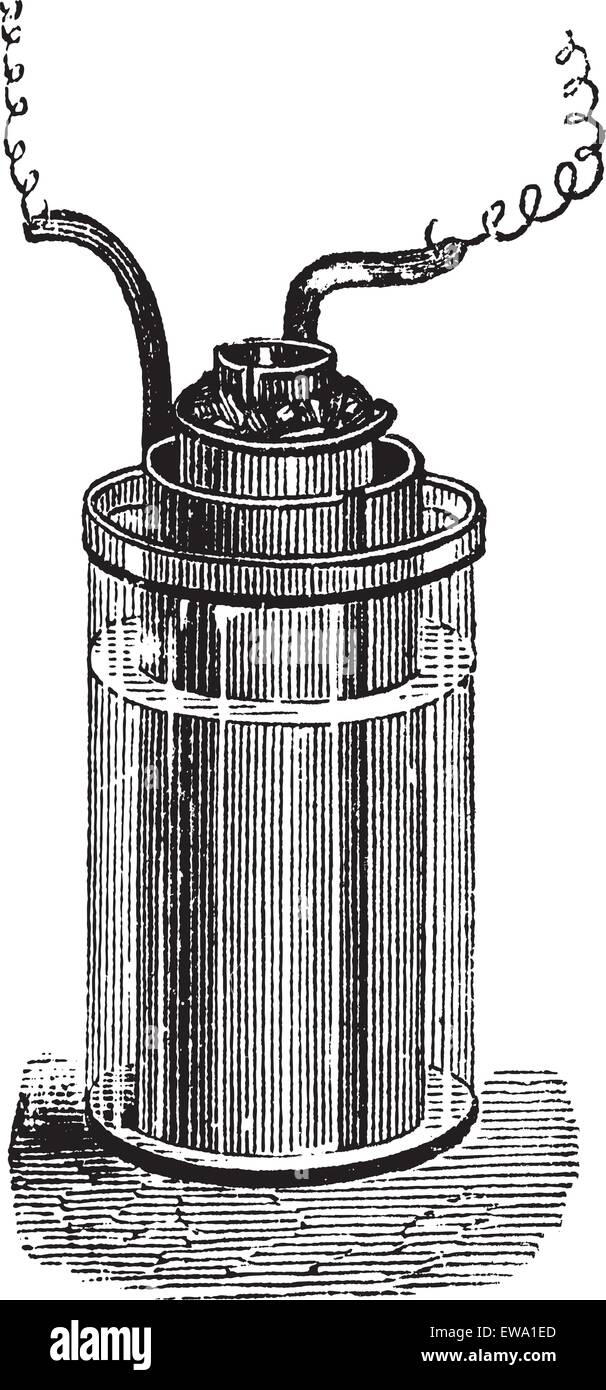 Daniell cell or gravity cell or crowfoot cell, vintage engraving. Old engraved illustration of Daniell cell, isolated on a white background. Stock Vectorhttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/stock-photo-daniell-cell-or-gravity-cell-or-crowfoot-cell-vintage-engraving-old-84428581.html
Daniell cell or gravity cell or crowfoot cell, vintage engraving. Old engraved illustration of Daniell cell, isolated on a white background. Stock Vectorhttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/stock-photo-daniell-cell-or-gravity-cell-or-crowfoot-cell-vintage-engraving-old-84428581.htmlRFEWA1ED–Daniell cell or gravity cell or crowfoot cell, vintage engraving. Old engraved illustration of Daniell cell, isolated on a white background.
 Callaud gravity cell:in 1860 the French inventor Callaud emproved the battery cell with a copper electrode and a zinc anode in a jar filled with copper sulfate and distilled water. His invention became the preferred battery for U.S. and U.K. telegraph systems. Stock Photohttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/callaud-gravity-cellin-1860-the-french-inventor-callaud-emproved-the-battery-cell-with-a-copper-electrode-and-a-zinc-anode-in-a-jar-filled-with-copper-sulfate-and-distilled-water-his-invention-became-the-preferred-battery-for-us-and-uk-telegraph-systems-image232013372.html
Callaud gravity cell:in 1860 the French inventor Callaud emproved the battery cell with a copper electrode and a zinc anode in a jar filled with copper sulfate and distilled water. His invention became the preferred battery for U.S. and U.K. telegraph systems. Stock Photohttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/callaud-gravity-cellin-1860-the-french-inventor-callaud-emproved-the-battery-cell-with-a-copper-electrode-and-a-zinc-anode-in-a-jar-filled-with-copper-sulfate-and-distilled-water-his-invention-became-the-preferred-battery-for-us-and-uk-telegraph-systems-image232013372.htmlRFRDD3BT–Callaud gravity cell:in 1860 the French inventor Callaud emproved the battery cell with a copper electrode and a zinc anode in a jar filled with copper sulfate and distilled water. His invention became the preferred battery for U.S. and U.K. telegraph systems.
 STAR TOWN - FEBRUARY 4: Model of Mir space station in Cosmonaut Training Center on February 4, 2012 in Star town near Moscow, Russia. Mir space statio Stock Photohttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/star-town-february-4-model-of-mir-space-station-in-cosmonaut-training-center-on-february-4-2012-in-star-town-near-moscow-russia-mir-space-statio-image604053362.html
STAR TOWN - FEBRUARY 4: Model of Mir space station in Cosmonaut Training Center on February 4, 2012 in Star town near Moscow, Russia. Mir space statio Stock Photohttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/star-town-february-4-model-of-mir-space-station-in-cosmonaut-training-center-on-february-4-2012-in-star-town-near-moscow-russia-mir-space-statio-image604053362.htmlRF2X2N06A–STAR TOWN - FEBRUARY 4: Model of Mir space station in Cosmonaut Training Center on February 4, 2012 in Star town near Moscow, Russia. Mir space statio
 Green battery glowing with plus and minus signs fully charged isolated on black background with copy space 3d illustration Stock Photohttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/green-battery-glowing-with-plus-and-minus-signs-fully-charged-isolated-on-black-background-with-copy-space-3d-illustration-image511426047.html
Green battery glowing with plus and minus signs fully charged isolated on black background with copy space 3d illustration Stock Photohttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/green-battery-glowing-with-plus-and-minus-signs-fully-charged-isolated-on-black-background-with-copy-space-3d-illustration-image511426047.htmlRF2MM1D3Y–Green battery glowing with plus and minus signs fully charged isolated on black background with copy space 3d illustration
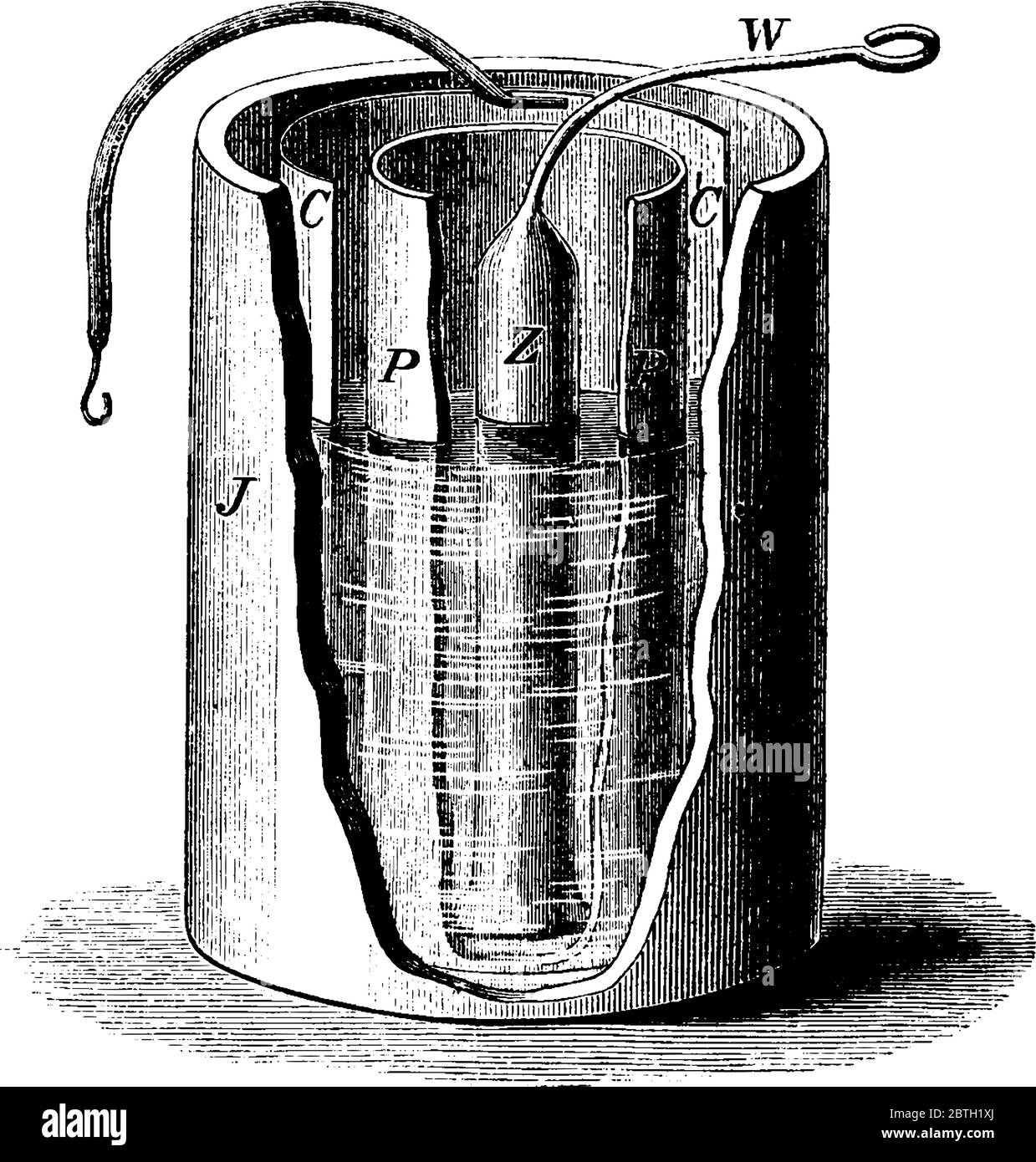 The Daniell cell was a great improvement over the voltaic pile used in the early days of battery development. The Daniell cell's theoretical voltage i Stock Vectorhttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/the-daniell-cell-was-a-great-improvement-over-the-voltaic-pile-used-in-the-early-days-of-battery-development-the-daniell-cells-theoretical-voltage-i-image359333818.html
The Daniell cell was a great improvement over the voltaic pile used in the early days of battery development. The Daniell cell's theoretical voltage i Stock Vectorhttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/the-daniell-cell-was-a-great-improvement-over-the-voltaic-pile-used-in-the-early-days-of-battery-development-the-daniell-cells-theoretical-voltage-i-image359333818.htmlRF2BTH1XJ–The Daniell cell was a great improvement over the voltaic pile used in the early days of battery development. The Daniell cell's theoretical voltage i
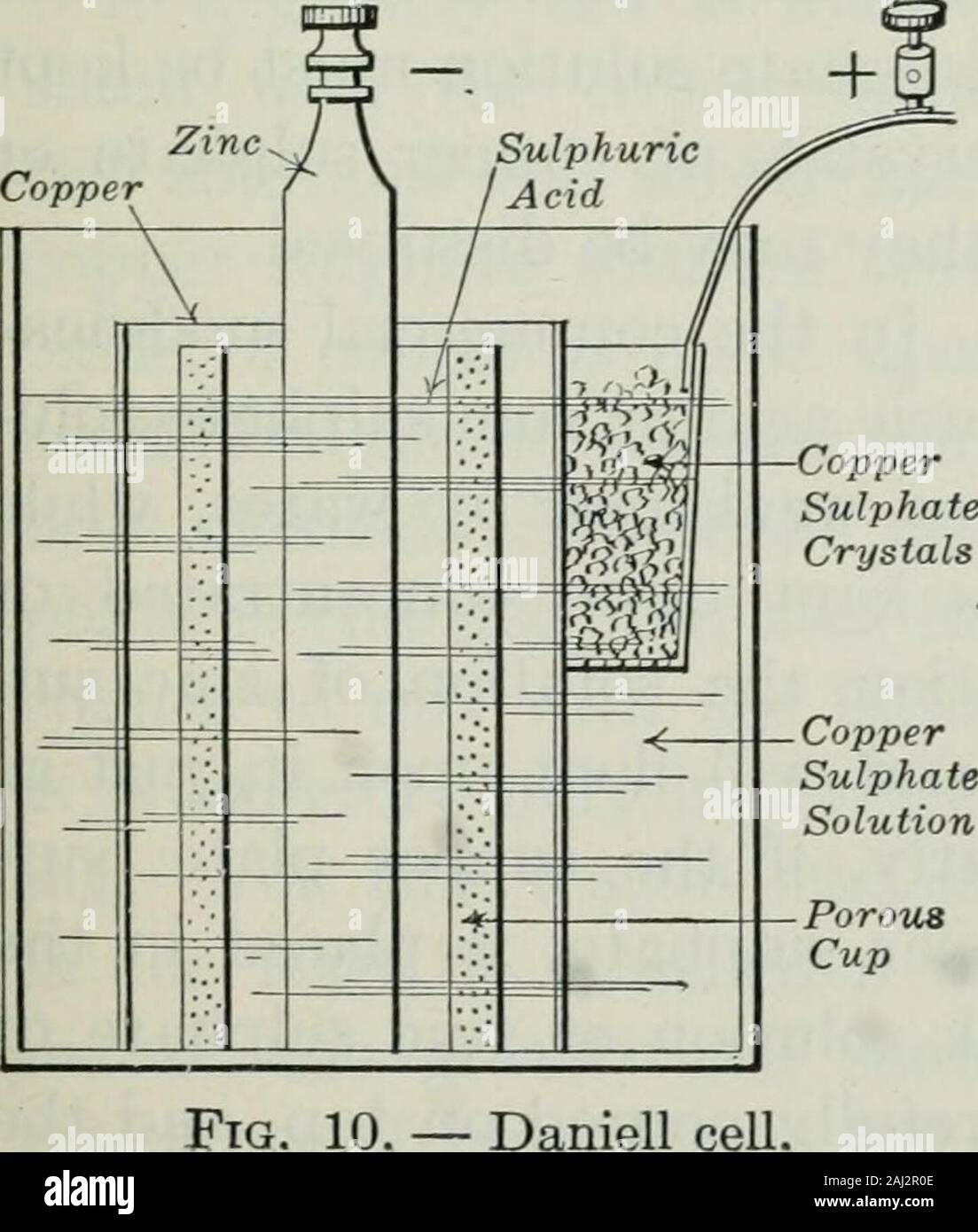 An elementary book on electricity and magnetism and their applications . mical depolariza-tion. Daniell cell. The thirdmethod of depolarizing introducesmore complicated chemical reac-tions, but concerning these wreneed not give much detail. Bythis method cells are constructedwhich give excellent results incontinuous service, and which are,therefore, called closed-circuit cells.One of these is probably the mostcommonly used of any form ofwet battery. This is the or-dinary gravity battery, or coppersulphate battery, which is so much used in telegraphy. The original Daniell cell(Fig. 10), from wh Stock Photohttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/an-elementary-book-on-electricity-and-magnetism-and-their-applications-mical-depolariza-tion-daniell-cell-the-thirdmethod-of-depolarizing-introducesmore-complicated-chemical-reac-tions-but-concerning-these-wreneed-not-give-much-detail-bythis-method-cells-are-constructedwhich-give-excellent-results-incontinuous-service-and-which-aretherefore-called-closed-circuit-cellsone-of-these-is-probably-the-mostcommonly-used-of-any-form-ofwet-battery-this-is-the-or-dinary-gravity-battery-or-coppersulphate-battery-which-is-so-much-used-in-telegraphy-the-original-daniell-cellfig-10-from-wh-image338122750.html
An elementary book on electricity and magnetism and their applications . mical depolariza-tion. Daniell cell. The thirdmethod of depolarizing introducesmore complicated chemical reac-tions, but concerning these wreneed not give much detail. Bythis method cells are constructedwhich give excellent results incontinuous service, and which are,therefore, called closed-circuit cells.One of these is probably the mostcommonly used of any form ofwet battery. This is the or-dinary gravity battery, or coppersulphate battery, which is so much used in telegraphy. The original Daniell cell(Fig. 10), from wh Stock Photohttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/an-elementary-book-on-electricity-and-magnetism-and-their-applications-mical-depolariza-tion-daniell-cell-the-thirdmethod-of-depolarizing-introducesmore-complicated-chemical-reac-tions-but-concerning-these-wreneed-not-give-much-detail-bythis-method-cells-are-constructedwhich-give-excellent-results-incontinuous-service-and-which-aretherefore-called-closed-circuit-cellsone-of-these-is-probably-the-mostcommonly-used-of-any-form-ofwet-battery-this-is-the-or-dinary-gravity-battery-or-coppersulphate-battery-which-is-so-much-used-in-telegraphy-the-original-daniell-cellfig-10-from-wh-image338122750.htmlRM2AJ2R0E–An elementary book on electricity and magnetism and their applications . mical depolariza-tion. Daniell cell. The thirdmethod of depolarizing introducesmore complicated chemical reac-tions, but concerning these wreneed not give much detail. Bythis method cells are constructedwhich give excellent results incontinuous service, and which are,therefore, called closed-circuit cells.One of these is probably the mostcommonly used of any form ofwet battery. This is the or-dinary gravity battery, or coppersulphate battery, which is so much used in telegraphy. The original Daniell cell(Fig. 10), from wh
 States that the particles present in a cell become proportionally larger with increasing size of the cell, vintage line drawing or engraving illustrat Stock Vectorhttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/states-that-the-particles-present-in-a-cell-become-proportionally-larger-with-increasing-size-of-the-cell-vintage-line-drawing-or-engraving-illustrat-image359329621.html
States that the particles present in a cell become proportionally larger with increasing size of the cell, vintage line drawing or engraving illustrat Stock Vectorhttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/states-that-the-particles-present-in-a-cell-become-proportionally-larger-with-increasing-size-of-the-cell-vintage-line-drawing-or-engraving-illustrat-image359329621.htmlRF2BTGTGN–States that the particles present in a cell become proportionally larger with increasing size of the cell, vintage line drawing or engraving illustrat
 accumulator dry battery secondary battery secondary cell storage cell storage device wet battery power positive negative Stock Photohttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/accumulator-dry-battery-secondary-battery-secondary-cell-storage-cell-image2947575.html
accumulator dry battery secondary battery secondary cell storage cell storage device wet battery power positive negative Stock Photohttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/accumulator-dry-battery-secondary-battery-secondary-cell-storage-cell-image2947575.htmlRMAPR9F8–accumulator dry battery secondary battery secondary cell storage cell storage device wet battery power positive negative
 A well used car battery acid hydrometer which is used for checking wet cell battery electrolyte levels. Stock Photohttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/a-well-used-car-battery-acid-hydrometer-which-is-used-for-checking-wet-cell-battery-electrolyte-levels-image330895478.html
A well used car battery acid hydrometer which is used for checking wet cell battery electrolyte levels. Stock Photohttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/a-well-used-car-battery-acid-hydrometer-which-is-used-for-checking-wet-cell-battery-electrolyte-levels-image330895478.htmlRF2A69GFJ–A well used car battery acid hydrometer which is used for checking wet cell battery electrolyte levels.
 Green battery glowing with plus and minus signs fully charged on black podium with green accent lights 3d illustration Stock Photohttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/green-battery-glowing-with-plus-and-minus-signs-fully-charged-on-black-podium-with-green-accent-lights-3d-illustration-image511426045.html
Green battery glowing with plus and minus signs fully charged on black podium with green accent lights 3d illustration Stock Photohttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/green-battery-glowing-with-plus-and-minus-signs-fully-charged-on-black-podium-with-green-accent-lights-3d-illustration-image511426045.htmlRF2MM1D3W–Green battery glowing with plus and minus signs fully charged on black podium with green accent lights 3d illustration
 The Pharmaceutical era . furicacid formed diffuses through the liquid and dissolves thezinc plate, this reaction being the source of the energydeveloped by the battery. In order to keep the cell in good working order it isnecessary to draw off the soluriou of zinc sulfate fromtime to time and replace it with water, and also to keepthe- bottom Niippliid with crystals of copiK<r sulfate. The gravity cell, because of its cbeapiies.s. continuousaction and slight expense of maintenance, is the one gen-erally eiu|doyed upon telegroph lines, and similar cir- CIlilN. The Denitll Cell.- The Daniell Stock Photohttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/the-pharmaceutical-era-furicacid-formed-diffuses-through-the-liquid-and-dissolves-thezinc-plate-this-reaction-being-the-source-of-the-energydeveloped-by-the-battery-in-order-to-keep-the-cell-in-good-working-order-it-isnecessary-to-draw-off-the-soluriou-of-zinc-sulfate-fromtime-to-time-and-replace-it-with-water-and-also-to-keepthe-bottom-niippliid-with-crystals-of-copikltr-sulfate-the-gravity-cell-because-of-its-cbeapiiess-continuousaction-and-slight-expense-of-maintenance-is-the-one-gen-erally-eiudoyed-upon-telegroph-lines-and-similar-cir-cililn-the-denitll-cell-the-daniell-image339247764.html
The Pharmaceutical era . furicacid formed diffuses through the liquid and dissolves thezinc plate, this reaction being the source of the energydeveloped by the battery. In order to keep the cell in good working order it isnecessary to draw off the soluriou of zinc sulfate fromtime to time and replace it with water, and also to keepthe- bottom Niippliid with crystals of copiK<r sulfate. The gravity cell, because of its cbeapiies.s. continuousaction and slight expense of maintenance, is the one gen-erally eiu|doyed upon telegroph lines, and similar cir- CIlilN. The Denitll Cell.- The Daniell Stock Photohttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/the-pharmaceutical-era-furicacid-formed-diffuses-through-the-liquid-and-dissolves-thezinc-plate-this-reaction-being-the-source-of-the-energydeveloped-by-the-battery-in-order-to-keep-the-cell-in-good-working-order-it-isnecessary-to-draw-off-the-soluriou-of-zinc-sulfate-fromtime-to-time-and-replace-it-with-water-and-also-to-keepthe-bottom-niippliid-with-crystals-of-copikltr-sulfate-the-gravity-cell-because-of-its-cbeapiiess-continuousaction-and-slight-expense-of-maintenance-is-the-one-gen-erally-eiudoyed-upon-telegroph-lines-and-similar-cir-cililn-the-denitll-cell-the-daniell-image339247764.htmlRM2AKX1YG–The Pharmaceutical era . furicacid formed diffuses through the liquid and dissolves thezinc plate, this reaction being the source of the energydeveloped by the battery. In order to keep the cell in good working order it isnecessary to draw off the soluriou of zinc sulfate fromtime to time and replace it with water, and also to keepthe- bottom Niippliid with crystals of copiK<r sulfate. The gravity cell, because of its cbeapiies.s. continuousaction and slight expense of maintenance, is the one gen-erally eiu|doyed upon telegroph lines, and similar cir- CIlilN. The Denitll Cell.- The Daniell
 Green battery glowing with plus and minus sign fully charged floating over black podium with green accent lights 3d illustration Stock Photohttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/green-battery-glowing-with-plus-and-minus-sign-fully-charged-floating-over-black-podium-with-green-accent-lights-3d-illustration-image511426046.html
Green battery glowing with plus and minus sign fully charged floating over black podium with green accent lights 3d illustration Stock Photohttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/green-battery-glowing-with-plus-and-minus-sign-fully-charged-floating-over-black-podium-with-green-accent-lights-3d-illustration-image511426046.htmlRF2MM1D3X–Green battery glowing with plus and minus sign fully charged floating over black podium with green accent lights 3d illustration
 The electro-therapeutic guide, or, A thousand questions asked and answered . The Greuet Acid Cell Nested Acid Cell Galvanic Battery. The Blue Stone or Crow Foot, or Gravity Cell M, II If What is a Battery? A battery is composed of two or more cells. Those known as dry cellare best for portable batteries, for the reason that they are cheap, compact,clean and efficient and require no attention. In fact there is no such thingas a drycell. They are calleddry for want of a more expressive term.They contain a minimum amount of fluid which combines the chemicals toform the excitant in the hermeticall Stock Photohttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/the-electro-therapeutic-guide-or-a-thousand-questions-asked-and-answered-the-greuet-acid-cell-nested-acid-cell-galvanic-battery-the-blue-stone-or-crow-foot-or-gravity-cell-m-ii-if-what-is-a-battery-a-battery-is-composed-of-two-or-more-cells-those-known-as-dry-cellare-best-for-portable-batteries-for-the-reason-that-they-are-cheap-compactclean-and-efficient-and-require-no-attention-in-fact-there-is-no-such-thingas-a-drycell-they-are-calleddry-for-want-of-a-more-expressive-termthey-contain-a-minimum-amount-of-fluid-which-combines-the-chemicals-toform-the-excitant-in-the-hermeticall-image342684124.html
The electro-therapeutic guide, or, A thousand questions asked and answered . The Greuet Acid Cell Nested Acid Cell Galvanic Battery. The Blue Stone or Crow Foot, or Gravity Cell M, II If What is a Battery? A battery is composed of two or more cells. Those known as dry cellare best for portable batteries, for the reason that they are cheap, compact,clean and efficient and require no attention. In fact there is no such thingas a drycell. They are calleddry for want of a more expressive term.They contain a minimum amount of fluid which combines the chemicals toform the excitant in the hermeticall Stock Photohttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/the-electro-therapeutic-guide-or-a-thousand-questions-asked-and-answered-the-greuet-acid-cell-nested-acid-cell-galvanic-battery-the-blue-stone-or-crow-foot-or-gravity-cell-m-ii-if-what-is-a-battery-a-battery-is-composed-of-two-or-more-cells-those-known-as-dry-cellare-best-for-portable-batteries-for-the-reason-that-they-are-cheap-compactclean-and-efficient-and-require-no-attention-in-fact-there-is-no-such-thingas-a-drycell-they-are-calleddry-for-want-of-a-more-expressive-termthey-contain-a-minimum-amount-of-fluid-which-combines-the-chemicals-toform-the-excitant-in-the-hermeticall-image342684124.htmlRM2AWEH2M–The electro-therapeutic guide, or, A thousand questions asked and answered . The Greuet Acid Cell Nested Acid Cell Galvanic Battery. The Blue Stone or Crow Foot, or Gravity Cell M, II If What is a Battery? A battery is composed of two or more cells. Those known as dry cellare best for portable batteries, for the reason that they are cheap, compact,clean and efficient and require no attention. In fact there is no such thingas a drycell. They are calleddry for want of a more expressive term.They contain a minimum amount of fluid which combines the chemicals toform the excitant in the hermeticall
 The American watchmaker and jeweler; an encyclopedia for the horologist, jeweler, gold and silversmith .. . Fig. 118. The Gravity Battery. In consequence of the trouble caused by theprecipitation of the copper on the porous cell in the Daniell battery,Cromwell F. Varley, in 1854, while experimenting, found that the differ-?ence in specific gravity between solutions of sulphate of copper and sul-phate of zinc was sufficient in itself to entirely separate them, the coppersolution lying at the bottom of the cell, and the zinc solution remaining 130 Electro-Plating, Etc.. Fig. 119. superposed upon Stock Photohttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/the-american-watchmaker-and-jeweler-an-encyclopedia-for-the-horologist-jeweler-gold-and-silversmith-fig-118-the-gravity-battery-in-consequence-of-the-trouble-caused-by-theprecipitation-of-the-copper-on-the-porous-cell-in-the-daniell-batterycromwell-f-varley-in-1854-while-experimenting-found-that-the-differ-ence-in-specific-gravity-between-solutions-of-sulphate-of-copper-and-sul-phate-of-zinc-was-sufficient-in-itself-to-entirely-separate-them-the-coppersolution-lying-at-the-bottom-of-the-cell-and-the-zinc-solution-remaining-130-electro-plating-etc-fig-119-superposed-upon-image338934620.html
The American watchmaker and jeweler; an encyclopedia for the horologist, jeweler, gold and silversmith .. . Fig. 118. The Gravity Battery. In consequence of the trouble caused by theprecipitation of the copper on the porous cell in the Daniell battery,Cromwell F. Varley, in 1854, while experimenting, found that the differ-?ence in specific gravity between solutions of sulphate of copper and sul-phate of zinc was sufficient in itself to entirely separate them, the coppersolution lying at the bottom of the cell, and the zinc solution remaining 130 Electro-Plating, Etc.. Fig. 119. superposed upon Stock Photohttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/the-american-watchmaker-and-jeweler-an-encyclopedia-for-the-horologist-jeweler-gold-and-silversmith-fig-118-the-gravity-battery-in-consequence-of-the-trouble-caused-by-theprecipitation-of-the-copper-on-the-porous-cell-in-the-daniell-batterycromwell-f-varley-in-1854-while-experimenting-found-that-the-differ-ence-in-specific-gravity-between-solutions-of-sulphate-of-copper-and-sul-phate-of-zinc-was-sufficient-in-itself-to-entirely-separate-them-the-coppersolution-lying-at-the-bottom-of-the-cell-and-the-zinc-solution-remaining-130-electro-plating-etc-fig-119-superposed-upon-image338934620.htmlRM2AKBPFT–The American watchmaker and jeweler; an encyclopedia for the horologist, jeweler, gold and silversmith .. . Fig. 118. The Gravity Battery. In consequence of the trouble caused by theprecipitation of the copper on the porous cell in the Daniell battery,Cromwell F. Varley, in 1854, while experimenting, found that the differ-?ence in specific gravity between solutions of sulphate of copper and sul-phate of zinc was sufficient in itself to entirely separate them, the coppersolution lying at the bottom of the cell, and the zinc solution remaining 130 Electro-Plating, Etc.. Fig. 119. superposed upon
 . The Pharmaceutical era. ncreases inbulk. The free sulfuricacid formed diffuses through the liquid and dissolves thezinc plate, this reaction being the source of the energydeveloped by the battery. In order to keep the cell in good working order it isnecessary to draw off the solution of zinc sulfate fromtime to time and replace it with water, and also to keepthe bottom supplied with crystals of copper sulfate. The gravity cell, because of its cheapness, continuousaction and slight expense of maintenance, is the one gen-erally employed upon telegraph lines, and similar cir-cuits. The Daniell Stock Photohttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/the-pharmaceutical-era-ncreases-inbulk-the-free-sulfuricacid-formed-diffuses-through-the-liquid-and-dissolves-thezinc-plate-this-reaction-being-the-source-of-the-energydeveloped-by-the-battery-in-order-to-keep-the-cell-in-good-working-order-it-isnecessary-to-draw-off-the-solution-of-zinc-sulfate-fromtime-to-time-and-replace-it-with-water-and-also-to-keepthe-bottom-supplied-with-crystals-of-copper-sulfate-the-gravity-cell-because-of-its-cheapness-continuousaction-and-slight-expense-of-maintenance-is-the-one-gen-erally-employed-upon-telegraph-lines-and-similar-cir-cuits-the-daniell-image370370233.html
. The Pharmaceutical era. ncreases inbulk. The free sulfuricacid formed diffuses through the liquid and dissolves thezinc plate, this reaction being the source of the energydeveloped by the battery. In order to keep the cell in good working order it isnecessary to draw off the solution of zinc sulfate fromtime to time and replace it with water, and also to keepthe bottom supplied with crystals of copper sulfate. The gravity cell, because of its cheapness, continuousaction and slight expense of maintenance, is the one gen-erally employed upon telegraph lines, and similar cir-cuits. The Daniell Stock Photohttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/the-pharmaceutical-era-ncreases-inbulk-the-free-sulfuricacid-formed-diffuses-through-the-liquid-and-dissolves-thezinc-plate-this-reaction-being-the-source-of-the-energydeveloped-by-the-battery-in-order-to-keep-the-cell-in-good-working-order-it-isnecessary-to-draw-off-the-solution-of-zinc-sulfate-fromtime-to-time-and-replace-it-with-water-and-also-to-keepthe-bottom-supplied-with-crystals-of-copper-sulfate-the-gravity-cell-because-of-its-cheapness-continuousaction-and-slight-expense-of-maintenance-is-the-one-gen-erally-employed-upon-telegraph-lines-and-similar-cir-cuits-the-daniell-image370370233.htmlRM2CEFR09–. The Pharmaceutical era. ncreases inbulk. The free sulfuricacid formed diffuses through the liquid and dissolves thezinc plate, this reaction being the source of the energydeveloped by the battery. In order to keep the cell in good working order it isnecessary to draw off the solution of zinc sulfate fromtime to time and replace it with water, and also to keepthe bottom supplied with crystals of copper sulfate. The gravity cell, because of its cheapness, continuousaction and slight expense of maintenance, is the one gen-erally employed upon telegraph lines, and similar cir-cuits. The Daniell
 . Railway and locomotive engineering : a practical journal of railway motive power and rolling stock . ell [h gravity of tin electrolyti ho ild never be allow dlo fall below 1.15, and 1.185 to M95 iaill. general practice. DESCRIPTION. The plates in each cell oi battery an join, d together by burning the plate terminal tnon bus I111 This burning is simply awelding in which the heat of a blowpipe is used to weld the parts together. The plates are then assembled inwater-light, acid-proof tanks or jars.tll.iss jar^ are usually employed for thesmall sizi -. although rubber has comeinto use of late. Stock Photohttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/railway-and-locomotive-engineering-a-practical-journal-of-railway-motive-power-and-rolling-stock-ell-h-gravity-of-tin-electrolyti-ho-ild-never-be-allow-dlo-fall-below-115-and-1185-to-m95-iaill-general-practice-description-the-plates-in-each-cell-oi-battery-an-join-d-together-by-burning-the-plate-terminal-tnon-bus-i111-this-burning-is-simply-awelding-in-which-the-heat-of-a-blowpipe-is-used-to-weld-the-parts-together-the-plates-are-then-assembled-inwater-light-acid-proof-tanks-or-jarstlliss-jar-are-usually-employed-for-thesmall-sizi-although-rubber-has-comeinto-use-of-late-image371837886.html
. Railway and locomotive engineering : a practical journal of railway motive power and rolling stock . ell [h gravity of tin electrolyti ho ild never be allow dlo fall below 1.15, and 1.185 to M95 iaill. general practice. DESCRIPTION. The plates in each cell oi battery an join, d together by burning the plate terminal tnon bus I111 This burning is simply awelding in which the heat of a blowpipe is used to weld the parts together. The plates are then assembled inwater-light, acid-proof tanks or jars.tll.iss jar^ are usually employed for thesmall sizi -. although rubber has comeinto use of late. Stock Photohttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/railway-and-locomotive-engineering-a-practical-journal-of-railway-motive-power-and-rolling-stock-ell-h-gravity-of-tin-electrolyti-ho-ild-never-be-allow-dlo-fall-below-115-and-1185-to-m95-iaill-general-practice-description-the-plates-in-each-cell-oi-battery-an-join-d-together-by-burning-the-plate-terminal-tnon-bus-i111-this-burning-is-simply-awelding-in-which-the-heat-of-a-blowpipe-is-used-to-weld-the-parts-together-the-plates-are-then-assembled-inwater-light-acid-proof-tanks-or-jarstlliss-jar-are-usually-employed-for-thesmall-sizi-although-rubber-has-comeinto-use-of-late-image371837886.htmlRM2CGXK0E–. Railway and locomotive engineering : a practical journal of railway motive power and rolling stock . ell [h gravity of tin electrolyti ho ild never be allow dlo fall below 1.15, and 1.185 to M95 iaill. general practice. DESCRIPTION. The plates in each cell oi battery an join, d together by burning the plate terminal tnon bus I111 This burning is simply awelding in which the heat of a blowpipe is used to weld the parts together. The plates are then assembled inwater-light, acid-proof tanks or jars.tll.iss jar^ are usually employed for thesmall sizi -. although rubber has comeinto use of late.
 . Storage battery manual, including principles of storage battery construction and design, with the application of storage of batteries to the naval service . has been reduced to thepoint such that little efficiency is obtained with further working of theplates. In other words, plates which have become lean have practicallyreached the end of their useful life. There is shown in Fig. 5 an illustrationof a lean plate. Definitions and Nomenclatuee of Pakts 19 Maximum Gravity.—The highest specific gravity attained by the electro-lyte of the cell through continued charging, thus indicating that no Stock Photohttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/storage-battery-manual-including-principles-of-storage-battery-construction-and-design-with-the-application-of-storage-of-batteries-to-the-naval-service-has-been-reduced-to-thepoint-such-that-little-efficiency-is-obtained-with-further-working-of-theplates-in-other-words-plates-which-have-become-lean-have-practicallyreached-the-end-of-their-useful-life-there-is-shown-in-fig-5-an-illustrationof-a-lean-plate-definitions-and-nomenclatuee-of-pakts-19-maximum-gravitythe-highest-specific-gravity-attained-by-the-electro-lyte-of-the-cell-through-continued-charging-thus-indicating-that-no-image370169030.html
. Storage battery manual, including principles of storage battery construction and design, with the application of storage of batteries to the naval service . has been reduced to thepoint such that little efficiency is obtained with further working of theplates. In other words, plates which have become lean have practicallyreached the end of their useful life. There is shown in Fig. 5 an illustrationof a lean plate. Definitions and Nomenclatuee of Pakts 19 Maximum Gravity.—The highest specific gravity attained by the electro-lyte of the cell through continued charging, thus indicating that no Stock Photohttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/storage-battery-manual-including-principles-of-storage-battery-construction-and-design-with-the-application-of-storage-of-batteries-to-the-naval-service-has-been-reduced-to-thepoint-such-that-little-efficiency-is-obtained-with-further-working-of-theplates-in-other-words-plates-which-have-become-lean-have-practicallyreached-the-end-of-their-useful-life-there-is-shown-in-fig-5-an-illustrationof-a-lean-plate-definitions-and-nomenclatuee-of-pakts-19-maximum-gravitythe-highest-specific-gravity-attained-by-the-electro-lyte-of-the-cell-through-continued-charging-thus-indicating-that-no-image370169030.htmlRM2CE6JAE–. Storage battery manual, including principles of storage battery construction and design, with the application of storage of batteries to the naval service . has been reduced to thepoint such that little efficiency is obtained with further working of theplates. In other words, plates which have become lean have practicallyreached the end of their useful life. There is shown in Fig. 5 an illustrationof a lean plate. Definitions and Nomenclatuee of Pakts 19 Maximum Gravity.—The highest specific gravity attained by the electro-lyte of the cell through continued charging, thus indicating that no
 . Storage battery manual, including principles of storage battery construction and design, with the application of storage of batteries to the naval service . lyte is practically directly pro-portional to the ampere-hours of discharge, and for this reason the specificgravity readings are in general more to be relied upon in determining thetrue state of discharge of the cell than are the voltage readings. However,in order to intelligently operate the storage battery cell routine readingsshould be taken of the voltage as well as the specific gravity. CHAPTEE Hi.DEFINITIONS AND NOMENCLATURE OF PA Stock Photohttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/storage-battery-manual-including-principles-of-storage-battery-construction-and-design-with-the-application-of-storage-of-batteries-to-the-naval-service-lyte-is-practically-directly-pro-portional-to-the-ampere-hours-of-discharge-and-for-this-reason-the-specificgravity-readings-are-in-general-more-to-be-relied-upon-in-determining-thetrue-state-of-discharge-of-the-cell-than-are-the-voltage-readings-howeverin-order-to-intelligently-operate-the-storage-battery-cell-routine-readingsshould-be-taken-of-the-voltage-as-well-as-the-specific-gravity-chaptee-hidefinitions-and-nomenclature-of-pa-image370168991.html
. Storage battery manual, including principles of storage battery construction and design, with the application of storage of batteries to the naval service . lyte is practically directly pro-portional to the ampere-hours of discharge, and for this reason the specificgravity readings are in general more to be relied upon in determining thetrue state of discharge of the cell than are the voltage readings. However,in order to intelligently operate the storage battery cell routine readingsshould be taken of the voltage as well as the specific gravity. CHAPTEE Hi.DEFINITIONS AND NOMENCLATURE OF PA Stock Photohttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/storage-battery-manual-including-principles-of-storage-battery-construction-and-design-with-the-application-of-storage-of-batteries-to-the-naval-service-lyte-is-practically-directly-pro-portional-to-the-ampere-hours-of-discharge-and-for-this-reason-the-specificgravity-readings-are-in-general-more-to-be-relied-upon-in-determining-thetrue-state-of-discharge-of-the-cell-than-are-the-voltage-readings-howeverin-order-to-intelligently-operate-the-storage-battery-cell-routine-readingsshould-be-taken-of-the-voltage-as-well-as-the-specific-gravity-chaptee-hidefinitions-and-nomenclature-of-pa-image370168991.htmlRM2CE6J93–. Storage battery manual, including principles of storage battery construction and design, with the application of storage of batteries to the naval service . lyte is practically directly pro-portional to the ampere-hours of discharge, and for this reason the specificgravity readings are in general more to be relied upon in determining thetrue state of discharge of the cell than are the voltage readings. However,in order to intelligently operate the storage battery cell routine readingsshould be taken of the voltage as well as the specific gravity. CHAPTEE Hi.DEFINITIONS AND NOMENCLATURE OF PA
 . Storage battery manual, including principles of storage battery construction and design, with the application of storage of batteries to the naval service . d as follows: First.—See that the exterior of the battery is thoroughly clean. Second.—Make inspection for loose or defective connectors and terminalposts, poor lead-burning, defective sealing of the cell cover, height of theelectrolyte in the cell and note how much water is required to bring it to theproper level over the tops of the plates and separators, specific gravity of theelectrolyte, and cracked jar, cover or other broken parts. Stock Photohttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/storage-battery-manual-including-principles-of-storage-battery-construction-and-design-with-the-application-of-storage-of-batteries-to-the-naval-service-d-as-follows-firstsee-that-the-exterior-of-the-battery-is-thoroughly-clean-secondmake-inspection-for-loose-or-defective-connectors-and-terminalposts-poor-lead-burning-defective-sealing-of-the-cell-cover-height-of-theelectrolyte-in-the-cell-and-note-how-much-water-is-required-to-bring-it-to-theproper-level-over-the-tops-of-the-plates-and-separators-specific-gravity-of-theelectrolyte-and-cracked-jar-cover-or-other-broken-parts-image370163742.html
. Storage battery manual, including principles of storage battery construction and design, with the application of storage of batteries to the naval service . d as follows: First.—See that the exterior of the battery is thoroughly clean. Second.—Make inspection for loose or defective connectors and terminalposts, poor lead-burning, defective sealing of the cell cover, height of theelectrolyte in the cell and note how much water is required to bring it to theproper level over the tops of the plates and separators, specific gravity of theelectrolyte, and cracked jar, cover or other broken parts. Stock Photohttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/storage-battery-manual-including-principles-of-storage-battery-construction-and-design-with-the-application-of-storage-of-batteries-to-the-naval-service-d-as-follows-firstsee-that-the-exterior-of-the-battery-is-thoroughly-clean-secondmake-inspection-for-loose-or-defective-connectors-and-terminalposts-poor-lead-burning-defective-sealing-of-the-cell-cover-height-of-theelectrolyte-in-the-cell-and-note-how-much-water-is-required-to-bring-it-to-theproper-level-over-the-tops-of-the-plates-and-separators-specific-gravity-of-theelectrolyte-and-cracked-jar-cover-or-other-broken-parts-image370163742.htmlRM2CE6BHJ–. Storage battery manual, including principles of storage battery construction and design, with the application of storage of batteries to the naval service . d as follows: First.—See that the exterior of the battery is thoroughly clean. Second.—Make inspection for loose or defective connectors and terminalposts, poor lead-burning, defective sealing of the cell cover, height of theelectrolyte in the cell and note how much water is required to bring it to theproper level over the tops of the plates and separators, specific gravity of theelectrolyte, and cracked jar, cover or other broken parts.
 . Storage battery manual, including principles of storage battery construction and design, with the application of storage of batteries to the naval service . one complete batteryof each type and to conduct capacity tests on it. The procedure for con-ducting such tests is in general as follows: 1. The following readings taken while battery is on charge and just priorto discharging: (a) Voltage of each cell. (b) Specific gravity of each cell. (c) Temperature of each cell. 2. Cut the battery in on discharge, regulate the current to the propervalue as quickly as possible. 3. After discharge has r Stock Photohttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/storage-battery-manual-including-principles-of-storage-battery-construction-and-design-with-the-application-of-storage-of-batteries-to-the-naval-service-one-complete-batteryof-each-type-and-to-conduct-capacity-tests-on-it-the-procedure-for-con-ducting-such-tests-is-in-general-as-follows-1-the-following-readings-taken-while-battery-is-on-charge-and-just-priorto-discharging-a-voltage-of-each-cell-b-specific-gravity-of-each-cell-c-temperature-of-each-cell-2-cut-the-battery-in-on-discharge-regulate-the-current-to-the-propervalue-as-quickly-as-possible-3-after-discharge-has-r-image370163005.html
. Storage battery manual, including principles of storage battery construction and design, with the application of storage of batteries to the naval service . one complete batteryof each type and to conduct capacity tests on it. The procedure for con-ducting such tests is in general as follows: 1. The following readings taken while battery is on charge and just priorto discharging: (a) Voltage of each cell. (b) Specific gravity of each cell. (c) Temperature of each cell. 2. Cut the battery in on discharge, regulate the current to the propervalue as quickly as possible. 3. After discharge has r Stock Photohttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/storage-battery-manual-including-principles-of-storage-battery-construction-and-design-with-the-application-of-storage-of-batteries-to-the-naval-service-one-complete-batteryof-each-type-and-to-conduct-capacity-tests-on-it-the-procedure-for-con-ducting-such-tests-is-in-general-as-follows-1-the-following-readings-taken-while-battery-is-on-charge-and-just-priorto-discharging-a-voltage-of-each-cell-b-specific-gravity-of-each-cell-c-temperature-of-each-cell-2-cut-the-battery-in-on-discharge-regulate-the-current-to-the-propervalue-as-quickly-as-possible-3-after-discharge-has-r-image370163005.htmlRM2CE6AK9–. Storage battery manual, including principles of storage battery construction and design, with the application of storage of batteries to the naval service . one complete batteryof each type and to conduct capacity tests on it. The procedure for con-ducting such tests is in general as follows: 1. The following readings taken while battery is on charge and just priorto discharging: (a) Voltage of each cell. (b) Specific gravity of each cell. (c) Temperature of each cell. 2. Cut the battery in on discharge, regulate the current to the propervalue as quickly as possible. 3. After discharge has r
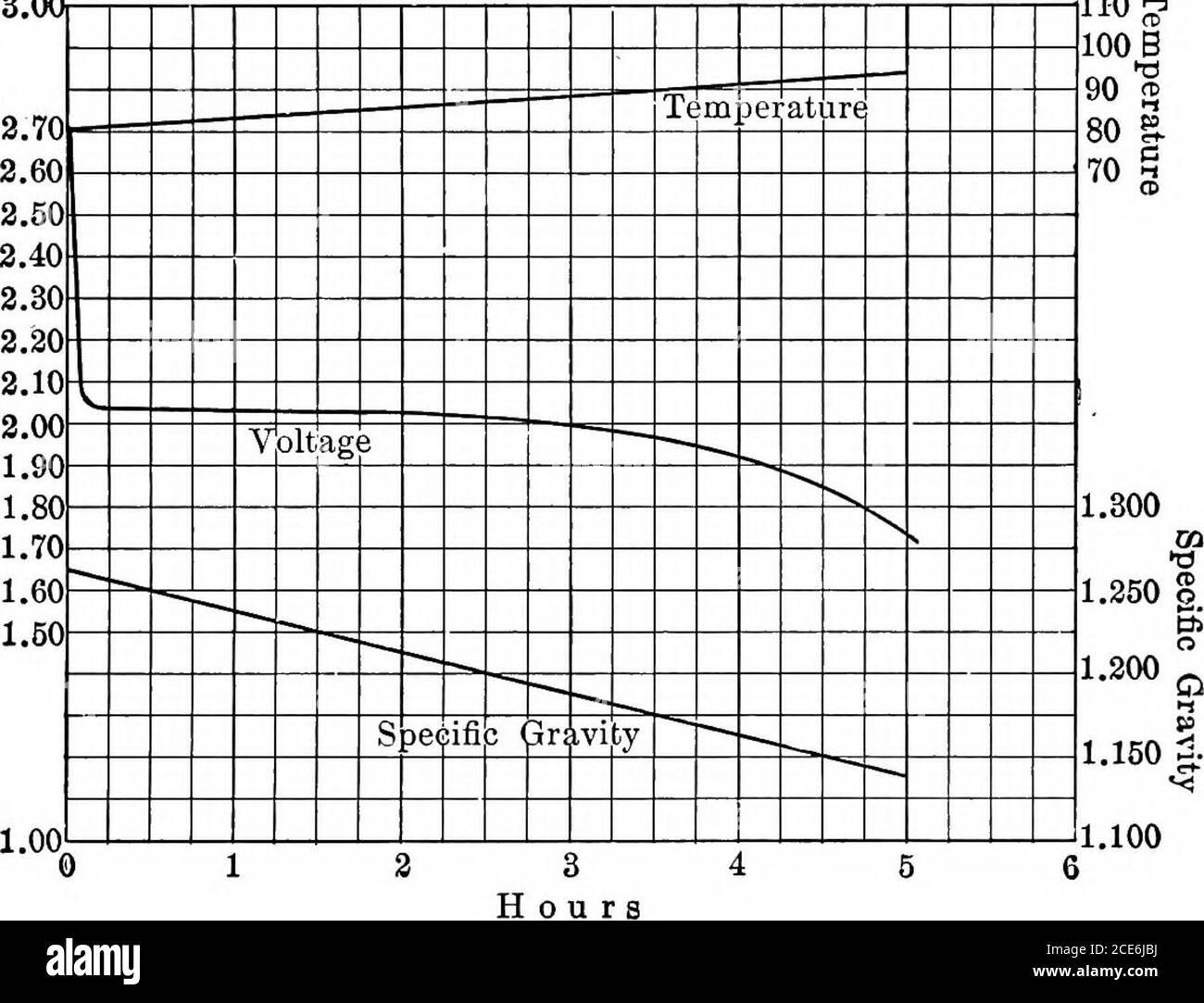 . Storage battery manual, including principles of storage battery construction and design, with the application of storage of batteries to the naval service . is a rapid increase in the cell voltage, until 2.65to 2.70 volts is reached and at which time there is practically no lead-sulphateremaining in the plates, hence the specific gravity of the electrolyte ceases torise and the cell may be said to be in a fully charged condition. If the charge The Lead-Acid Storage Battery Cell 11 be continued beyond this point the temperature of the cell rises rapidly andthe cell in reality resolves itself Stock Photohttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/storage-battery-manual-including-principles-of-storage-battery-construction-and-design-with-the-application-of-storage-of-batteries-to-the-naval-service-is-a-rapid-increase-in-the-cell-voltage-until-265to-270-volts-is-reached-and-at-which-time-there-is-practically-no-lead-sulphateremaining-in-the-plates-hence-the-specific-gravity-of-the-electrolyte-ceases-torise-and-the-cell-may-be-said-to-be-in-a-fully-charged-condition-if-the-charge-the-lead-acid-storage-battery-cell-11-be-continued-beyond-this-point-the-temperature-of-the-cell-rises-rapidly-andthe-cell-in-reality-resolves-itself-image370169062.html
. Storage battery manual, including principles of storage battery construction and design, with the application of storage of batteries to the naval service . is a rapid increase in the cell voltage, until 2.65to 2.70 volts is reached and at which time there is practically no lead-sulphateremaining in the plates, hence the specific gravity of the electrolyte ceases torise and the cell may be said to be in a fully charged condition. If the charge The Lead-Acid Storage Battery Cell 11 be continued beyond this point the temperature of the cell rises rapidly andthe cell in reality resolves itself Stock Photohttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/storage-battery-manual-including-principles-of-storage-battery-construction-and-design-with-the-application-of-storage-of-batteries-to-the-naval-service-is-a-rapid-increase-in-the-cell-voltage-until-265to-270-volts-is-reached-and-at-which-time-there-is-practically-no-lead-sulphateremaining-in-the-plates-hence-the-specific-gravity-of-the-electrolyte-ceases-torise-and-the-cell-may-be-said-to-be-in-a-fully-charged-condition-if-the-charge-the-lead-acid-storage-battery-cell-11-be-continued-beyond-this-point-the-temperature-of-the-cell-rises-rapidly-andthe-cell-in-reality-resolves-itself-image370169062.htmlRM2CE6JBJ–. Storage battery manual, including principles of storage battery construction and design, with the application of storage of batteries to the naval service . is a rapid increase in the cell voltage, until 2.65to 2.70 volts is reached and at which time there is practically no lead-sulphateremaining in the plates, hence the specific gravity of the electrolyte ceases torise and the cell may be said to be in a fully charged condition. If the charge The Lead-Acid Storage Battery Cell 11 be continued beyond this point the temperature of the cell rises rapidly andthe cell in reality resolves itself
 . Storage battery manual, including principles of storage battery construction and design, with the application of storage of batteries to the naval service . stakable signs of chlorine gas. Reduction- in capacity.Battery will not hold charge.Abnormal heating of cell. Frozen slush formed on the sur-face of electrolyte. Due to allow-ing battery to stand in dischargedcondition in freezing weather. Visual inspection. Caused by allowing acid to slop onthe wood parts. How rectified. Give battery a treatingcharge, then add acid as neces-sary to bring specific gravity torequired value. Reduce the spe Stock Photohttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/storage-battery-manual-including-principles-of-storage-battery-construction-and-design-with-the-application-of-storage-of-batteries-to-the-naval-service-stakable-signs-of-chlorine-gas-reduction-in-capacitybattery-will-not-hold-chargeabnormal-heating-of-cell-frozen-slush-formed-on-the-sur-face-of-electrolyte-due-to-allow-ing-battery-to-stand-in-dischargedcondition-in-freezing-weather-visual-inspection-caused-by-allowing-acid-to-slop-onthe-wood-parts-how-rectified-give-battery-a-treatingcharge-then-add-acid-as-neces-sary-to-bring-specific-gravity-torequired-value-reduce-the-spe-image370164064.html
. Storage battery manual, including principles of storage battery construction and design, with the application of storage of batteries to the naval service . stakable signs of chlorine gas. Reduction- in capacity.Battery will not hold charge.Abnormal heating of cell. Frozen slush formed on the sur-face of electrolyte. Due to allow-ing battery to stand in dischargedcondition in freezing weather. Visual inspection. Caused by allowing acid to slop onthe wood parts. How rectified. Give battery a treatingcharge, then add acid as neces-sary to bring specific gravity torequired value. Reduce the spe Stock Photohttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/storage-battery-manual-including-principles-of-storage-battery-construction-and-design-with-the-application-of-storage-of-batteries-to-the-naval-service-stakable-signs-of-chlorine-gas-reduction-in-capacitybattery-will-not-hold-chargeabnormal-heating-of-cell-frozen-slush-formed-on-the-sur-face-of-electrolyte-due-to-allow-ing-battery-to-stand-in-dischargedcondition-in-freezing-weather-visual-inspection-caused-by-allowing-acid-to-slop-onthe-wood-parts-how-rectified-give-battery-a-treatingcharge-then-add-acid-as-neces-sary-to-bring-specific-gravity-torequired-value-reduce-the-spe-image370164064.htmlRM2CE6C14–. Storage battery manual, including principles of storage battery construction and design, with the application of storage of batteries to the naval service . stakable signs of chlorine gas. Reduction- in capacity.Battery will not hold charge.Abnormal heating of cell. Frozen slush formed on the sur-face of electrolyte. Due to allow-ing battery to stand in dischargedcondition in freezing weather. Visual inspection. Caused by allowing acid to slop onthe wood parts. How rectified. Give battery a treatingcharge, then add acid as neces-sary to bring specific gravity torequired value. Reduce the spe
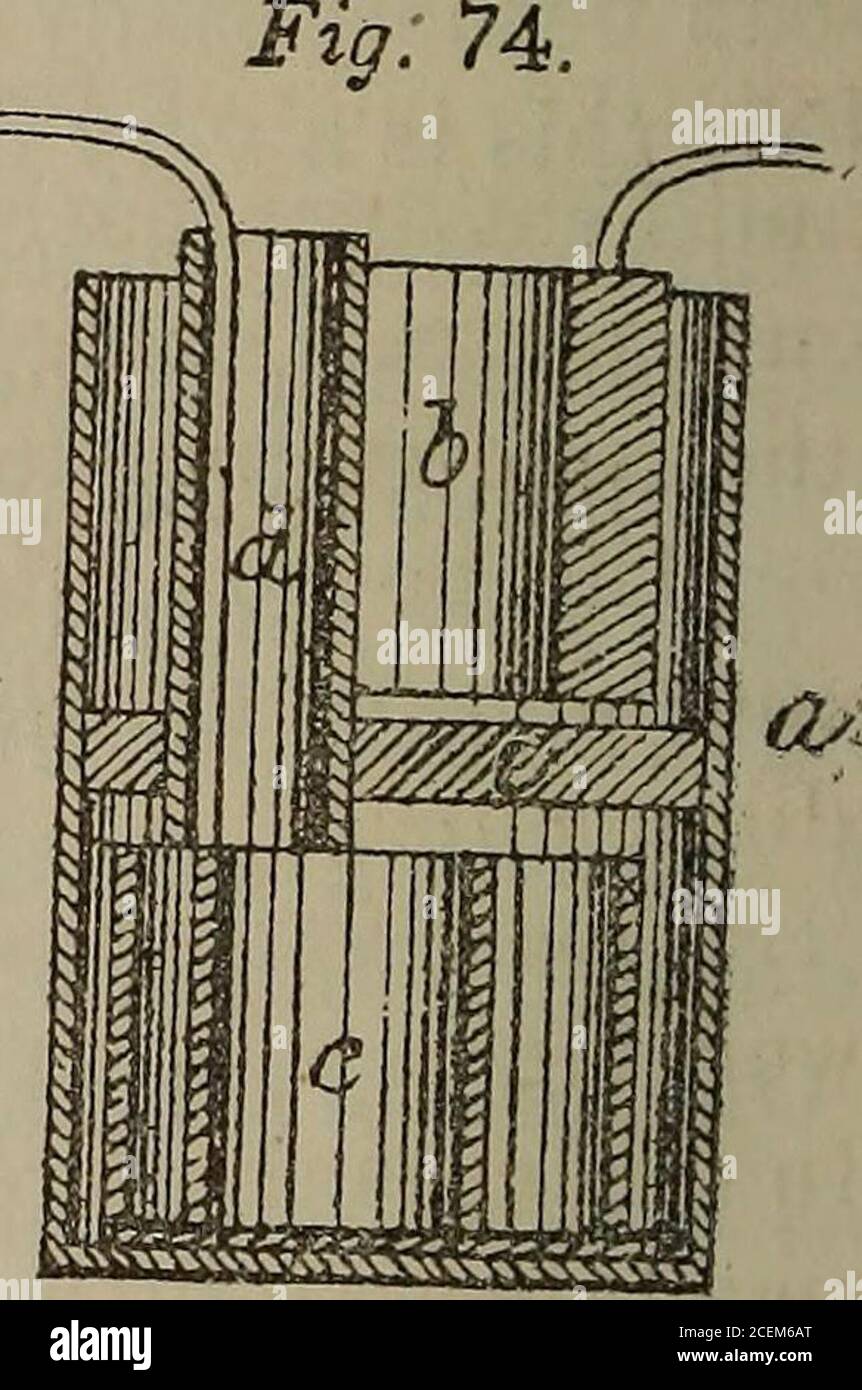 . Notes on torpedoes, offensive and defensive. up through a glazed earthenware cylinder, (d;) (e)is a porous diaphragm consisting of several thicknesses offlannel, fitting tightly round the glazed cylinder (d) andcompletely filling up the space between it and the outer cell(a.) This flannel diaphragm is the chief peculiarity of thisform of battery; it supplies the place of the porous cell inthe other combinations of Daniell, and is so placed that,taking advantage of the greater specific gravity of a solu-tion of sulphate of copper over a solution of sulphate ofzinc, each metal may be, to a gre Stock Photohttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/notes-on-torpedoes-offensive-and-defensive-up-through-a-glazed-earthenware-cylinder-d-eis-a-porous-diaphragm-consisting-of-several-thicknesses-offlannel-fitting-tightly-round-the-glazed-cylinder-d-andcompletely-filling-up-the-space-between-it-and-the-outer-cella-this-flannel-diaphragm-is-the-chief-peculiarity-of-thisform-of-battery-it-supplies-the-place-of-the-porous-cell-inthe-other-combinations-of-daniell-and-is-so-placed-thattaking-advantage-of-the-greater-specific-gravity-of-a-solu-tion-of-sulphate-of-copper-over-a-solution-of-sulphate-ofzinc-each-metal-may-be-to-a-gre-image370466960.html
. Notes on torpedoes, offensive and defensive. up through a glazed earthenware cylinder, (d;) (e)is a porous diaphragm consisting of several thicknesses offlannel, fitting tightly round the glazed cylinder (d) andcompletely filling up the space between it and the outer cell(a.) This flannel diaphragm is the chief peculiarity of thisform of battery; it supplies the place of the porous cell inthe other combinations of Daniell, and is so placed that,taking advantage of the greater specific gravity of a solu-tion of sulphate of copper over a solution of sulphate ofzinc, each metal may be, to a gre Stock Photohttps://www.alamy.com/image-license-details/?v=1https://www.alamy.com/notes-on-torpedoes-offensive-and-defensive-up-through-a-glazed-earthenware-cylinder-d-eis-a-porous-diaphragm-consisting-of-several-thicknesses-offlannel-fitting-tightly-round-the-glazed-cylinder-d-andcompletely-filling-up-the-space-between-it-and-the-outer-cella-this-flannel-diaphragm-is-the-chief-peculiarity-of-thisform-of-battery-it-supplies-the-place-of-the-porous-cell-inthe-other-combinations-of-daniell-and-is-so-placed-thattaking-advantage-of-the-greater-specific-gravity-of-a-solu-tion-of-sulphate-of-copper-over-a-solution-of-sulphate-ofzinc-each-metal-may-be-to-a-gre-image370466960.htmlRM2CEM6AT–. Notes on torpedoes, offensive and defensive. up through a glazed earthenware cylinder, (d;) (e)is a porous diaphragm consisting of several thicknesses offlannel, fitting tightly round the glazed cylinder (d) andcompletely filling up the space between it and the outer cell(a.) This flannel diaphragm is the chief peculiarity of thisform of battery; it supplies the place of the porous cell inthe other combinations of Daniell, and is so placed that,taking advantage of the greater specific gravity of a solu-tion of sulphate of copper over a solution of sulphate ofzinc, each metal may be, to a gre